How to Choose the Right Fluorescent Dye for Cell Staining
Choosing the right fluorescent dye is one of the most important early decisions in any cell staining workflow because the dye determines far more than the visible color in the final image. It shapes signal brightness, background behavior, live-cell compatibility, fixation tolerance, spectral overlap risk, and ultimately the biological meaning of the result. A dye that is bright but poorly matched to the sample or instrument can create misleading fluorescence, unstable localization, or unnecessary troubleshooting. A more suitable dye, even if less visually dramatic at first glance, often delivers clearer interpretation and better reproducibility across experiments. In modern fluorescence-based cell analysis, researchers rarely choose dyes in isolation. They select them in the context of sample type, staining target, readout platform, and whether the experiment emphasizes structural imaging, functional response, viability assessment, organelle localization, or multiparameter analysis. This means dye selection should be treated as a workflow design decision, not a purchasing step at the end of planning. The best dye is the one that fits the biology, the protocol, and the optical system at the same time.
What Are Fluorescent Dyes and Why Are They Important in Cell Analysis?
Fluorescent dyes are light-responsive molecules that absorb energy at one wavelength and emit it at a longer wavelength, allowing cellular structures, biochemical states, and functional changes to be visualized with high sensitivity. In cell analysis, they serve as optical translators: they convert otherwise invisible biological information into measurable fluorescence signals that can be detected by microscopy, imaging systems, and flow-based platforms. This is why fluorescent dyes are not simply colorful reagents. They are signal-generating tools that make it possible to distinguish cells, compartments, states, and responses that would be difficult or impossible to interpret using transmitted light alone. Their importance has grown with the expansion of modern fluorescence workflows. Researchers now routinely need to analyze not only morphology, but also organelle localization, membrane properties, intracellular transport, live/dead status, metabolic behavior, and multiparameter cell states within the same experimental framework. Fluorescent dyes make these questions experimentally accessible because they can be selected for different excitation and emission windows, different chemical behaviors, and different biological targets. In practice, they allow cell analysis to move from simple visualization toward more structured, information-rich interpretation.
