![]()
Cell Tracking Dyes: How to Label and Follow Cells Over Time
04-24-2026
Cell staining for follow-up studies is not only about making a cell population visible at one moment in time. In many experiments, researchers need to know where labeled cells move, whether they remain distinguishable after co-culture, whether they can still be identified after repeated observation, and whether a fluorescent label continues to report cell identity without being mistaken for background or transferred signal. This is the practical role of cell tracking dyes. Rather than acting as short-lived decorative stains, they are used to mark a selected cell population so that its position, behavior, and persistence can be followed across a defined observation window. In real workflows, this may involve tracking migration, distinguishing one population from another, monitoring labeled cells in a mixed system, or preserving cell identity during downstream imaging and phenotypic analysis.
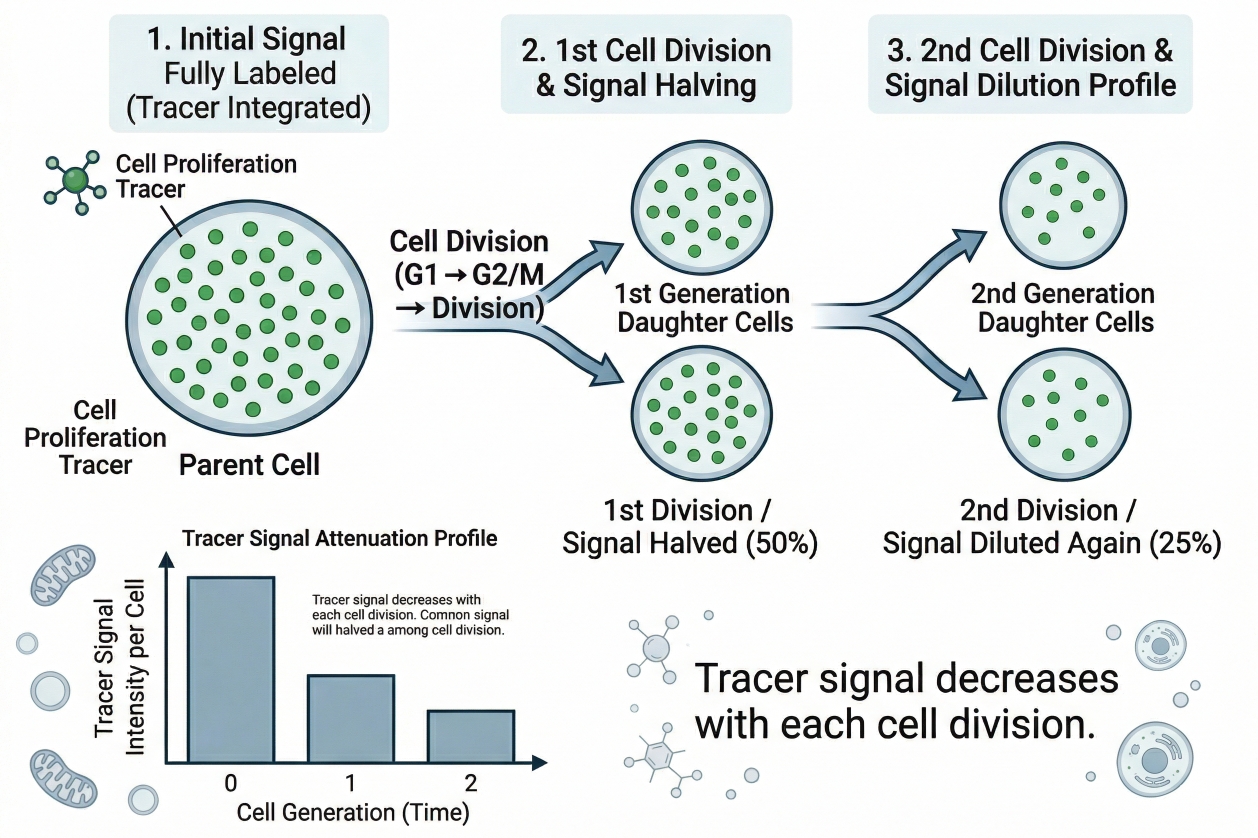
Cell Proliferation Tracer: Principles, Fluorescent Staining, and Dye Selection
04-24-2026
Cell staining for proliferation analysis is not only about making cells visible. In many studies, researchers need to know whether cells are actually dividing, how many rounds of division have occurred, whether only part of a population is proliferating, and whether that behavior changes after stimulation, treatment, co-culture, or environmental shift. This is where a cell proliferation tracer becomes especially useful. Unlike a general fluorescent label that mainly reports location or short-term cell status, a proliferation tracer is designed to support division tracking over time. In practice, that means the fluorescent signal must be introduced in a way that is stable enough to remain associated with the labeled cell population, yet interpretable enough to reveal how that signal changes as cells divide.
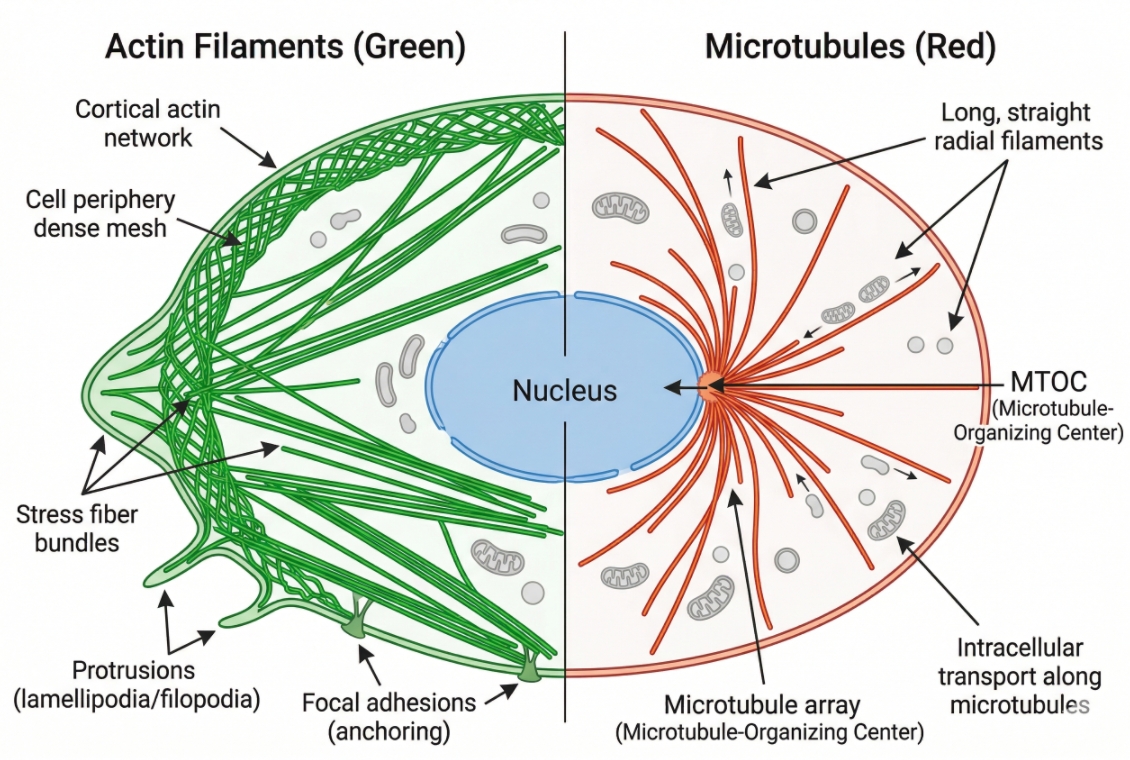
Cytoskeleton Staining: Principles, Probe Selection, and Workflow Design
04-24-2026
Cell staining of the cytoskeleton is one of the most informative approaches in fluorescence-based cell analysis because the cytoskeleton is not a single decorative structure. It is the architectural framework that helps define cell shape, intracellular organization, force distribution, surface protrusions, division-related remodeling, and how cells respond to their local environment. In many experiments, researchers are not simply trying to make filaments visible. They want to understand whether actin remains organized into stress fibers, whether cortical actin becomes redistributed, whether microtubules retain a continuous radial network, whether spindle-like structures appear during mitosis, and whether those structural changes are robust enough to support side-by-side interpretation across treatment groups. That is why cytoskeleton staining needs to be planned as a readout strategy rather than treated as a routine add-on channel.
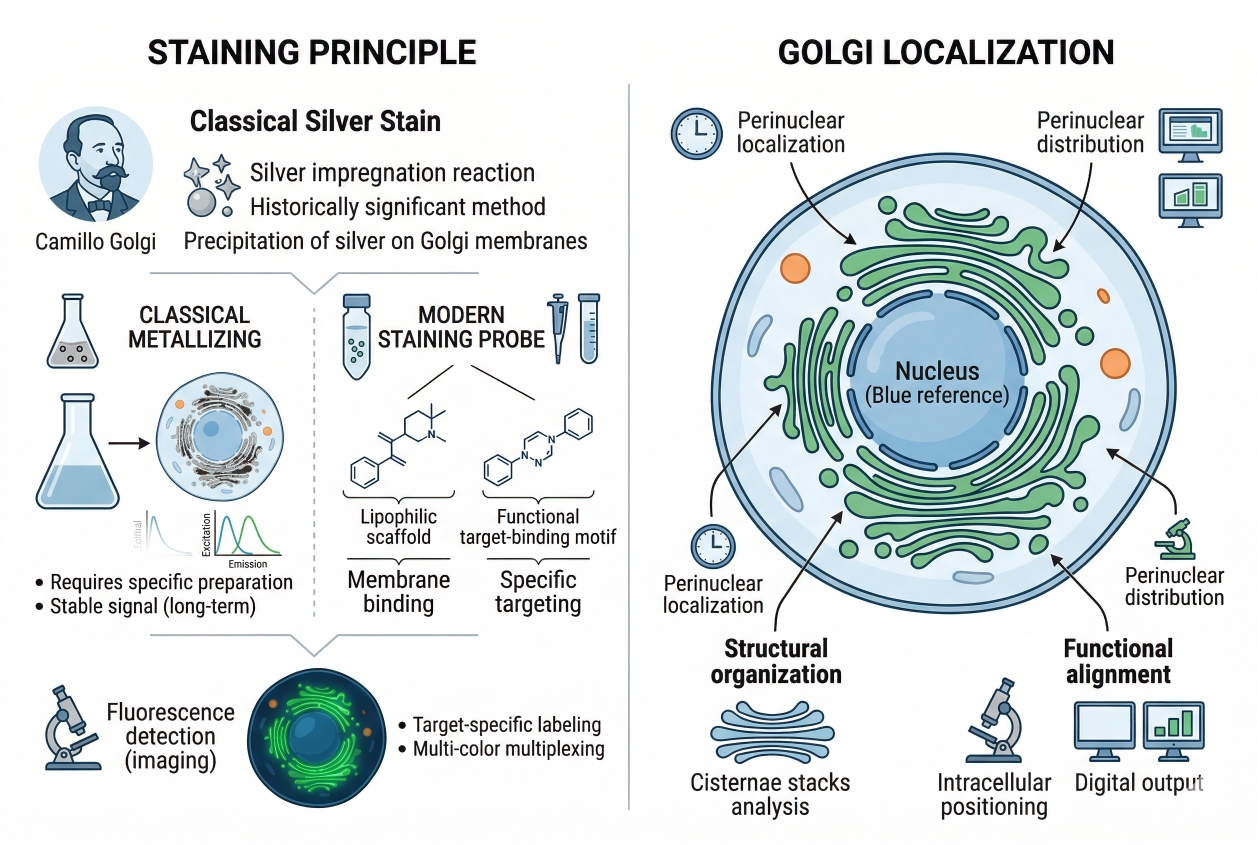
Golgi Fluorescent Staining: Principles, Probe Selection, Workflow, and Troubleshooting
04-24-2026
Cell staining of the Golgi apparatus is widely used in fluorescence-based cell analysis because the Golgi is not just another intracellular landmark. It sits at the center of membrane trafficking, cargo processing, and intracellular organization, so changes in Golgi appearance often carry more interpretive weight than a simple position marker. In many imaging workflows, researchers do not only want to confirm that a Golgi-like structure is present. They want to understand whether the Golgi remains compact and perinuclear, whether it appears dispersed after treatment, whether it can be distinguished cleanly from endoplasmic reticulum-like background, and whether the observed signal supports structural interpretation or a broader trafficking-related conclusion. That is why Golgi fluorescent staining needs to be approached as a workflow decision rather than a purely decorative labeling step. In practice, Golgi staining often becomes most valuable when it is integrated into cell imaging workflows that also include nuclear, ER, lysosomal, membrane, or cytoskeletal channels. Under those conditions, the Golgi channel provides spatial context that can strengthen intracellular interpretation, but only if the staining strategy is matched to the experiment rather than chosen by familiarity alone. Researchers who need a broader comparison of organelle labeling strategies can also explore organelle cell staining, while users evaluating reagent options for this workflow may review golgi fluorescent probes.