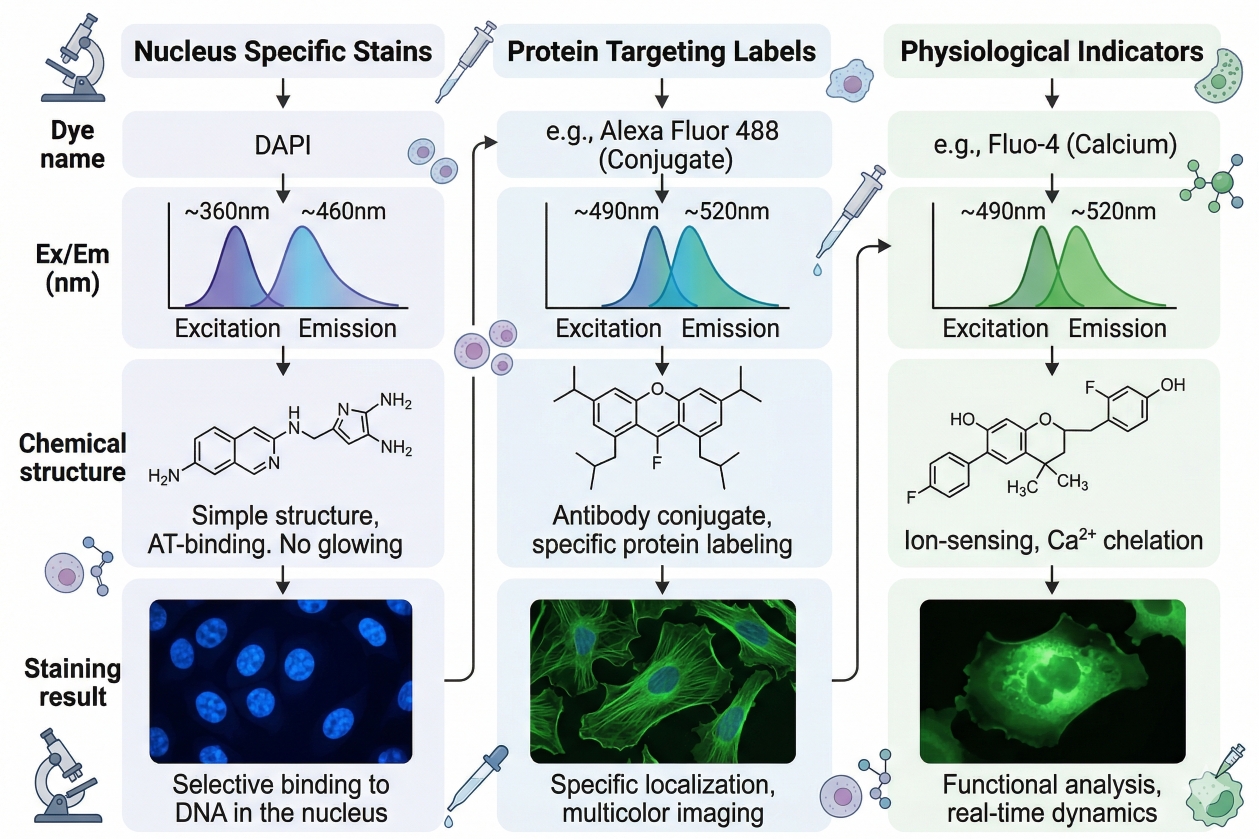 Fig. 1. Fluorescent dyes enable cell analysis by converting structural and functional cellular information into detectable optical signals (BOC Sciences Authorized).
Fig. 1. Fluorescent dyes enable cell analysis by converting structural and functional cellular information into detectable optical signals (BOC Sciences Authorized).
How Fluorescent Dyes Generate Detectable Signals?
Although fluorescence is often described simply in terms of excitation and emission wavelengths, real experimental performance depends on multiple linked properties. Signal intensity is affected by extinction coefficient and quantum yield, while practical usability is also shaped by photostability, environmental sensitivity, background behavior, and how well the dye can be separated from autofluorescence. A dye that is theoretically bright may still perform poorly if it bleaches rapidly, accumulates nonspecifically, or emits in a region crowded by other fluorophores. The biological environment also matters. Some dyes become brighter when they bind nucleic acids or partition into membranes, while others change fluorescence in response to polarity, pH, or transmembrane potential. Some dyes are almost purely structural labels, whereas others act as reporters of cell state. This means the detected fluorescence signal is not always a straightforward measure of dye amount. It may instead reflect a biological interaction, intracellular localization, or chemical environment. For that reason, fluorescent dye selection must always account for both optical behavior and biological mechanism.
Why Fluorescent Dyes Are Widely Used in Modern Cell Analysis?
Fluorescent dyes are widely used because they are compatible with a broad range of research questions and instrumentation formats. They support high-contrast imaging, rapid population-level discrimination, live-cell observation, endpoint structural analysis, and multicolor experiments in which several cellular properties are observed at once. This flexibility makes them valuable not only in exploratory cell biology, but also in assay development, treatment-response studies, screening, localization analysis, and workflow quality control. Another major advantage is modularity. Researchers can pair different dye classes with different experimental priorities: nuclear dyes for segmentation, membrane dyes for morphology, organelle dyes for compartment analysis, viability dyes for sample quality control, and functional indicators for dynamic cellular processes. This broad utility explains why fluorescent dyes are central to product categories such as fluorescent dyes, fluorescent probes, cell and organelle stains, and more specialized application-oriented staining systems.
What Is Cell Staining and What Does It Help Researchers Observe?
Cell staining is the deliberate use of dyes, probes, or labeled reagents to visualize and analyze cells according to structural, biochemical, or functional differences. In fluorescence workflows, staining is not merely a decorative step that makes cells easier to see. It is a method for turning a biological feature into a detectable readout. Depending on the stain selected, the signal may reflect where a structure is located, whether a membrane is intact, how an organelle is distributed, whether a metabolic substrate is taken up, or whether a cell population differs in condition or state. Because cells are highly complex and many of their most important features are transparent under ordinary observation, staining is often what makes cellular interpretation possible. It helps researchers define cell boundaries, identify subcellular compartments, distinguish viable and non-viable populations, compare treated and untreated samples, and design multiparameter assays that integrate morphology, location, and function. In that sense, cell staining is not just a visualization technique. It is one of the most widely used interfaces between cell biology and optical analysis.
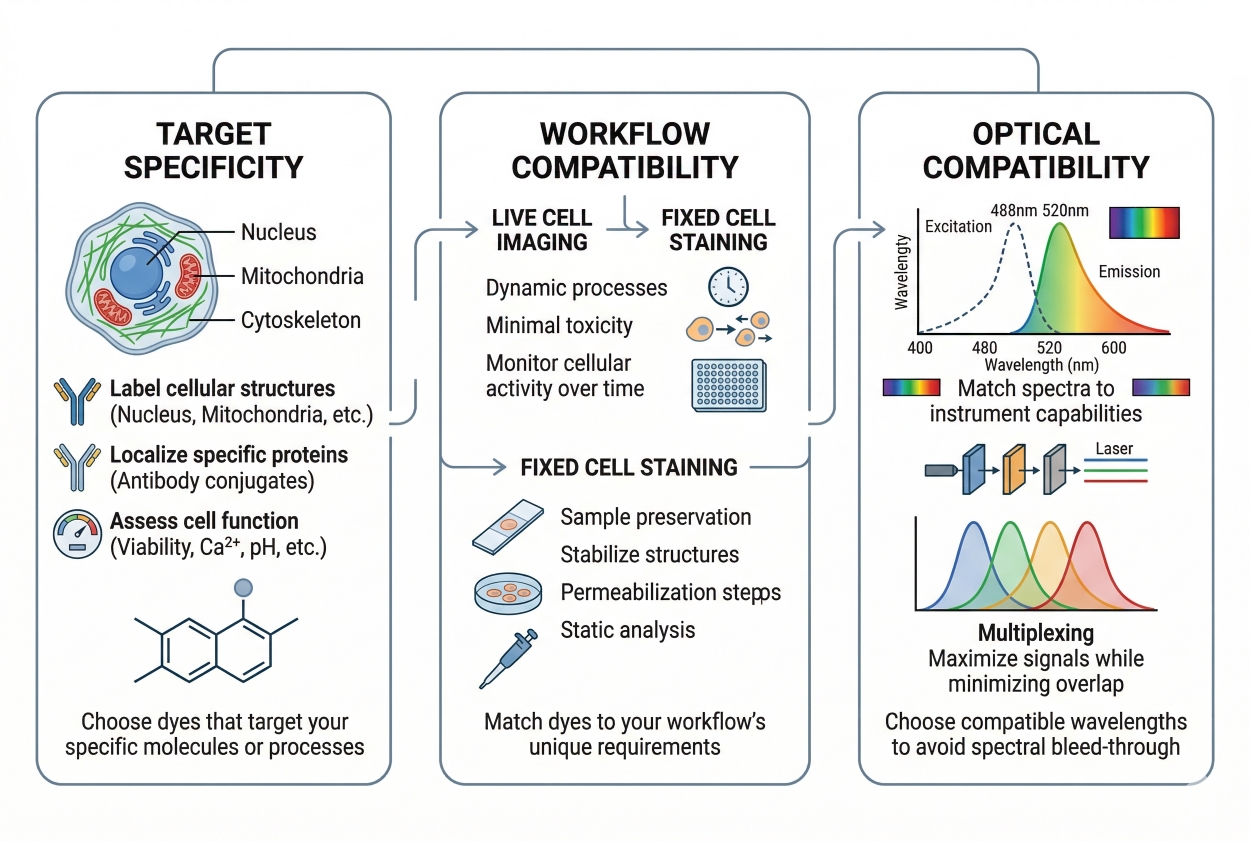 Fig. 2. Choosing the right fluorescent dye requires balancing target specificity, workflow fit, and optical compatibility (BOC Sciences Authorized).