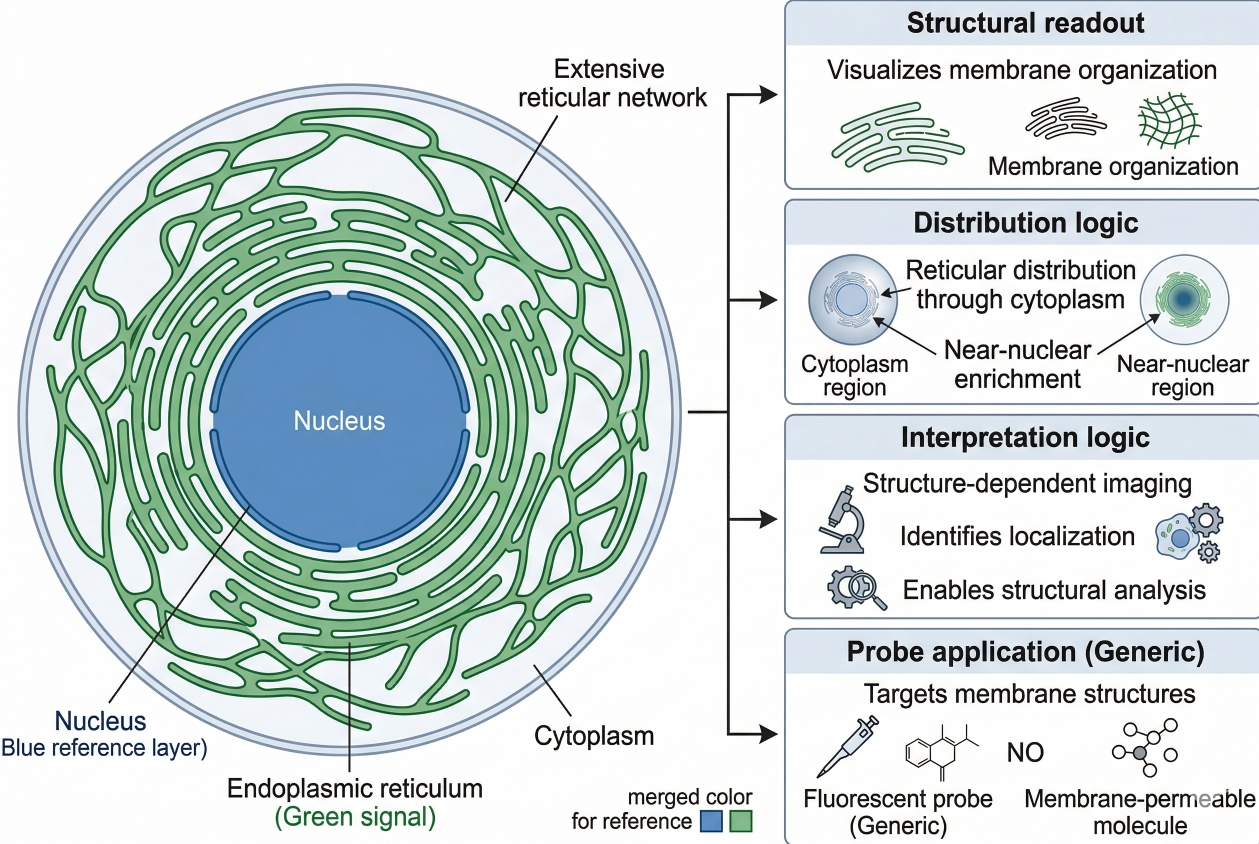
Endoplasmic Reticulum Staining: Principles, Dye Selection, and Workflow Design
04-24-2026
Cell staining of the endoplasmic reticulum is widely used in fluorescence-based cell analysis because ER organization is tightly linked to intracellular architecture, membrane dynamics, organelle interaction, and image interpretation quality. In many experiments, researchers are not simply trying to confirm that the endoplasmic reticulum is present. They want to understand whether the ER forms a fine reticular network, whether the perinuclear region appears dense or expanded, whether sheet-like and tubular features remain distinguishable, whether a treatment alters ER distribution relative to the nucleus, and whether the ER channel can be combined reliably with other fluorescence readouts. That is why endoplasmic reticulum staining is more demanding than routine nuclear labeling or simple membrane counterstaining. A useful ER staining page therefore needs to do more than list dyes. It should explain what ER fluorescence is actually showing, how probe mechanism influences interpretation, why live-cell and fixed-cell workflows should not be treated as interchangeable, and how to build a staining workflow that supports a biologically credible conclusion rather than a visually attractive but ambiguous image.
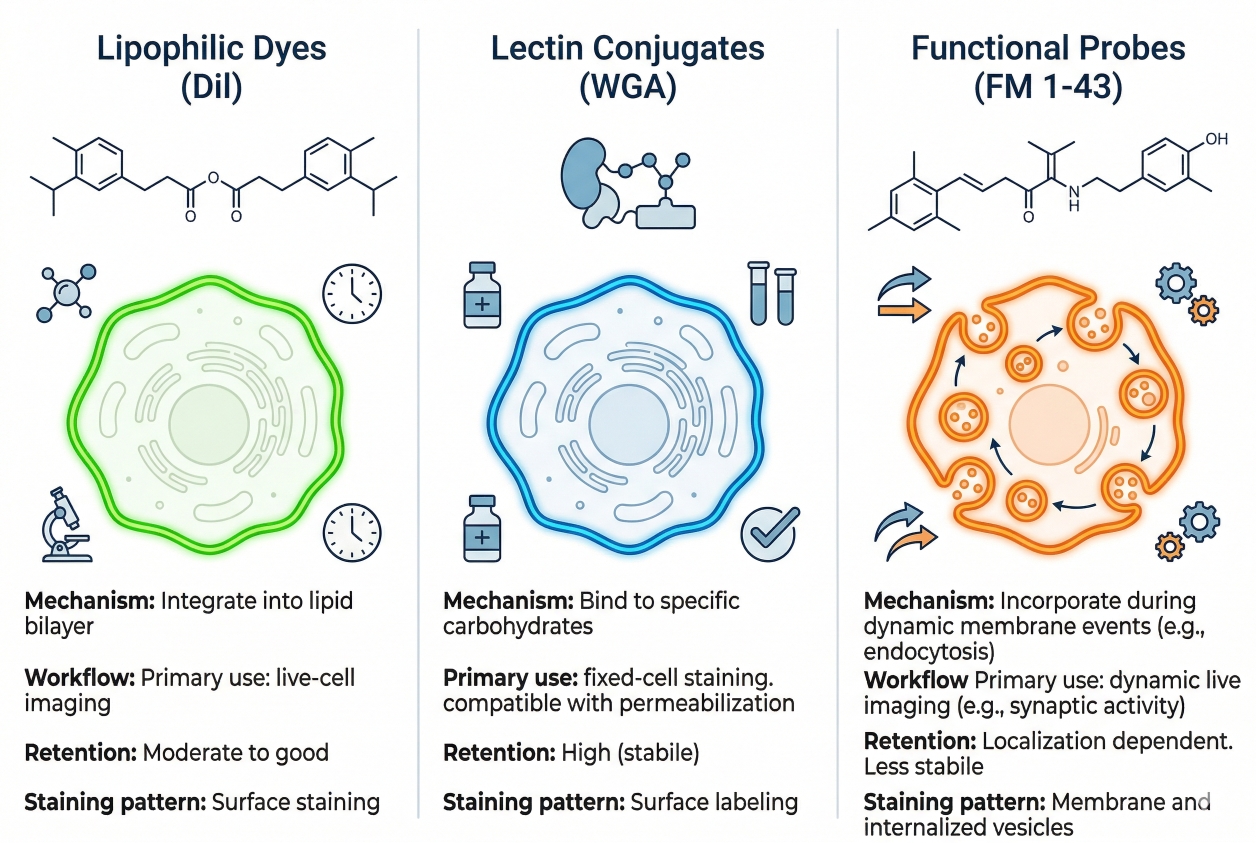
Cell Membrane Staining Guide: Fluorescent Dyes, Applications, and Key Considerations
04-24-2026
Cell membrane staining is widely used in fluorescence-based cell analysis because the plasma membrane provides the most direct visual definition of where one cell ends and another begins. In many imaging workflows, researchers are not only trying to confirm that cells are present. They need to determine whether cell boundaries remain continuous, whether membrane contours appear smooth or irregular, whether cells spread broadly or stay compact, and whether neighboring cells can be separated clearly enough for morphology comparison or image segmentation. This is why membrane staining often becomes a central structural channel rather than a decorative add-on. A membrane stain that produces bright fluorescence but weak boundary logic can mislead downstream interpretation, especially in dense cultures, multicolor imaging panels, and experiments where morphology or cell-cell contact matters. A useful membrane staining page therefore needs to do more than list dye names. It should explain what membrane staining means in fluorescence imaging, how different membrane fluorescent dye classes behave, and how workflow choices affect whether a membrane signal truly supports boundary-level interpretation.
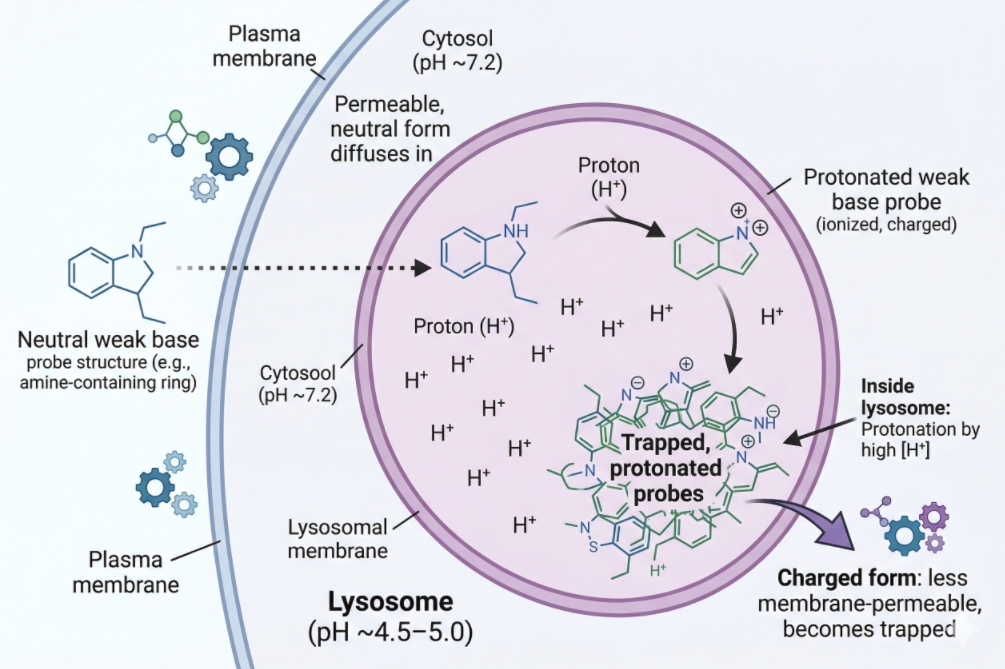
Lysosome Staining in Cells: Choosing Probes and Avoiding Misread Signals
04-24-2026
Lysosome staining is widely used in fluorescence-based cell analysis because lysosomes are central to intracellular degradation, vesicle maturation, cargo turnover, and organelle trafficking. In practice, however, lysosome staining is easy to overinterpret. Many researchers expect a bright punctate lysosomal signal to mean they have specifically labeled lysosomes, when in reality a large fraction of commonly used live-cell lysosome probes accumulate because they partition into acidic compartments. That distinction matters. A punctate acidic-vesicle pattern can be highly useful for comparing treatment groups, tracking vesicle redistribution, or adding organelle context to cell imaging workflows, but it does not automatically prove lysosome identity on its own. A useful lysosome staining page therefore needs to do more than introduce LysoTracker dyes. It should explain what these probes really report, where the interpretation boundaries are, why fixation can distort signal meaning, and how to design a staining workflow that reduces false confidence in acidic-compartment readouts.
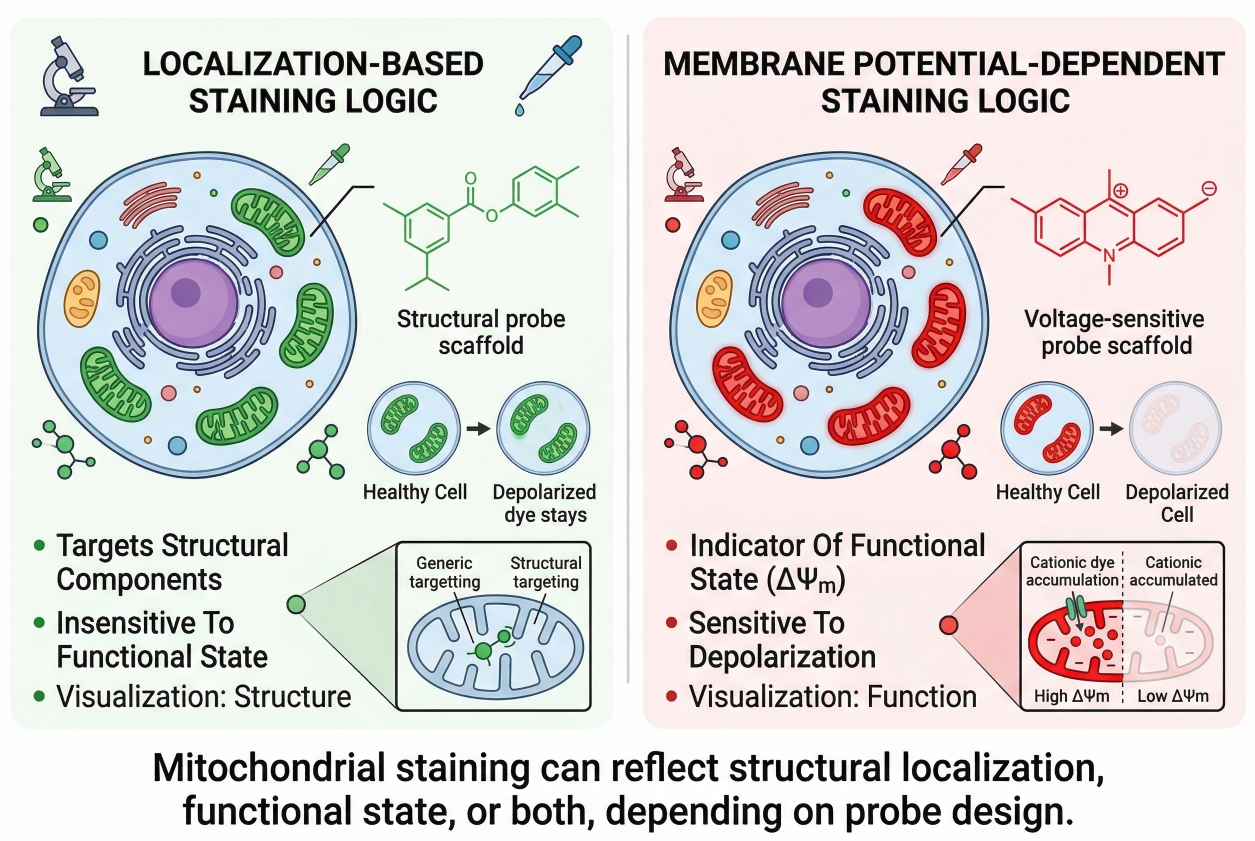
Mitochondrial Staining in Cells: Dyes, Workflow, and Interpretation
04-24-2026
Cell staining of mitochondria is widely used in fluorescence-based cell analysis because mitochondrial organization is closely tied to cellular state, intracellular distribution, and imaging readout quality. In many experiments, researchers do not only want to know whether mitochondria are present. They want to understand how mitochondria are arranged in the cytoplasm, whether the network appears fragmented or elongated, whether signal intensity changes after treatment, and whether those changes should be interpreted as structural redistribution or as a shift in mitochondrial condition. That is why mitochondrial staining is often more demanding than routine labeling of nuclei or plasma membrane. The same experiment may contain both localization information and function-linked signal behavior, and these two layers are not always easy to separate. A dye can appear to “lose mitochondrial staining” even when mitochondria are still present, or it can produce bright signal that looks convincing while actually reflecting non-ideal handling, overload, or interpretation bias. A useful mitochondrial staining page therefore needs to do more than list dyes. It should help researchers understand the logic behind signal generation, the consequences of sample handling, and the workflow choices that determine whether mitochondrial fluorescence will support a reliable biological conclusion.