Fig. 2. Choosing the right fluorescent dye requires balancing target specificity, workflow fit, and optical compatibility (BOC Sciences Authorized).
Structural Staining vs Functional Staining
A useful way to understand cell staining is to divide it into structural and functional purposes. Structural staining is mainly concerned with location and organization. It helps answer questions such as where the nucleus is, whether cell boundaries are intact, how organelles are arranged, or whether morphology differs between conditions. Nuclear stains, membrane stains, and many organelle-targeted dyes are often used in this way. Functional staining answers a different class of question. Instead of showing only where something is, it is used to indicate what state a cell or compartment is in. Examples include viability dyes, membrane potential-sensitive dyes, uptake probes, and other environment-responsive fluorophores. These stains are especially valuable when the biological question involves activity, condition, or physiological change. However, they also require more careful interpretation because the fluorescence often depends on a dynamic mechanism rather than a static location alone.
What Cell Staining Helps Researchers Observe at the Whole-Cell Level?
At the whole-cell level, staining helps define cell size, shape, morphology, population heterogeneity, and changes associated with treatment or environmental stress. In imaging, this may include distinguishing round versus spread morphologies, identifying membrane blebbing, assessing attachment quality, or evaluating whether a culture looks structurally uniform. In population-based workflows, staining may help separate intact cells from debris or distinguish one subpopulation from another based on fluorescence behavior. This whole-cell perspective is often essential before any more specific interpretation begins. Researchers may need to know whether the cells are healthy enough to analyze, whether the sample is morphologically comparable across replicates, or whether a structural change has occurred before investigating a finer biochemical mechanism.
What Cell Staining Helps Researchers Observe at the Subcellular Level?
At the subcellular level, cell staining is used to examine nuclei, membranes, mitochondria, lysosomes, endoplasmic reticulum, Golgi structures, lipids, and cytoskeletal elements. These staining patterns help researchers understand localization, compartment identity, intracellular redistribution, and spatial relationships between cellular components. Such analysis is central to compartment-specific biology, because many meaningful changes in cell state occur not through the appearance of a completely new structure, but through relocation, accumulation, depletion, or altered organization within existing structures. This is why subcellular cell staining is foundational in cell imaging, localization analysis, organelle comparison, and multicolor microscopy. Without staining, many intracellular features would remain unresolved or would lack the contrast needed for consistent interpretation.
Why Cell Staining Quality Directly Affects Downstream Interpretation?
Cell staining quality determines whether downstream data can be trusted. A visually bright signal is not necessarily a biologically meaningful one. Poorly matched stains can create diffuse background, false co-localization, excessive uptake in damaged cells, weak retention during washing, or strong spectral interference in multicolor experiments. These problems can affect not only the stain itself, but the interpretation of the entire workflow. For this reason, staining quality should be judged in terms of specificity, reproducibility, biological plausibility, and compatibility with the rest of the assay. If the stain does not match the sample type, target logic, or acquisition method, the result may still look attractive while being analytically weak. This is why cell staining design must be integrated with protocol planning, dye selection, and platform-specific strategy rather than handled as an isolated technical step.
Need help matching fluorescent dyes to your imaging system or assay workflow?
We can help align dye selection with your microscope configuration, detection channels, and experimental goals to achieve clear and reliable results.
Major Types of Fluorescent Dyes Used for Cell Staining
Fluorescent dyes used for cell staining can be grouped into several practical classes according to what they label and how they behave in cells. This classification is useful because dye selection is rarely solved by choosing a fluorophore family alone. Researchers usually need to decide which biological category of stain is appropriate first, and only then determine which specific dye within that category offers the right spectral, chemical, and workflow properties. In other words, a successful staining strategy often begins with dye function before moving to dye identity. These major classes are not perfectly isolated from one another. Some dyes can serve more than one purpose, and some functional probes also provide structural contrast. Still, organizing dyes into broad classes makes selection more rational and reduces the risk of choosing a reagent that is bright but conceptually mismatched to the assay. The categories below cover the most common fluorescent dye types used in cell staining workflows.