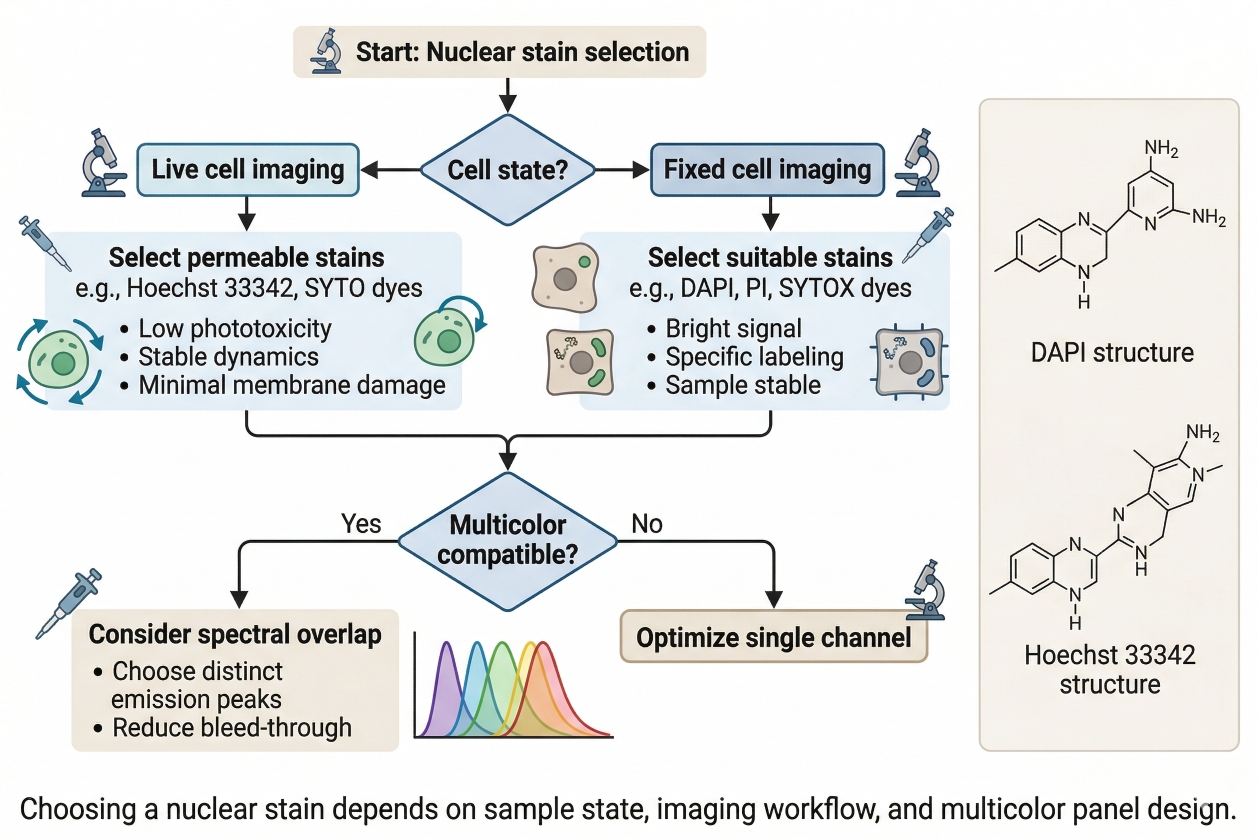
A Practical Guide to Nuclear Staining in Cells: Principles, Selection, and Applications
04-24-2026
Nuclear staining is one of the most widely used techniques in fluorescence-based cell analysis because it solves several experimental needs at the same time. It helps researchers identify cells, define intracellular orientation, support image segmentation, normalize field-to-field comparisons, and interpret how other fluorescent signals are positioned relative to a stable structural reference. In microscopy, a reliable nuclear channel often determines whether downstream organelle or localization data can be interpreted cleanly. In phenotypic screening and comparative imaging, it frequently acts as the analytical anchor that makes multiparameter workflows scalable and reproducible. Even though nuclear staining is common, choosing the right nuclear stain is not a trivial decision. Different dyes vary in permeability, brightness, photostability, background behavior, fixation compatibility, and suitability for multicolor workflows. A stain that works well in fixed-cell endpoint imaging may not be appropriate for live-cell observation. A dye that produces strong nuclear contrast by itself may become a poor option if it occupies a critical channel in a more complex panel. In drug discovery, biotech, and CDMO research settings, where experiments often require strong reproducibility across large datasets or multiple conditions, nuclear staining should be treated as part of workflow design rather than as a routine add-on.
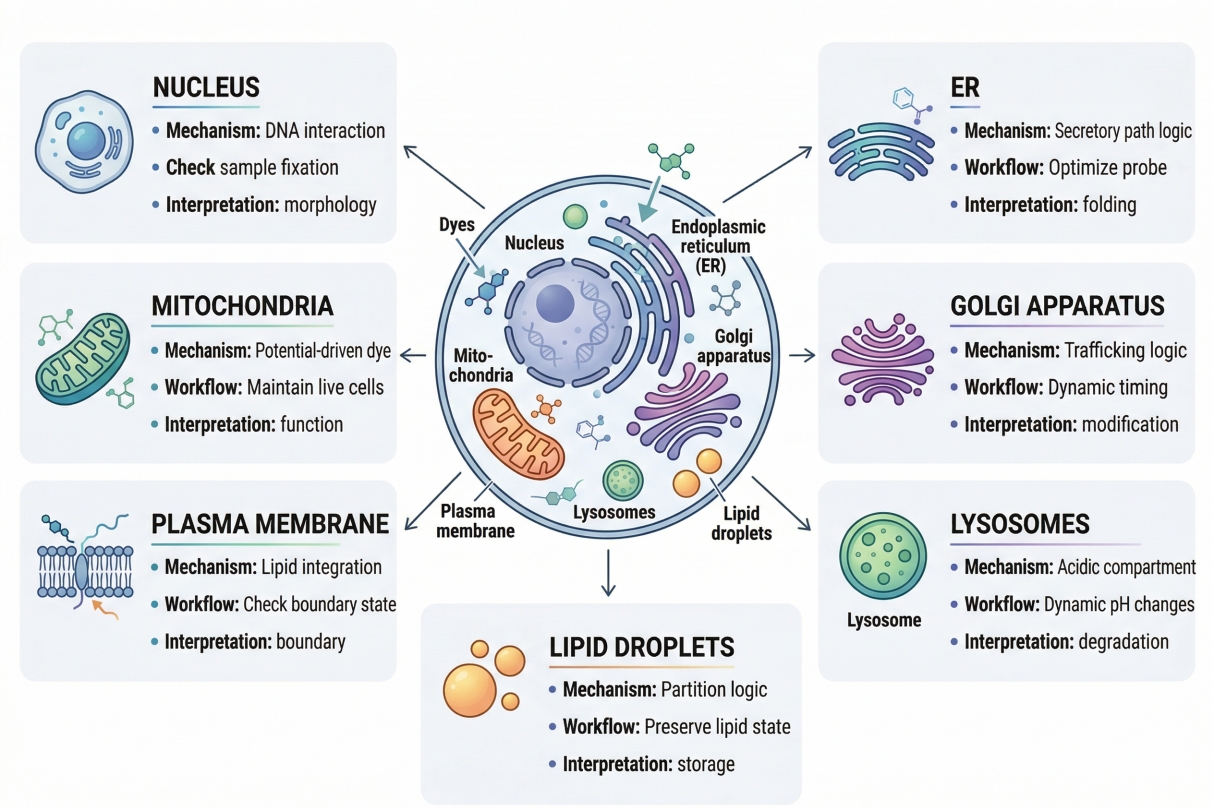
Organelle-Specific Cell Staining: Mitochondria, Nucleus, Membrane and Beyond
04-24-2026
Organelle cell staining is not just a more detailed version of general cell staining. For many researchers, it is the point where a fluorescence experiment becomes genuinely informative. When the question is no longer “Are the cells there?” but rather “What is happening inside the cells?”, broad whole-cell contrast quickly becomes insufficient. Researchers need to know whether mitochondria are redistributed, whether lysosomes are expanded or clustered, whether the plasma membrane remains well defined, whether the nucleus can serve as a reliable reference layer, and whether lipid-associated structures are changing in ways that affect interpretation. In these situations, organelle-specific staining is not a visual preference. It is a decision about analytical precision. This is also why organelle staining is one of the most misunderstood areas in fluorescence workflow design. A stain that appears bright and visually attractive can still fail the real experimental need if it does not reflect the correct compartment, if its signal depends on a functional property that changes during sample handling, or if it cannot be combined reliably with other dyes in a multicolor experiment. Many staining failures happen because researchers choose an organelle stain based on familiarity, color, or a general label claim without fully checking how the stain enters cells, what drives retention, whether it remains interpretable after fixation, and how it behaves alongside other channels. The result may be a strong image but a weak conclusion.
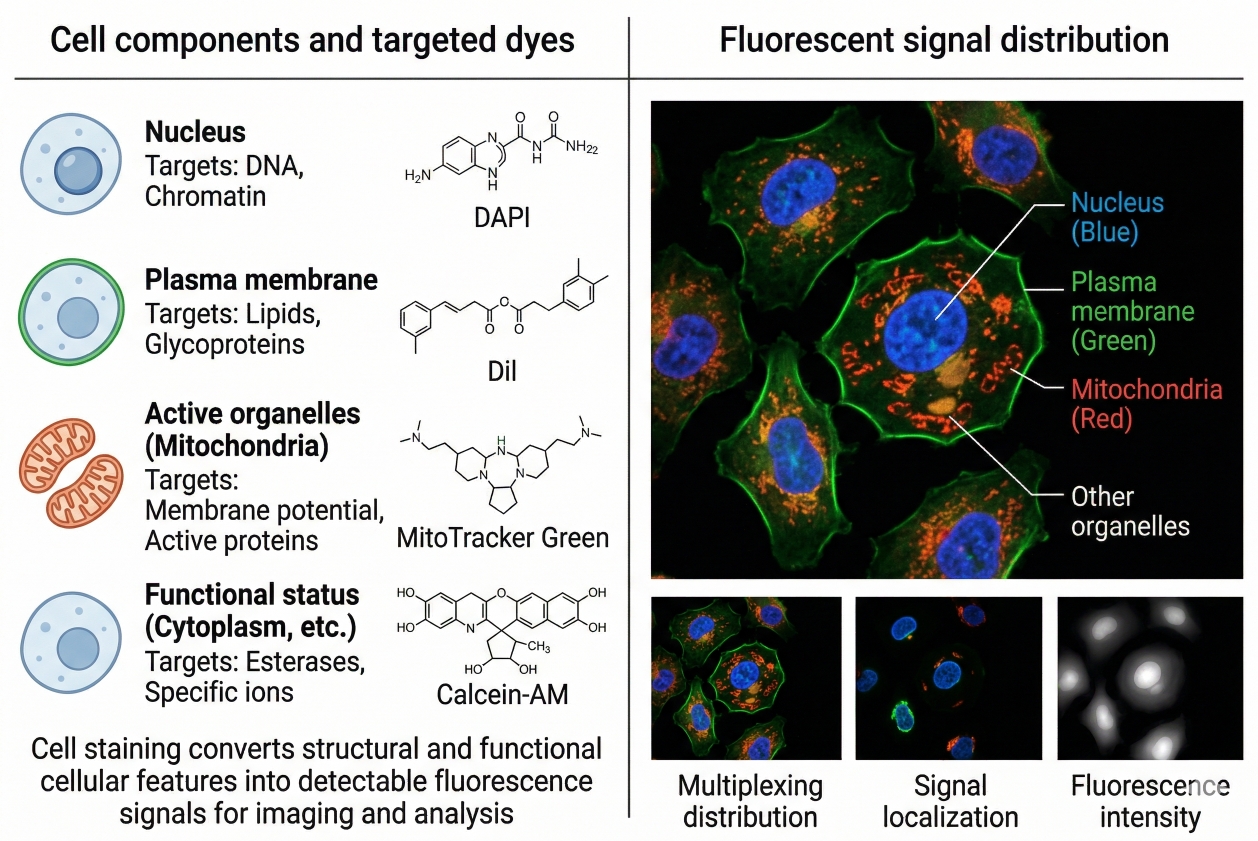
Common Problems in Cell Staining and How to Fix Them
04-24-2026
Cell staining is one of the most widely used approaches in fluorescence-based cell analysis because it turns otherwise difficult-to-see structures, compartments, and cellular states into readable optical signals. But in practice, successful staining depends on far more than adding a dye and taking an image. Sample condition, dye chemistry, buffer composition, incubation time, washing stringency, fixation strategy, and acquisition settings all influence whether the final signal is specific, interpretable, and reproducible. When these variables are not aligned, cell staining can generate high background, weak fluorescence, uneven labeling, misleading co-localization, or signal changes that are incorrectly interpreted as biology. That is why troubleshooting cell staining should never begin with a single assumption such as “the dye concentration must be wrong” or “the microscope settings are bad.” Most staining failures are multi-factorial. A weak signal may be caused by poor target accessibility, but it may also reflect photobleaching, poor sample health, or a dye that is fundamentally mismatched to the workflow. High background may arise from free dye residue, autofluorescence, non-specific staining, or a fixation-related artifact. The most effective way to solve these problems is to understand how the full workflow behaves from sample preparation to readout.
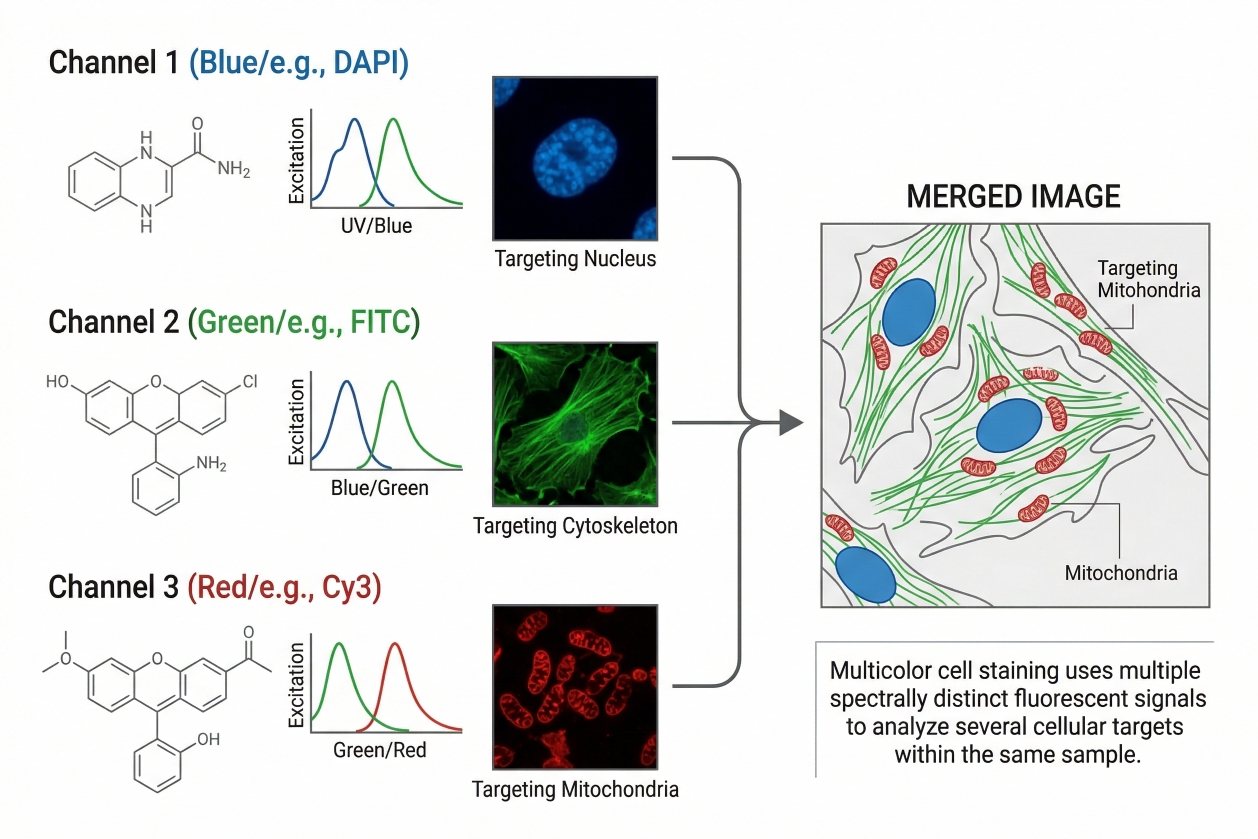
Multicolor Cell Staining: Principles, Dye Selection, and Panel Design
04-24-2026
Multicolor cell staining has become a core strategy in modern fluorescence-based cell analysis because many biological questions cannot be answered by a single stain alone. Researchers often need to examine several cellular features at the same time, such as nuclear position, membrane integrity, organelle distribution, viability status, and the expression or localization of multiple targets within the same sample. By combining fluorophores with distinguishable spectral properties, multicolor staining makes it possible to analyze these signals in parallel rather than through separate experiments that are harder to compare directly. The value of this approach is clear, but so is the difficulty. A multicolor panel can fail even when every individual stain appears reasonable on its own. Spectral overlap, signal spillover, unequal fluorophore brightness, poor control design, autofluorescence, and incompatibility between live-cell and fixed-cell workflows can all undermine interpretation. That is why multicolor cell staining is not simply an extension of single-color staining. It requires a more deliberate framework that connects the biological question, the instrument configuration, the marker hierarchy, and the fluorophore selection strategy.