Nuclear Dyes
Nuclear dyes are used to label cell nuclei for counting, segmentation, morphology analysis, and spatial reference in multicolor experiments. They are among the most widely used stains because the nucleus provides a stable and easily interpretable landmark in many workflows. In fluorescence microscopy, nuclear dyes help define cell number, support image segmentation, and create a reliable counterstain that improves contextual interpretation of other channels. These dyes often interact with DNA or related nuclear components and are commonly associated with broader product areas such as DNA stains and nuclear fluorescent probes. When selecting among nuclear dyes, researchers should consider permeability, fixation compatibility, emission channel occupancy, and whether the stain is intended for live cells, fixed cells, or both. A useful nuclear stain is not just bright. It must also remain clean, segmentable, and compatible with the rest of the panel.
Cell Membrane Dyes
Cell membrane dyes are designed to highlight the plasma membrane, define cell boundaries, or report on membrane-associated behavior. They are valuable when researchers need to distinguish individual cell outlines, assess morphology, visualize membrane dynamics, or incorporate boundary information into multicolor imaging. In some cases, membrane dyes are also chosen because they offer broad whole-cell contrast that supports cell counting or tracking in mixed samples. Within this category, not all dyes behave in the same way. Some are general membrane labels, while others are sensitive to membrane potential, lipid environment, or local membrane organization. This is why membrane dye selection should reflect whether the goal is structural boundary definition or a more functional membrane-associated readout. Relevant categories include cell membrane fluorescent probes and related membrane-active fluorescence tools.
Organelle-Specific Fluorescent Dyes
Organelle-specific dyes are selected when the experiment requires clear discrimination of intracellular compartments rather than whole-cell contrast alone. These dyes may target mitochondria, lysosomes, Golgi structures, endoplasmic reticulum, lipid-associated compartments, or other subcellular domains. Their value lies in enabling localization analysis, compartment comparison, and interpretation of intracellular distribution patterns under different treatment or environmental conditions. Organelle dyes are especially important because localization often carries biological meaning. A treatment may not change whether a signal exists, but instead where it appears or whether a compartment changes shape, abundance, or relative organization. This is why organelle-directed staining is foundational for spatial cell biology and related fluorescence workflows. Product areas that support this class include mitochondrial fluorescent probes, lysosomal fluorescent probes, endoplasmic reticulum fluorescent probes, Golgi fluorescent probes, and lipid fluorescent probes. In practice, these dyes should be chosen with particular attention to whether the experiment is live or fixed, since retention and targeting logic may change dramatically after processing.
Viability and Live/Dead Staining Dyes
Viability dyes are used to distinguish live cells from dead or membrane-compromised cells and are therefore critical in both imaging and population-based analysis. Some work by taking advantage of membrane integrity, others depend on intracellular conversion or retention, and some are designed to preserve viability information into later workflow stages. Their importance lies in quality control as much as in biological measurement. Dead or severely damaged cells often generate misleading fluorescence, so identifying them can improve the interpretability of the entire experiment. This category overlaps with live/dead discrimination, treatment-response analysis, and in some cases with apoptosis-related workflows. Selection should be guided by whether the assay remains live, whether a post-fix stage is planned, and how the viability channel fits within the broader optical layout. A viability dye should not be selected solely because it is convenient. It should be chosen because its staining logic matches the biological state the experiment needs to distinguish.