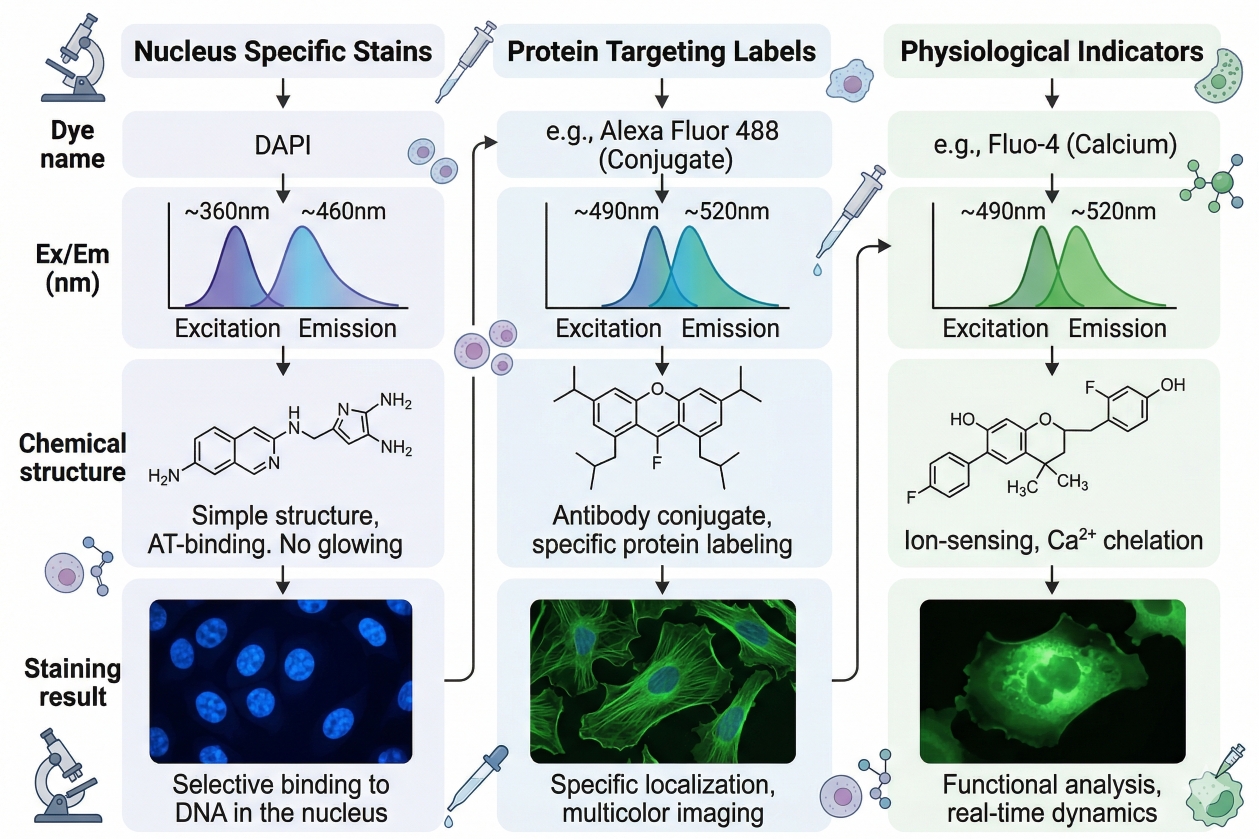
How to Choose the Right Fluorescent Dye for Cell Staining
04-24-2026
Choosing the right fluorescent dye is one of the most important early decisions in any cell staining workflow because the dye determines far more than the visible color in the final image. It shapes signal brightness, background behavior, live-cell compatibility, fixation tolerance, spectral overlap risk, and ultimately the biological meaning of the result. A dye that is bright but poorly matched to the sample or instrument can create misleading fluorescence, unstable localization, or unnecessary troubleshooting. A more suitable dye, even if less visually dramatic at first glance, often delivers clearer interpretation and better reproducibility across experiments. In modern fluorescence-based cell analysis, researchers rarely choose dyes in isolation. They select them in the context of sample type, staining target, readout platform, and whether the experiment emphasizes structural imaging, functional response, viability assessment, organelle localization, or multiparameter analysis. This means dye selection should be treated as a workflow design decision, not a purchasing step at the end of planning. The best dye is the one that fits the biology, the protocol, and the optical system at the same time.
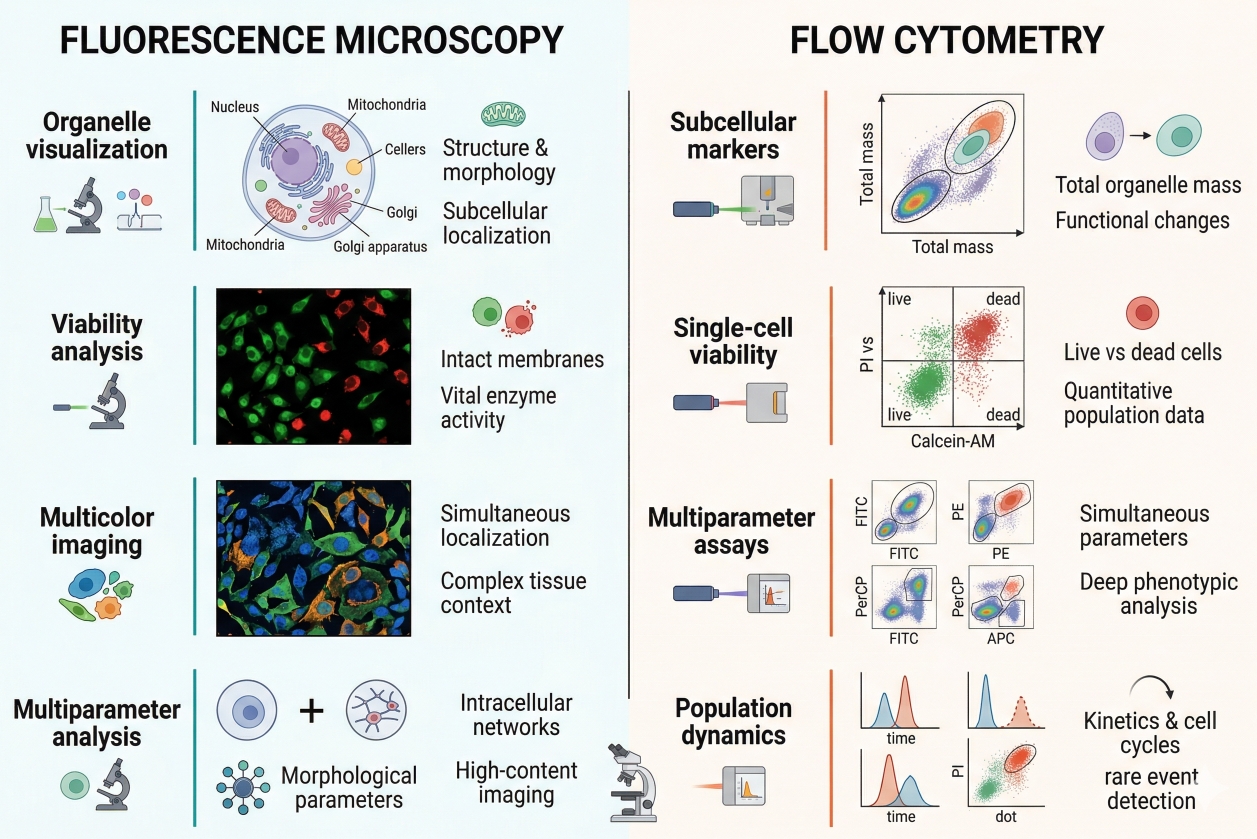
Cell Staining Protocol: Step-by-Step Guide for Reliable Results
04-24-2026
A reliable cell staining workflow begins long before a dye is added to a dish, slide, or tube. The quality of the final signal depends on whether the sample state, staining goal, detection platform, dye chemistry, incubation conditions, washing logic, and image acquisition settings were aligned from the start. When these elements are planned well, cell staining can reveal cellular structure, organelle localization, membrane behavior, viability state, or population-level differences with strong interpretability. When they are mismatched, even bright fluorescence can produce misleading images, unstable signal, or poor reproducibility. This page is designed as a practical protocol guide rather than a narrow recipe for one stain. Different experiments require different dyes, buffers, fixation strategies, and acquisition approaches, so a useful protocol page should explain the logic of each step and show how researchers can adjust the workflow without losing analytical quality. That is especially important in fluorescence-based assays, where signal strength alone is never enough. A technically successful result must also preserve biological meaning, minimize background, and remain compatible with the readout platform.
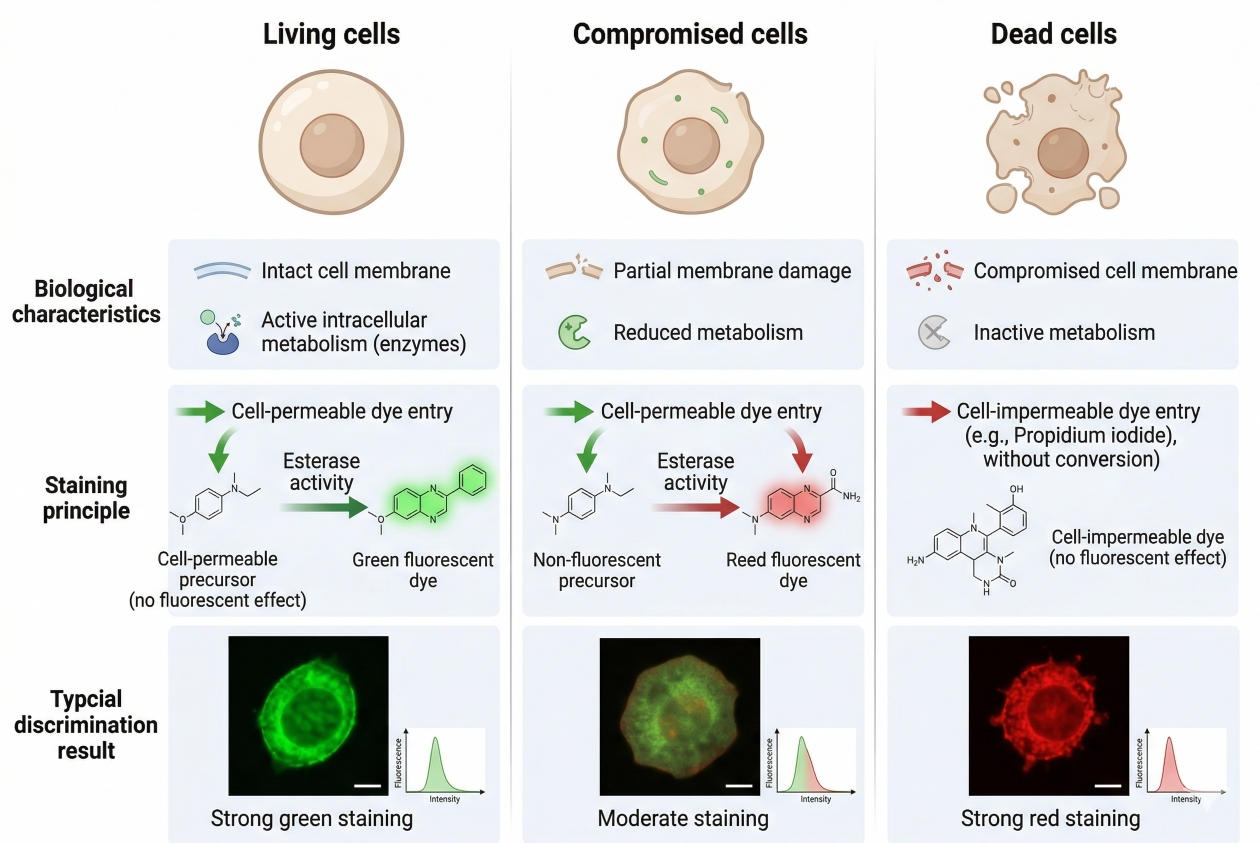
Cell Viability Staining: Fluorescent Methods to Distinguish Live and Dead Cells
04-24-2026
Cell staining workflows often depend on one basic question before any target-specific analysis begins: are the cells alive, dead, or in a compromised transitional state that could distort the readout? That is why cell viability staining is one of the most important control layers in fluorescence-based experiments. A viability stain does more than separate intact cells from damaged cells. It helps researchers judge sample quality, improve data interpretation, reduce false positives from non-specific fluorescence, and decide whether a downstream image, population analysis, or multicolor panel is biologically meaningful. In practical terms, viability information often determines whether the rest of the experiment can be trusted.
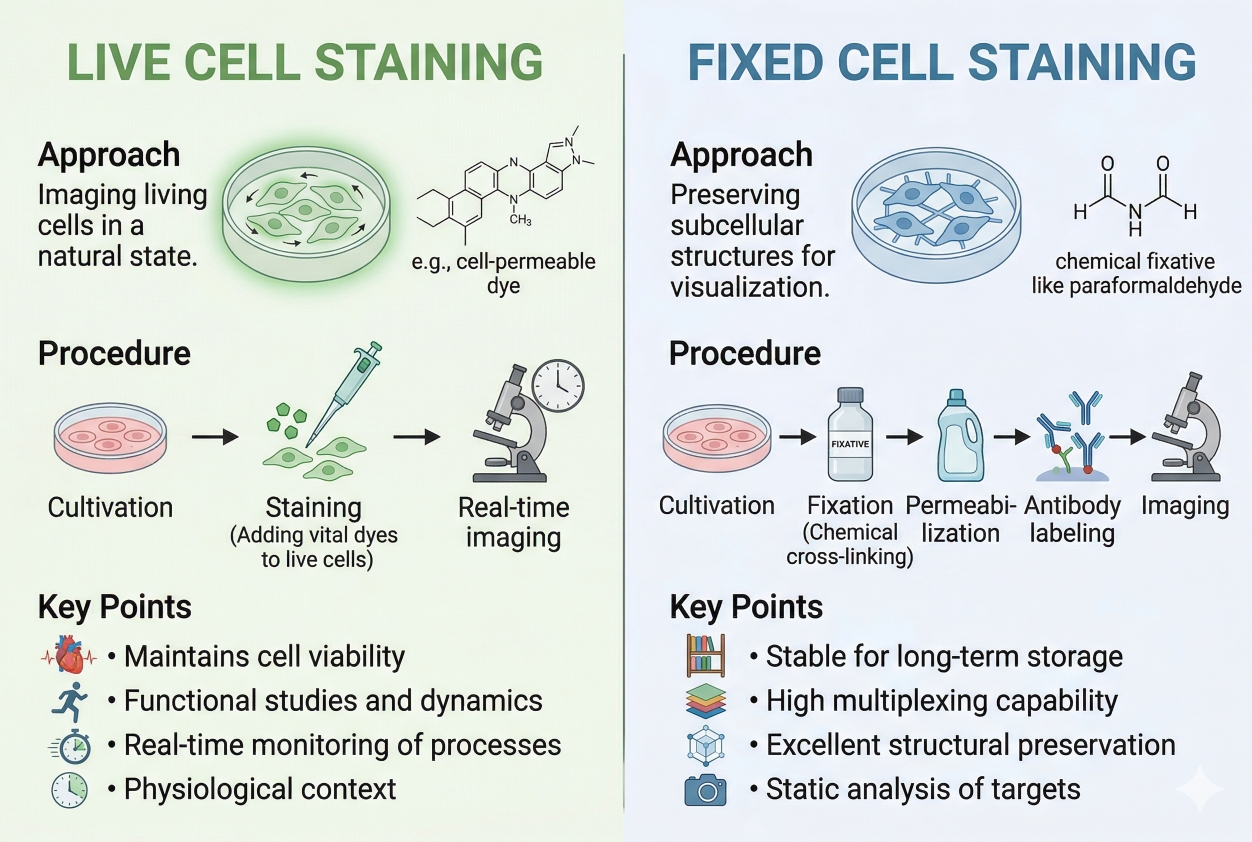
Live Cell Staining vs Fixed Cell Staining: How to Choose the Right Strategy
04-24-2026
Choosing between live cell staining and fixed cell staining is one of the most important early decisions in a fluorescence experiment because it affects what kind of biology you can observe, which probes or antibodies will work, how stable the signal will be, and how reproducible the final data will be. Live cell staining is designed for researchers who need to follow dynamic processes in real time, such as membrane trafficking, mitochondrial behavior, organelle movement, or short-term changes in cell state. Fixed cell staining, by contrast, is usually the better fit when the priority is structural preservation, long acquisition windows, intracellular target accessibility, or a more controlled endpoint workflow. In practice, this is not simply a technical preference. It is a strategy choice that shapes sample handling, dye selection, imaging setup, control design, and downstream interpretation.
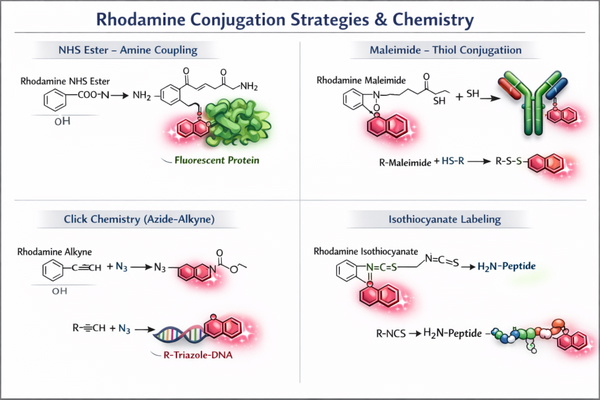
Rhodamine Labeling Strategies Designed for Your Specific Biomolecules
12-25-2025
Rhodamine labeling, as a key fluorescence labeling technique for biomolecules, has become an indispensable tool in cell imaging, protein localization, nucleic acid analysis, and molecular interaction studies due to its high brightness, excellent photostability, and broad applicability. Through customized Rhodamine labeling strategies tailored for proteins, antibodies, nucleic acids, peptides, and small-molecule probes, researchers can achieve high-sensitivity detection, precise visualization, and multichannel colocalization analysis, enabling in-depth investigation of dynamic processes and molecular mechanisms within complex biological systems.
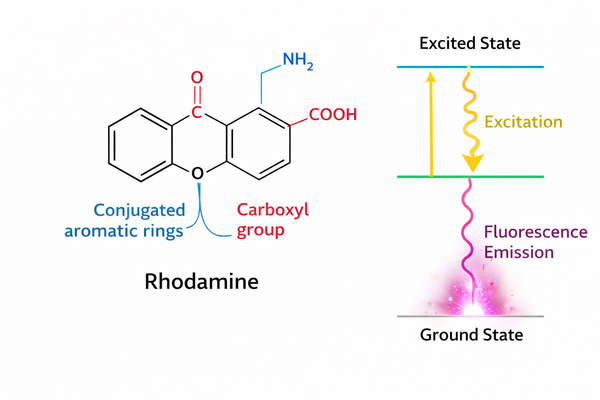
Rhodamine Spectral Properties and Fluorescence Mechanism Explained for Research Use
12-25-2025
Rhodamine dyes, as core fluorophores in bioanalytical and optical imaging applications, have become one of the most widely used fluorescent probe families in chemical biology research. This is due to their excellent molar extinction coefficients, extremely high quantum yields, and outstanding photophysical stability in complex physiological environments. These dyes are based on the rigid xanthene core structure, which effectively suppresses non-radiative energy dissipation, enabling high-brightness photon emission in the excited state. A deep understanding of rhodamine’s spectral properties and the underlying electronic transition mechanisms is critically important for researchers to precisely control experimental parameters, develop novel fluorescent sensors, and enhance the sensitivity of biomolecular detection.
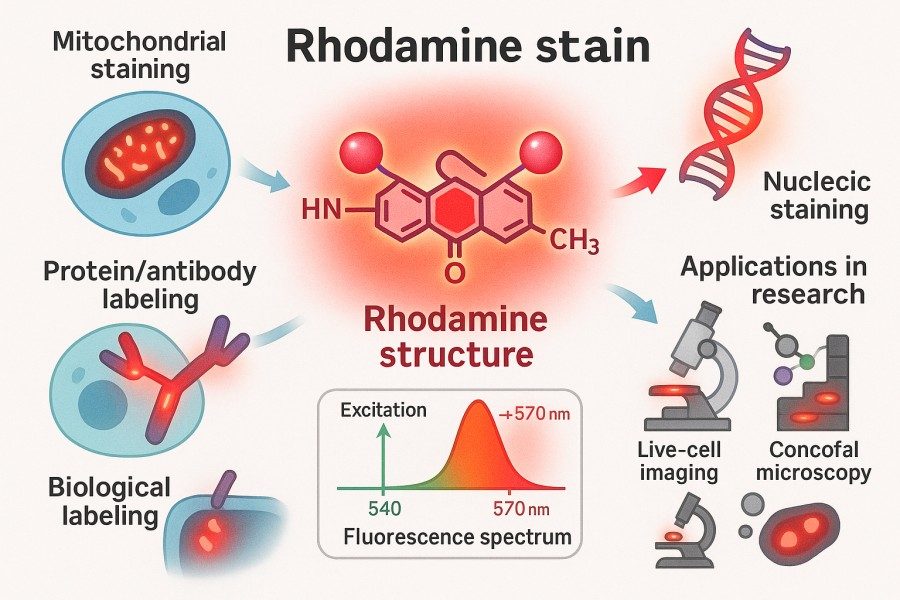
Rhodamine vs Other Fluorophores: How to Choose the Best Dye?
10-31-2025
Rhodamine, one of the most representative fluorescent dyes in life science research, is highly favored for its exceptional brightness, outstanding photostability, and extensive range of derivatives. Its unique chemical structure allows versatile functional modifications, enabling efficient conjugation with proteins, nucleic acids, and small molecules. Rhodamine dyes are widely applied in cell imaging, flow cytometry, and in vivo fluorescence tracking. With the continuous advancement of fluorescence technologies, other dyes such as Fluorescein, Cyanine, Alexa Fluor, and BODIPY have also demonstrated distinct advantages in terms of performance and application. Given the wide variety of fluorescent dyes available, researchers must evaluate multiple parameters—including excitation and emission wavelengths, quantum yield, photostability, and biocompatibility—to select the most suitable dye for their experimental needs. This article provides an in-depth comparison of Rhodamine and other major fluorescent dyes, offering a scientific guide to dye selection and helping researchers achieve higher signal intensity and detection precision across diverse experimental systems.
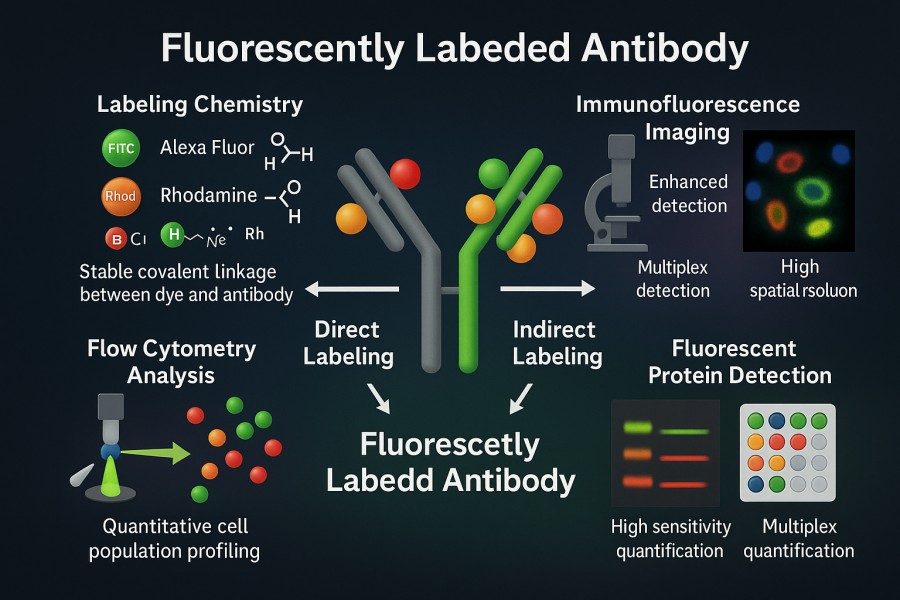
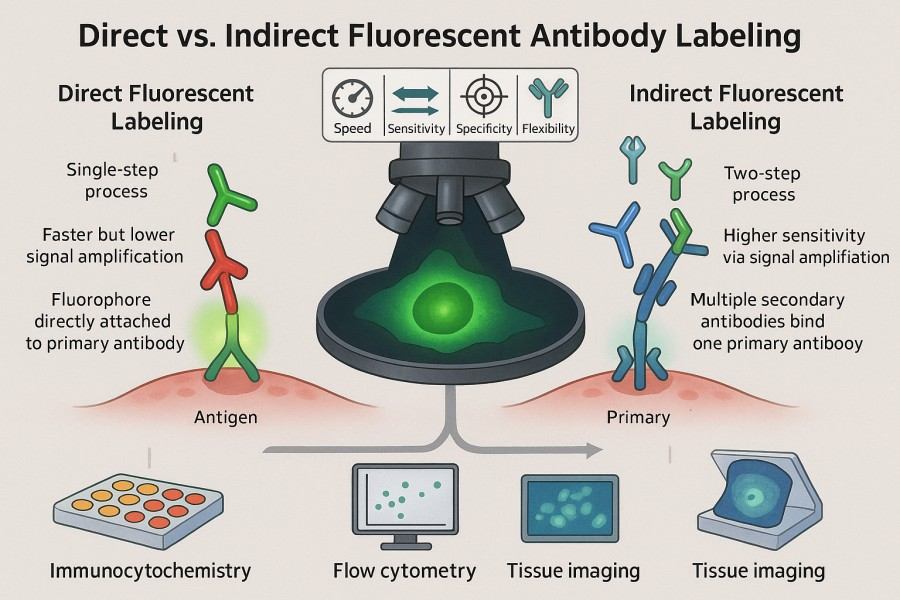
Direct vs. Indirect Fluorescent Antibody Labeling: Which Is Right for Your Research?
10-31-2025
Fluorescent labeled antibodies have become indispensable tools in modern life science research, offering high sensitivity and visualization capabilities for protein detection, cell imaging, and multiplexed analysis. Whether in basic research or clinical diagnostics, scientists rely on these labeled antibodies for accurate identification and quantification of target molecules. In experimental design, choosing between direct fluorescent antibody labeling and indirect fluorescent antibody labeling is a critical decision, as each approach presents distinct advantages and limitations in terms of sensitivity, specificity, operational complexity, and signal stability. By thoroughly understanding the principles and applications of both methods, researchers can optimize their strategies to achieve more efficient and reliable protein detection and visualization outcomes.
A Practical Guide to Fluorescent Labeling of Antibodies: Principles, Challenges, and Applications
10-31-2025
Antibody labeling, as a core technology in modern biological research and diagnostic experiments, enables highly sensitive detection and visualization of target proteins by coupling specific antibodies with fluorescent dyes. Fluorescently labeled antibodies not only reveal the spatial distribution and dynamic changes of proteins inside and outside cells but are also widely applied in flow cytometry, fluorescence microscopy imaging, Western blotting, and high-throughput screening platforms. With the advancement of multicolor labeling techniques and high-performance fluorescent dyes, researchers can analyze multiple target molecules simultaneously within a single experiment, greatly enhancing experimental efficiency and data reliability.