Cell Tracking and Proliferation-Related Dyes
Cell tracking dyes are used when the experiment involves following cell movement, migration, redistribution, or signal persistence across time. In proliferation-related workflows, the same or related dye classes may help track dilution of signal over successive divisions or maintain a long enough labeling window for short- to medium-term observation. These dyes are especially useful in longitudinal designs, co-culture studies, migration assays, and experiments that require population follow-up after initial labeling. Unlike static structural stains, tracking dyes must be judged by retention behavior, transfer risk, persistence, and how cleanly they remain associated with the intended cells over time. These requirements make them a distinct category within cell staining, even when they share fluorophore families with more conventional imaging labels. Product groups such as cell proliferation tracer fluorescent probes are especially relevant in this area.
Environment-Sensitive and Functional Fluorescent Probes
Environment-sensitive dyes and functional probes are used when researchers need the fluorescence signal to reflect a biological state rather than only a location. These may include dyes responsive to membrane potential, pH, ion concentration, oxidative conditions, or specific uptake pathways. Their value lies in providing mechanistically informative fluorescence rather than purely structural contrast. In these workflows, signal interpretation is closely tied to the chemistry of the probe and the physiological condition of the sample. Because they are state-responsive, these probes demand more careful interpretation than many structural stains. A change in fluorescence may reflect altered cell physiology, treatment effect, transport behavior, or compartment condition rather than a simple difference in abundance. Relevant product categories include pH indicators, ion fluorescent probes, calcium, chloride and other indicators, and nitric oxide and ROS probes. These dyes are especially valuable when cell staining is intended to report on physiological condition rather than static structure.
Why Choosing the Right Fluorescent Dye Matters More Than Just Picking a Color?
Many dye-selection problems begin when fluorophore choice is reduced to color matching. Although excitation and emission channels are important, they are only part of the selection process. A dye that fits a convenient channel may still fail because it is too dim for the target abundance, too toxic for live cells, too unstable under repeated illumination, or incompatible with fixation and washing steps. Color alone does not determine whether a signal is trustworthy.
- How Dye Choice Affects Signal Quality: Signal quality is shaped by brightness, background behavior, localization fidelity, and resistance to bleaching. Two dyes emitting in similar spectral regions can perform very differently in cells depending on how strongly they accumulate, whether they aggregate, and how cleanly they separate from autofluorescence. A stronger dye selection process therefore considers how the signal will behave in the biological sample, not just how the fluorophore behaves in a spectral chart.
- How Dye Properties Influence Cell Compatibility: A dye suitable for fixed staining may disturb live cells, while a live-cell-compatible stain may fail after fixation or prolonged washing. The correct choice depends on whether the experiment prioritizes dynamic observation, structural preservation, or downstream multiparameter processing. This is why platform fit and sample condition are often more important than dye popularity.
- Why Poor Dye Selection Can Create Misleading Results: Poor dye selection can generate convincing but misleading fluorescence. Non-specific accumulation may look like biological localization. Weak retention may be mistaken for a treatment effect. Spectral overlap may create apparent co-localization that is only optical interference. These problems often consume more time than careful selection would have required at the beginning. In that sense, dye choice is a preventive step as much as a technical one.
Core Criteria for Choosing a Fluorescent Dye for Cell Staining
Once the target and workflow are defined, dye selection should be evaluated through a practical set of criteria. No single criterion is sufficient on its own. A dye may excel in brightness but fail in cell compatibility. Another may be stable and convenient but occupy a problematic channel. The best selection process therefore uses several criteria together.
- Brightness and Signal Intensity: Brightness matters because low signal can make interpretation unstable, especially in low-abundance targets or rapid acquisition workflows. However, brightness should be judged in context. A very bright dye that produces high background or self-quenching may be less useful than a moderately bright dye that localizes cleanly and behaves consistently. Brightness should therefore be considered as usable signal rather than theoretical intensity alone.
- Photostability: Photostability is essential in repeated imaging, time-lapse observation, and any workflow where the sample may experience prolonged illumination. Dyes that bleach rapidly can reduce quantitative consistency and distort spatial interpretation if one channel fades more quickly than another. In practical selection, photostability is especially important for dynamic imaging, confocal acquisition, and multichannel experiments where repeated scanning is unavoidable.
- Water Solubility and Handling Behavior: Solubility influences how predictably a dye can be prepared, diluted, and applied. Poorly handled dyes may precipitate, aggregate, or behave inconsistently across samples. Some dyes are easier to formulate and more forgiving in common buffers, while others require tighter control over solvent use or stock handling. Good handling behavior reduces variability and supports smoother protocol execution.
- Spectral Compatibility with the Instrument: A dye must match the excitation source, filter set, or detector layout available in the actual experiment. Spectral compatibility is not simply a matter of whether excitation is possible. It also includes how cleanly the emission can be separated from autofluorescence and adjacent channels. A fluorophore with ideal properties on paper is still a poor choice if the instrument cannot excite or detect it cleanly within the planned panel.
- Fixation Tolerance and Retention Behavior: Some dyes remain informative after fixation, while others lose meaning once the sample is processed. This criterion is especially important when a workflow might start live and later move into fixed analysis, or when additional staining layers will be added after the initial step. Retention should never be assumed. It should be part of the selection logic from the outset.
- Cytotoxicity and Compatibility with Live-Cell Workflows: In live-cell staining, compatibility with cell health is critical. A dye may provide strong contrast but still perturb membrane properties, organelle behavior, or overall viability. For live workflows, the appropriate dye is not just the one that stains effectively, but the one that does so with acceptable disturbance under the real observation window of the assay.
Common Mistakes in Fluorescent Dye Selection and How to Avoid Them
Many dye-selection failures are predictable because they arise from recurring planning errors. Recognizing these errors early can save substantial time during protocol development and reduce false interpretation later.
- Choosing Dyes Without Considering the Readout Platform: A dye that performs well in one platform may be less suitable in another. Microscopy, endpoint imaging, and population analysis each define "good signal" differently. Selecting a dye without considering how the data will be collected often produces avoidable problems in sensitivity or interpretability.
- Ignoring Spectral Overlap in Multicolor Panels: Channel crowding is one of the most common avoidable problems in fluorescence workflows. Even if each dye seems acceptable alone, the panel may fail if emission overlap is not considered early. This is especially true when channels are assigned based only on convenience rather than relative signal behavior and panel balance.
- Using Live-Cell Dyes in Incompatible Post-Fix Workflows: Another frequent mistake is expecting a live-cell stain to retain the same meaning after fixation. Unless retention and compatibility are known, this assumption can produce misleading endpoint results. If the workflow may include fixation, that should influence dye choice before the experiment begins.
- Overvaluing Brightness While Neglecting Specificity and Workflow Fit: Bright dyes are attractive, but excessive focus on brightness often hides more important issues such as specificity, retention, or biological compatibility. In many workflows, a cleaner and more stable dye yields better analysis than the brightest possible option.
A Practical Decision Framework for Choosing the Right Fluorescent Dye
A reliable decision framework starts from the biological question rather than from a product catalog. What needs to be seen or measured? Under what sample conditions? Through which platform? Once those questions are answered, the list of realistic dye choices becomes much more manageable.
- Start from the Biological Question: Determine whether the stain is needed for structure, function, viability, localization, tracking, or a combination of these. A clear biological purpose narrows the dye class immediately and prevents random browsing across unrelated fluorophores.
- Match the Dye to the Sample State and Workflow: Decide whether the sample will remain live, be fixed, or pass through multiple processing stages. The dye should match not only the target but also the handling pathway. This prevents later conflict between staining logic and protocol reality.
- Check Spectral Layout and Controls: Confirm that the chosen dye fits the available excitation and detection setup and that appropriate controls can be included. Even a good dye becomes a poor choice if it occupies the wrong channel or complicates interpretation unnecessarily.
- Validate with a Small-Scale Pilot Before Full Experiment Rollout: Before scaling the experiment, test the dye under the actual sample conditions and acquisition settings that will be used. A small pilot helps determine whether signal strength, localization, background, and retention behave as expected. In most cases, this step is more efficient than trying to solve avoidable problems after the main experiment has already begun.