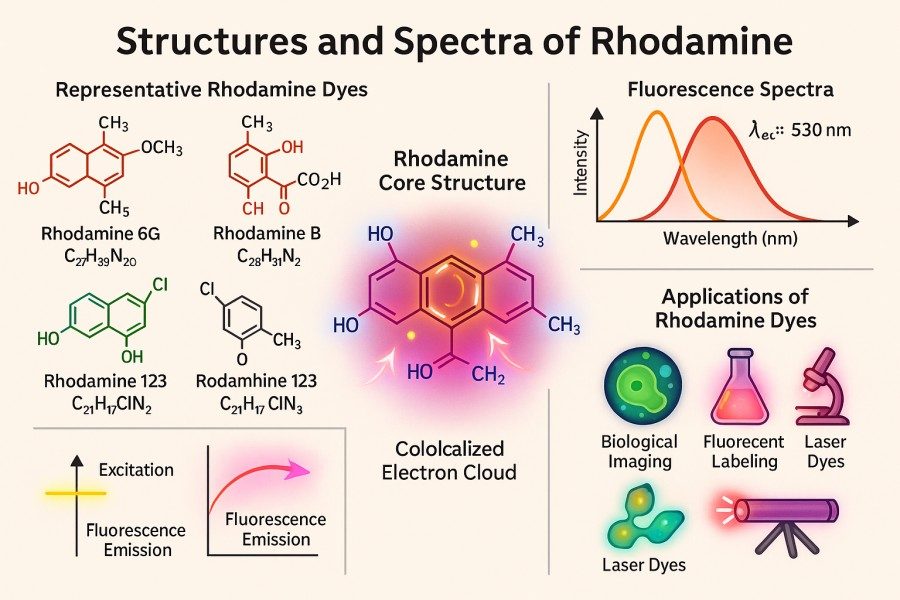
Comparing Rhodamine Derivatives: Rhodamine 6G, Rhodamine B, Rhodamine 123 and More
10-31-2025
Rhodamine dyes, as a class of classic fluorescent dyes, are widely used in fields such as bioimaging, flow cytometry, sensing probes, and materials science due to their high brightness, excellent photostability, and versatile chemical modification capabilities. With the increasing demands of modern research, scientists are paying more attention to the differences among various rhodamine derivatives in terms of spectral properties, labeling efficiency, and biocompatibility. This article systematically compares commonly used rhodamine derivatives, including Rhodamine 6G, Rhodamine B, Rhodamine 123, and other important derivatives, providing in-depth analysis on structural features, application scenarios, advantages and limitations, and experimental considerations, offering authoritative guidance and practical reference for researchers selecting and applying rhodamine dyes.
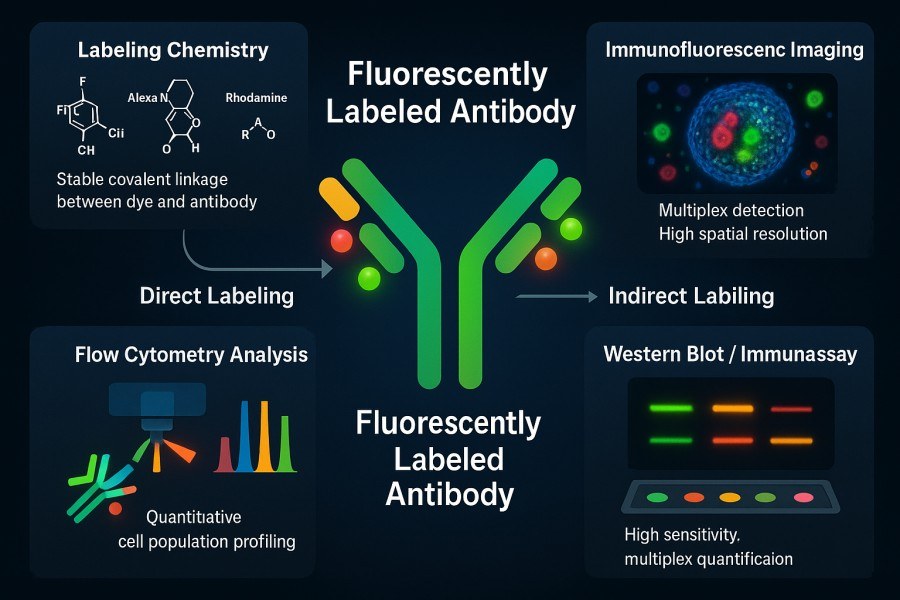
How to Select the Best Fluorophore for Reliable Antibody Labeling Results?
10-31-2025
Antibody labeling is an indispensable tool in modern life science research and diagnostic technologies. By conjugating antibodies with fluorophores, it enables highly sensitive and specific detection and visualization of target molecules. Choosing the right fluorophore not only affects signal intensity and image quality but also directly impacts the reliability of experiments and the reproducibility of data. In complex applications such as multicolor imaging, flow cytometry, or in vivo imaging, factors like excitation/emission wavelengths, photostability, brightness, and chemical compatibility of different fluorophores can significantly influence experimental outcomes. Therefore, scientifically evaluating fluorophore properties and matching them to experimental requirements is crucial for achieving high-quality antibody labeling results.
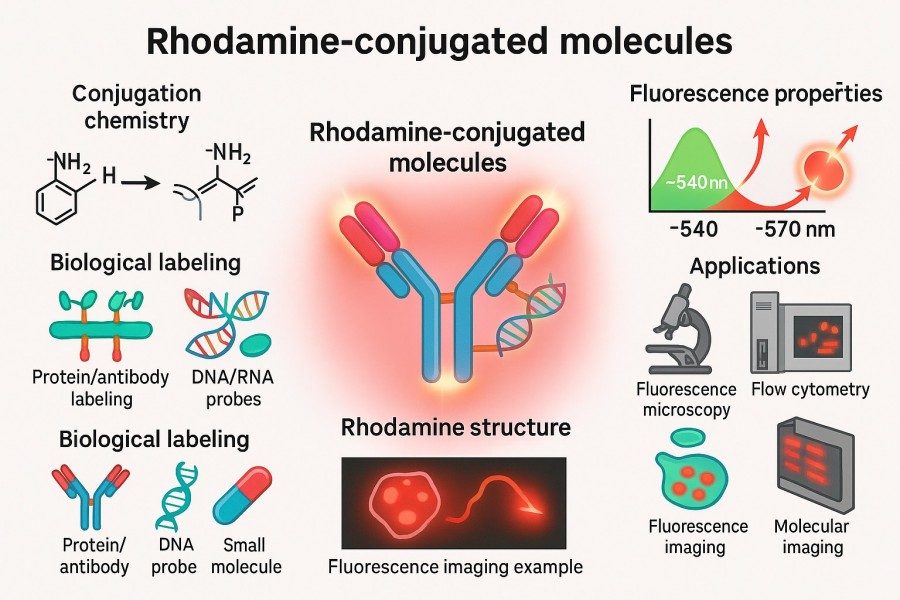
How to Optimize Rhodamine Conjugation Efficiency for Better Results?
10-07-2025
Rhodamine, as a high-brightness and stable fluorescent dye, plays a crucial role in biomolecular labeling and fluorescence imaging. However, achieving efficient Rhodamine conjugation depends not only on the quality of the dye itself but also on the choice of conjugation strategies, experimental conditions, and subsequent storage methods. This article provides an in-depth exploration of the concept, strategies, challenges, and optimization methods of Rhodamine conjugation, helping researchers achieve efficient labeling of antibodies, proteins, peptides, nucleic acids, and lipids to obtain reliable experimental results.
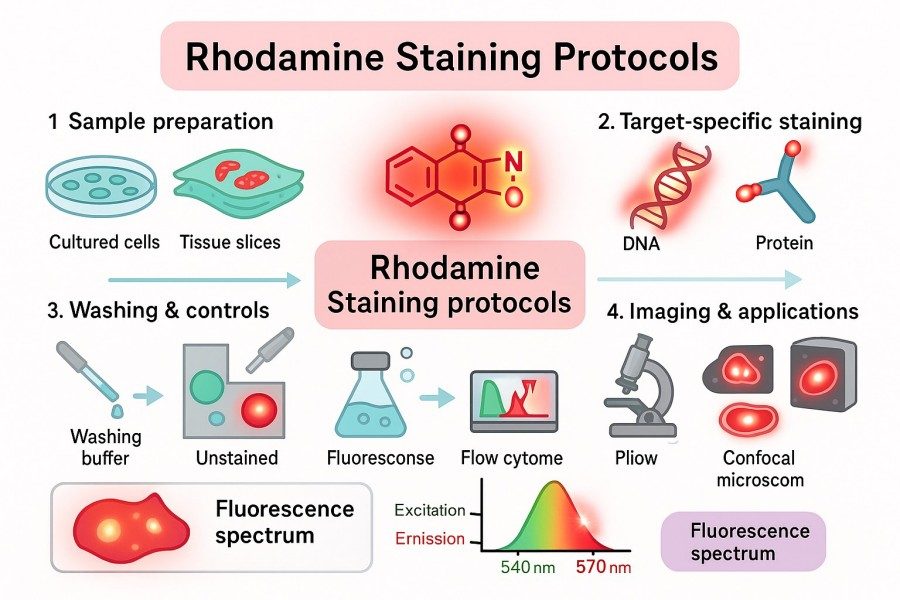
Rhodamine Staining Protocols: Tips for High-Quality and Reproducible Results
10-07-2025
Rhodamine dyes are widely used labeling tools among researchers due to their high brightness, photostability, and excellent cell permeability. Whether in cell staining, tissue section labeling, pathogen detection, or mitochondrial function studies, Rhodamine staining can provide high-resolution and reproducible imaging results. This article delves into rhodamine staining protocols from principles, types, common methods, optimization strategies, to practical applications, offering researchers practical tips to enhance staining quality and experimental reproducibility.
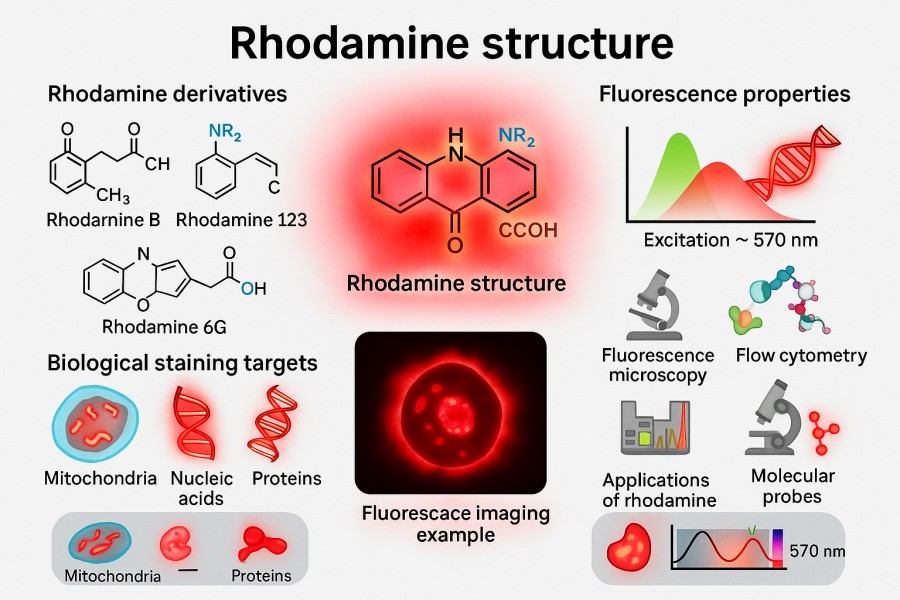
Rhodamine Synthesis: Overcoming Challenges in Fluorescent Applications
10-07-2025
Rhodamine dyes have become commonly used fluorescent reagents in scientific research and industry due to their excellent fluorescence performance, photostability, and broad applicability. In fields such as bioimaging, fluorescent probe design, and optoelectronic materials, the high brightness and tunable spectral properties of Rhodamine provide strong support for experiments. However, the synthesis of Rhodamine involves multiple chemical methods and technical routes, and the optimization of each step directly affects the dye's purity, stability, and optical properties. This article provides a comprehensive overview of Rhodamine synthesis strategies, key techniques, application directions, and best laboratory practices, aiming to offer researchers a systematic reference to achieve efficient and reproducible Rhodamine preparation and application.
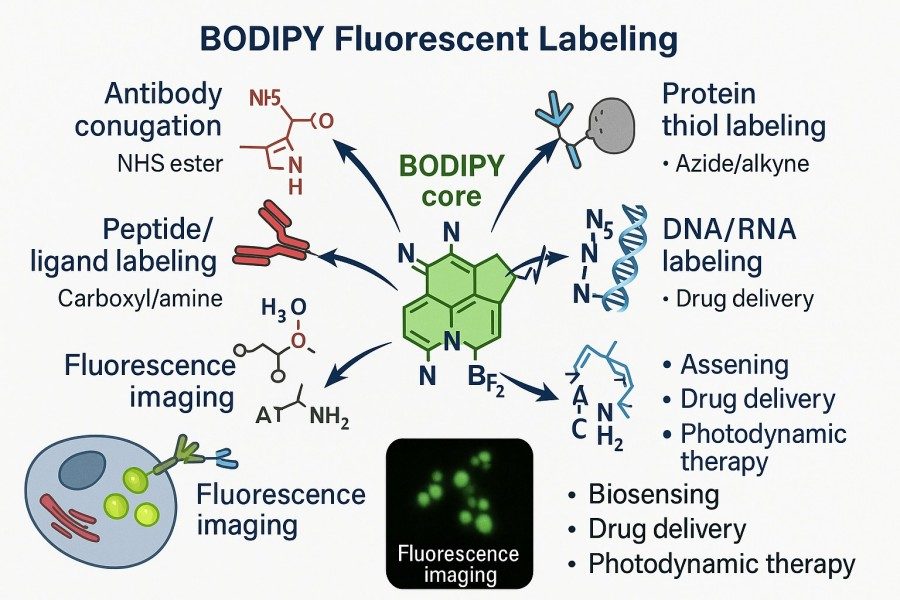
Mastering BODIPY Fluorescent Labeling: Techniques, Applications, and Expert Tips
08-29-2025
BODIPY (boron-dipyrromethene) dyes are gradually becoming the fluorescent tool of choice for researchers due to their high quantum yield, excellent photostability, narrow and tunable emission spectra, and extensive potential for molecular modification. Whether labeling proteins, peptides, lipids, or nucleic acids, BODIPY provides highly specific and bright signals, meeting the diverse needs of applications ranging from cellular imaging to high-throughput analysis. This article will explore BODIPY fluorescent labeling strategies, practical applications, and optimization tips, helping researchers overcome common challenges, enhance experimental efficiency and data reliability, and provide comprehensive technical guidance for both beginners and experienced scientists.
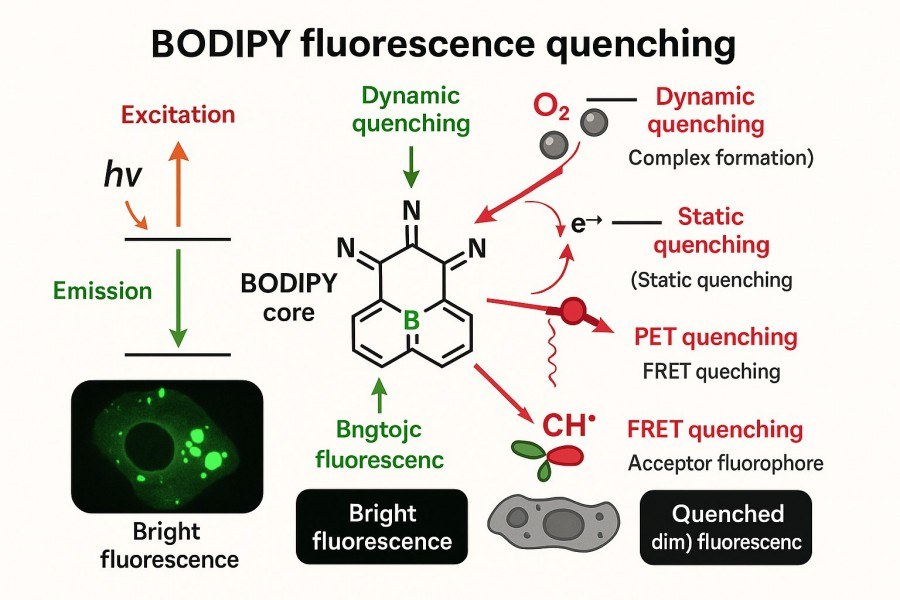
Understanding and Controlling Fluorescence Quenching in BODIPY Dyes for Optimal Probe Design
08-29-2025
BODIPY dyes, with their excellent photostability, high fluorescence quantum yields, and tunable spectral properties, have become indispensable fluorescent probes in bioimaging, optical sensing, and phototherapy research. However, in practical applications, fluorescence quenching is a critical factor that can affect BODIPY performance. Quenching not only reduces signal intensity but may also compromise probe selectivity and stability. Therefore, understanding quenching mechanisms, modulating structural factors, and implementing rational strategies are essential for developing high-performance BODIPY probes.
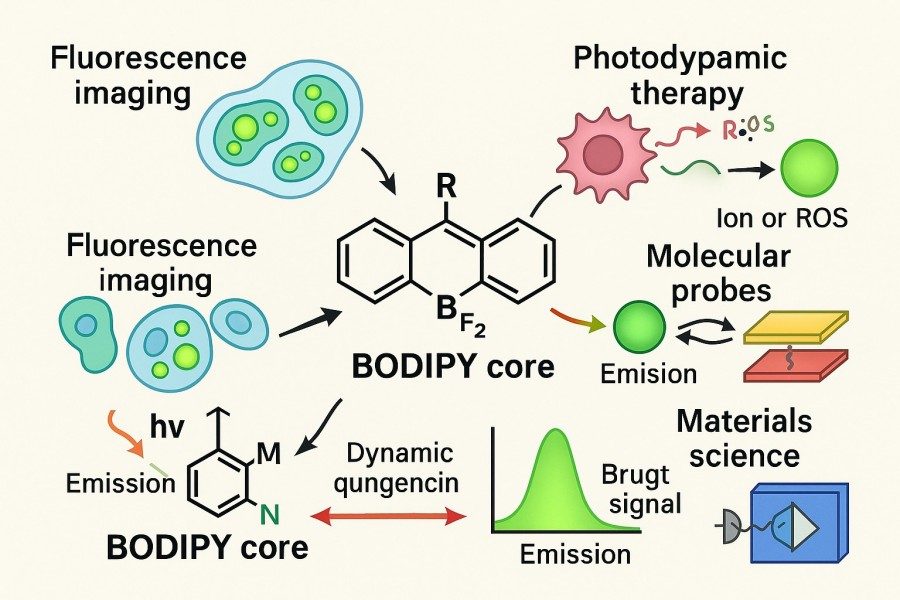
How to Modulate Photophysical, Photochemical, and Stability Properties of BODIPY Dyes?
08-29-2025
BODIPY dyes have become essential tools in fields such as bioimaging, photodynamic therapy, sensor development, and materials science due to their excellent photophysical properties, high fluorescence quantum yields, and chemical modifiability. However, native BODIPY suffers from certain limitations in emission wavelength, Stokes shift, photostability, and photochemical activity, making it challenging to meet the diverse requirements of different applications. To achieve precise utilization of high-performance BODIPY dyes, researchers need to systematically optimize their photophysical properties, photochemical behavior, and chemical/photostability through molecular structural modification and environmental tuning.
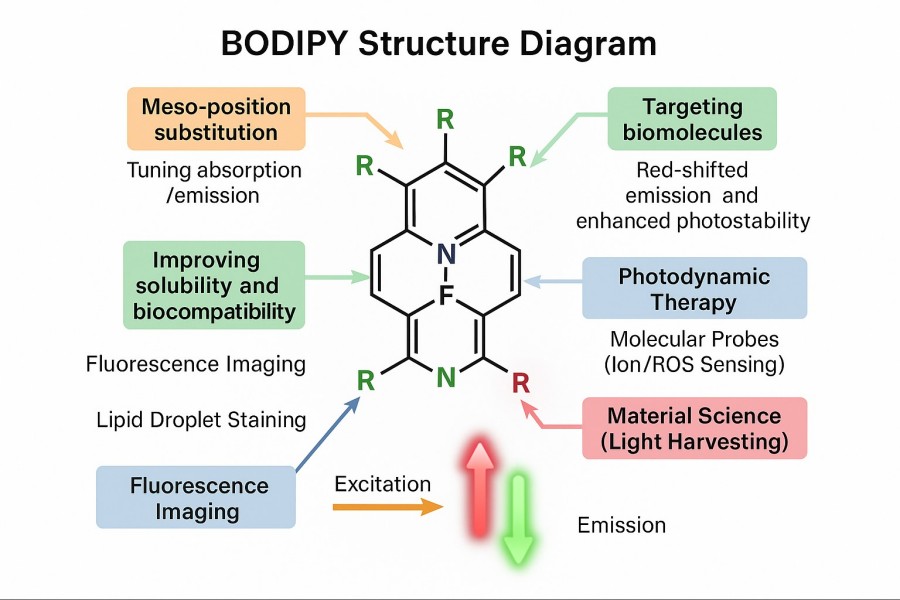
How Structural Modification of BODIPY Dyes Enhances Their Performance and Applicability?
08-29-2025
BODIPY dyes have gained widespread use in fluorescent probes, live-cell imaging, and optoelectronic materials due to their excellent photostability, sharp absorption and emission peaks, and high fluorescence quantum yield. However, native BODIPY molecules still have limitations in solubility, biocompatibility, spectral coverage, and targeting functionality. To meet the demands of complex experiments and advanced applications, scientists have systematically optimized BODIPY through structural modifications. Strategies such as core substitution, introduction of hydrophilic groups, bioconjugation, and smart responsive design not only enhance the optical performance and stability of the dyes but also enable advanced applications such as targeted delivery, functional imaging, and phototherapy. This article provides a comprehensive analysis of BODIPY structural modification methods and their value in scientific and industrial applications, helping researchers understand how customized design can improve dye performance and address common technical challenges in experiments.
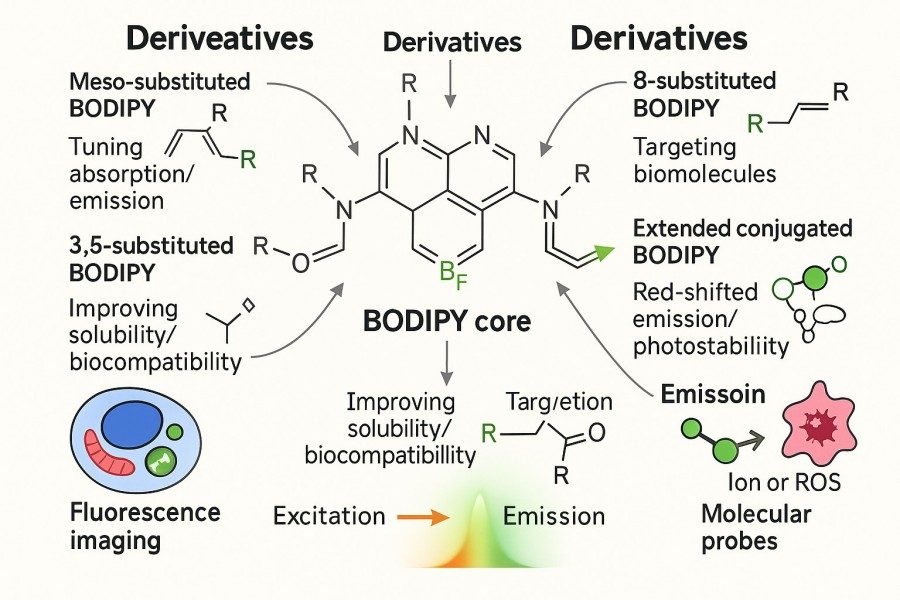
Understanding the Excitation and Emission Properties of BODIPY Dyes: A Guide for Optimal Fluorescent Probe Selection
08-29-2025
BODIPY (Boron-Dipyrromethene) dyes have become one of the most sought-after fluorescent tools for researchers due to their high fluorescence quantum yields, excellent photostability, and tunable spectral properties. However, different BODIPY derivatives exhibit significant variations in excitation and emission characteristics, and improper selection may result in weak signals, spectral crosstalk, or experimental failure. Therefore, a thorough understanding of BODIPY's optical principles, excitation/emission profiles, and structure–function relationships is essential for efficient experimental design and fluorescent probe optimization. This guide systematically explores the core structure, spectral properties, structural modification effects, and application-matching strategies of BODIPY dyes, helping researchers select the most suitable fluorescent probes for life sciences, materials science, and energy research.
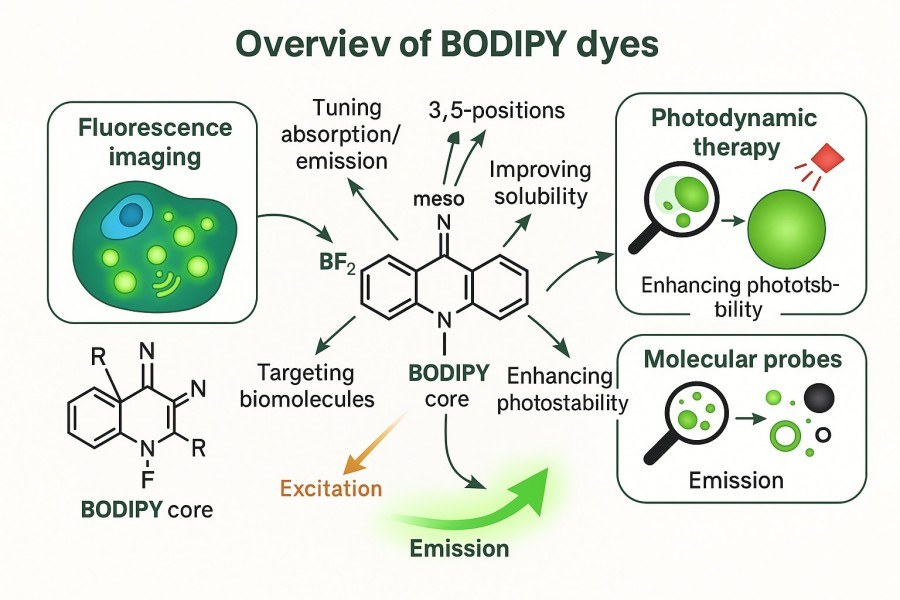
BODIPY Design, Synthesis and Functionalization: High-Performance BODIPY Derivatives
08-29-2025
In modern biochemistry and molecular imaging research, BODIPY (boron-dipyrromethene) dyes have become researchers' preferred fluorescent tools due to their excellent photophysical properties, chemical stability, and versatile derivatization potential. However, the design, synthesis, and functionalization of BODIPY dyes still face many challenges, including yield optimization, solubility control, and specific biomarking. This article provides a comprehensive analysis of BODIPY dyes—from the fundamentals, synthesis mechanisms, derivative development, and modification strategies to practical applications—highlighting their critical role in research and offering practical solutions for scientists.
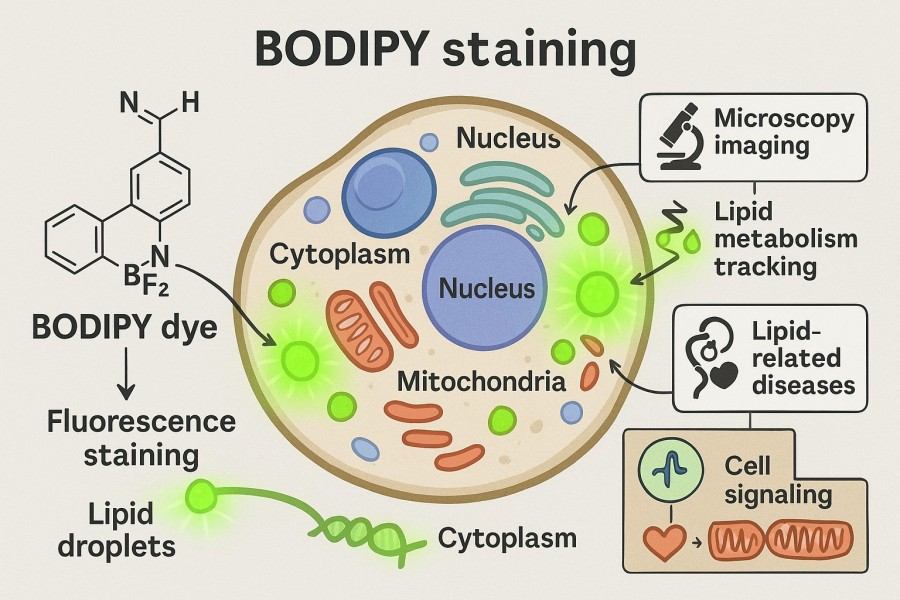
Complete Guide to BODIPY Staining for Reliable Research: Principle and Protocol
08-29-2025
BODIPY staining is a widely applied fluorescent labeling method in cell and tissue research, highly valued by researchers for its high sensitivity and stable signal. With the unique chemical structure and excellent optical properties of BODIPY dyes, scientists can directly visualize lipid droplet distribution, organelle function, and molecular dynamics. This technique not only plays an important role in fundamental cell biology and metabolic mechanism studies but is also extensively used in drug screening, disease model construction, and live-cell imaging. To achieve reliable experimental results, researchers need a deep understanding of the bodipy staining principle, master the bodipy staining protocol under different experimental systems, and flexibly optimize conditions based on sample characteristics.
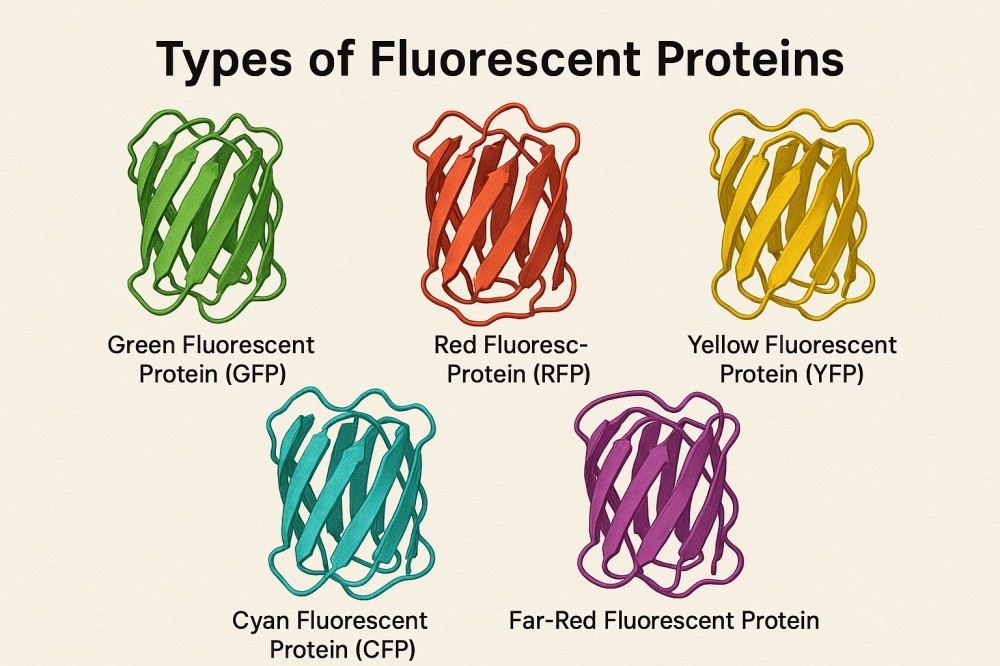
Comprehensive Guide to Fluorescent Proteins and Their Uses in Research
07-14-2025
Fluorescent proteins have their own optical characteristics and are often used as versatile fluorescent labeling tools in the life science research fields of cell imaging, protein tracking, and molecular detection. By using fluorescent proteins of different colors and spectral properties, complex multiplex labeling and dynamic monitoring can be achieved, greatly improving the sensitivity and accuracy of experiments. Structural, performance, and application studies of fluorescent proteins provide researchers with a reference to help them choose the right fluorescent protein, optimize experimental conditions, and promote the application of novel research.
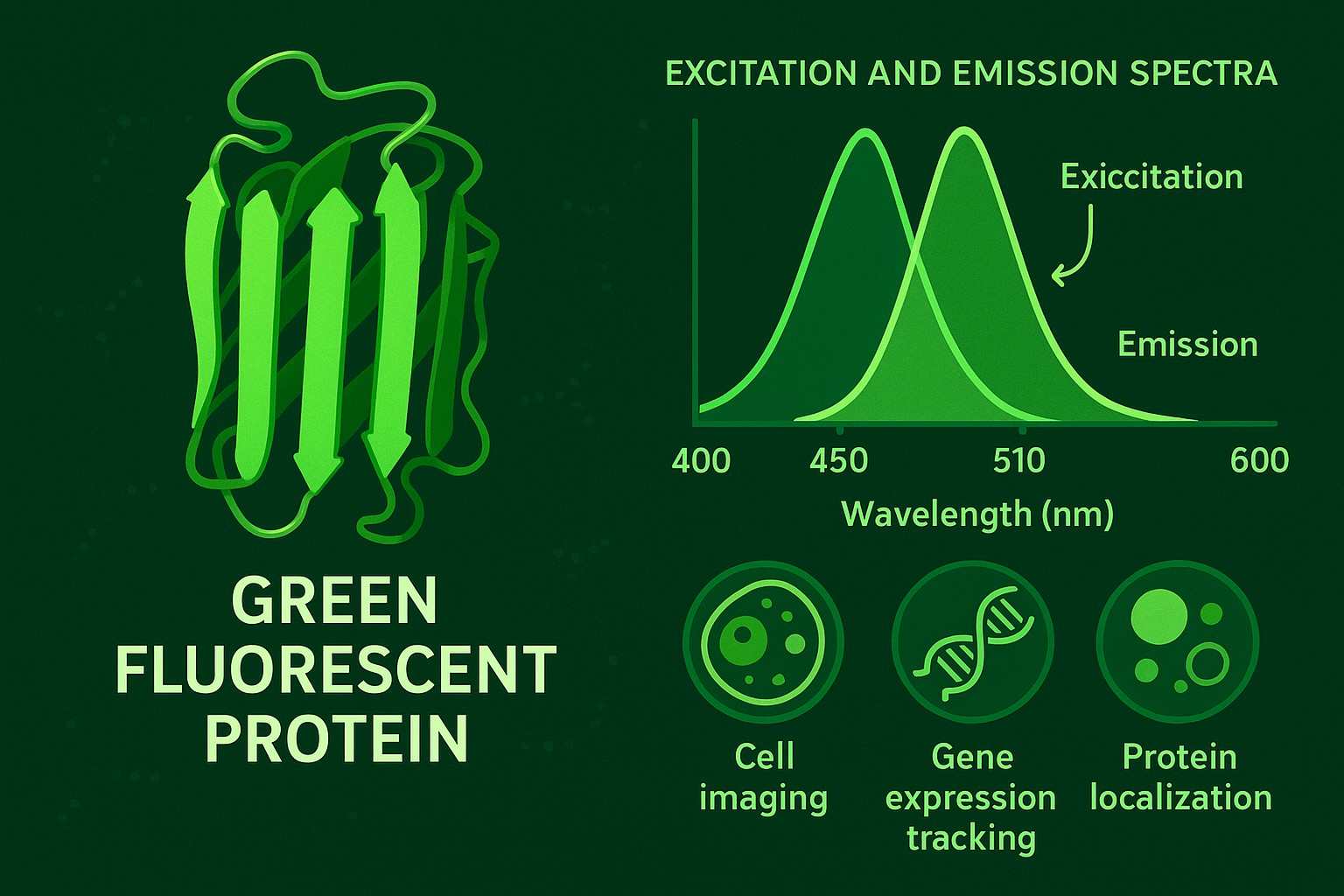
Green Fluorescent Protein: A Comprehensive Overview from Discovery to Application
07-14-2025
Green fluorescent protein (GFP), a naturally fluorescent protein from jellyfish, has been used for years to allow real-time, visual tracking of proteins and cellular imaging without interfering with the normal functions of a cell. The structure of GFP has undergone over a decade of development and optimization, leading to the creation and refinement of numerous mutants with significantly enhanced brightness, photostability, and spectral properties. GFPs and their derivatives are now used across molecular biology, cell biology, neuroscience, and drug discovery in areas such as the study of gene expression, protein localization, and cellular dynamics, and bioimaging.
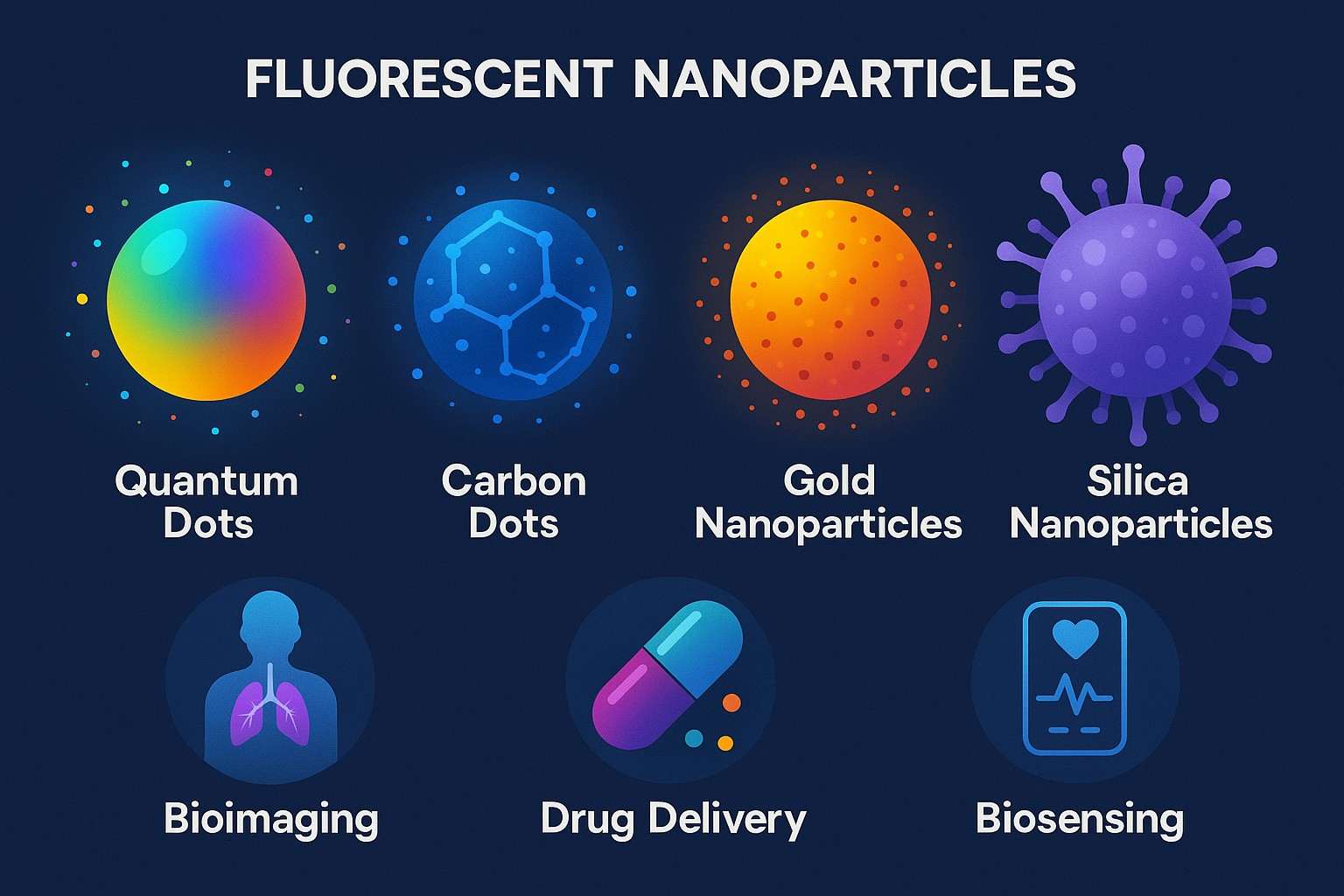
Overview of Fluorescent Nanoparticles: Applications, Challenges and Solutions
07-14-2025
Functional nanomaterials called fluorescent nanoparticles are particularly interesting for applications in bioimaging, disease diagnostics, drug delivery, and environmental sensing. Owing to their excellent optical characteristics, tunable sizes and shapes, and facile surface modification strategies, these fluorescent nanomaterials have shown great promise in biological fields as compared to conventional fluorescent dyes and proteins. They are important for conducting research and achieving success in the field of nanobiotechnology.

Mastering the Spectrum: A Comprehensive Guide to Cy3 and Cy5 Dyes
01-30-2025
In biomedical research fluorescent dyes serve as essential tools. During cell imaging and molecular biology experiments, scientists use fluorescent dyes to visualize biomolecules and cellular structures. Among many fluorescent dyes, Cy3 and Cy5 dyes distinguish themselves from other fluorescent dyes because of their distinctive spectral properties and extensive usage across various applications. The two dyes find extensive application across biological imaging and multiple scientific fields like molecular biology, proteomics, and genomics research.

Hoechst Dyes: Definition, Structure, Mechanism and Applications
01-30-2025
Fluorescent dyes are essential tools for biomedical researchers. Scientists can study living cells better through fluorescent dyes, which help them find new life science solutions quickly. From cell cycle analysis and apoptosis detection to live cell imaging, fluorescent dyes have a wide range of applications. Among all fluorescent dyes, Hoechst dyes stand apart from other fluorescent dyes because of their special features and extensive usage areas.

Fluorescent Probes: Definition, Structure, Types and Application
10-16-2024
The organism contains a multitude of chemical substances, including enzymes, proteins, and various cations and anions, which are of significant importance to human life activities and health. Additionally, there are numerous substances in nature that await testing, including antibiotics, disease markers, and pollutants, which merit further exploration by researchers. Conducting tests on these substances has positive implications, and various methods have been developed to detect specific analytes, such as chromatography, mass spectrometry, and biological methods. Each of these methods has its characteristics but tends to be time-consuming and more costly, and they cannot provide real-time information feedback.

Fluorescent Dyes: Definition, Mechanism, Types and Application
10-16-2024
Fluorescent dyes are a class of high-performance luminescent materials based on organic molecules, widely recognized for their excellent optical properties and broad application prospects. Among the various types of fluorescent dyes, rhodamine and fluorescein are the two most widely used, with extensive literature investigating their properties and applications. Other common fluorescent dyes include coumarins, polycyclic aromatic compounds, NBD-amines, naphthalimides, BODIPY dyes, cyanines, as well as thiazines and oxazines. These dyes typically exhibit good photostability, significant fluorescence intensity, and adjustable emission wavelengths, allowing them to play crucial roles in various fields such as biological imaging, optical sensing, laser technology, and display technology.

Coumarin Dyes: Definition, Structure, Benefits, Synthesis and Uses
10-16-2024
Coumarin dyes are a significant class of synthetic organic compounds derived from coumarin, known for their unique fluorescent properties and vibrant colors. These dyes are characterized by their benzopyranone structure, which contributes to their photophysical behavior, making them ideal for a wide range of applications in fields such as bioimaging, laser technology, and organic light-emitting diodes (OLEDs). As research progresses, coumarin dyes continue to evolve, leading to the development of novel derivatives with tailored functionalities for specific uses in the life sciences and materials science.

BODIPY Dyes: Definition, Structure, Synthesis and Uses
10-16-2024
BODIPY dyes (boron-dipyrromethene) are a class of fluorescent compounds known for their exceptional photophysical properties, including high fluorescence quantum yields, photostability, and tunable emission spectra. These dyes have gained significant attention in various scientific and industrial fields due to their versatility. With a well-defined structure based on a boron-fluorine complex and dipyrromethene backbone, BODIPY dyes can be easily modified for specific applications. Their uses span from bioimaging and chemical sensing to photodynamic therapy and organic electronics.

Cyanine Dyes: Definition, Structure, Types and Uses
10-16-2024
Cyanine dyes are a class of synthetic dyes widely used in scientific research and industrial applications due to their unique optical properties, including strong fluorescence and tunable absorption/emission wavelengths. These dyes are particularly valued in fields such as molecular imaging, biotechnology, and materials science for their ability to label biomolecules, enhance imaging techniques, and serve as fluorescent probes. The versatile structure of cyanine dyes allows for modifications that optimize their solubility, photostability, and specificity, making them essential tools in fluorescence-based assays, medical diagnostics, and therapeutic applications.

Rhodamine Dyes: Definition, Structure, Uses, Excitation and Emission
10-07-2024
Rhodamine is an important class of flavonoid dyes. Due to its extended π-conjugated system, its open form exhibits strong fluorescence. They are used as laser dyes and in studies for detecting polymer and oligonucleotide adsorption. Dendritic polymers containing rhodamine as one of their components may have potential applications in materials science. Rhodamine dyes are used as fluorescent labeling reagents in biology because of their excellent optical properties, such as high fluorescence quantum yield, long excitation wavelength, and high photostability.

Fluorescein Dyes: Definition, Structure, Synthesis and Uses
10-07-2024
Fluorescein, a synthetic organic compound, has become an indispensable tool in biological research due to its fluorescent properties. Its bright green fluorescence under ultraviolet or blue light makes it ideal for various labeling applications in cell biology, molecular biology, and immunology. The versatility of fluorescein and its derivatives, particularly fluorescein isothiocyanate (FITC), allows for the specific labeling of cells, proteins, antibodies, and nucleic acids, providing crucial insights into the structure and function of biological systems.