Choosing between different fluorescent dyes for your cell staining experiment?
We can help you evaluate dye properties such as brightness, photostability, cell permeability, and spectral compatibility for your specific application.
How to Match Fluorescent Dyes to Different Cell Staining Scenarios?
Dye selection becomes more practical when it is tied to the actual scenario in which the stain will be used. Instead of asking which dye is best in general, researchers should ask which dye type is best for a specific workflow. This approach reduces overgeneralization and makes selection easier to validate in pilot experiments. A dye that performs very well in one context may be a poor fit in another if the sample state, staining target, timing, or detection platform changes. Matching dyes to real staining scenarios is therefore one of the most effective ways to reduce false starts and improve experimental reproducibility.
Dyes for Live Cell Staining
Live-cell staining places the greatest emphasis on biological compatibility because the cells must remain intact enough for the observed signal to retain its meaning. In this scenario, the dye must do more than enter the sample and fluoresce. It must do so without causing excessive membrane disturbance, organelle stress, abnormal uptake behavior, or rapid signal decay during the actual observation window. This is why dyes chosen for live-cell staining are usually evaluated not only for brightness, but also for cytotoxicity, incubation speed, retention behavior, photostability, and compatibility with physiological media and imaging conditions.
In practical terms, live-cell dye selection depends heavily on what the experiment is trying to capture. If the goal is short-term structural visualization, a dye that labels quickly and cleanly with minimal perturbation may be sufficient. If the goal is dynamic observation, such as membrane trafficking, organelle movement, or real-time changes in cell state, the dye must also withstand repeated imaging and remain interpretable over time. This often favors fluorophores with stronger photostability and lower background accumulation. Researchers should also remember that live-cell stains are particularly sensitive to temperature, incubation duration, and light exposure, so a dye that seems promising in a static trial may still be suboptimal in a true time-lapse workflow. Another key consideration is whether the stain reports on structure or function. Some live-cell dyes simply provide localization, while others respond to membrane potential, intracellular polarity, uptake behavior, or other environmental variables. Those functional dyes can add more biological depth, but they also demand tighter control of experimental conditions because the signal may change for reasons unrelated to the main hypothesis. For live workflows, the best dye is therefore the one that provides usable signal with the least biological distortion under the actual imaging or analysis conditions being used.
Dyes for Fixed Cell Staining
Fixed-cell staining is usually selected when the workflow prioritizes structural preservation, intracellular accessibility, controlled endpoint analysis, or the ability to perform additional processing steps without the constraints of maintaining viable cells. In this scenario, dye selection shifts away from low-disturbance live compatibility and toward signal stability, fixation tolerance, resistance to washing, and compatibility with downstream permeabilization or multistep labeling. A dye that is difficult to use in living cells may become highly effective in fixed-cell analysis if it provides strong localization, stable fluorescence, and clean performance after processing. One of the main strengths of fixed-cell staining is that it allows broader access to intracellular targets. Because fixation can preserve architecture and permeabilization can open access to internal structures, researchers can use dyes that would otherwise not reach the intended compartment in live cells. This is especially relevant for nuclear, cytoskeletal, and certain organelle-oriented workflows where endpoint clarity matters more than dynamic observation. At the same time, fixed-cell dye selection still requires caution. Not every fluorophore retains its localization meaning after fixation, and not every dye tolerates processing equally well. Some signals remain stable and well defined, while others redistribute, weaken, or become harder to interpret after washing or chemical treatment.
Another important factor is timing flexibility. Because fixed samples no longer change biologically during acquisition, the protocol can often include more rigorous washing, stronger signal optimization, and longer imaging sessions. This tends to favor dyes that support reproducible structural contrast and cleaner endpoint analysis rather than rapid live compatibility. However, fixation can also create or amplify background, alter epitope exposure, and change how a dye interacts with cellular material. For that reason, fixed-cell dye selection should be based on whether the fluorophore remains meaningful after the full processing sequence, not just on how well it performed before the sample was fixed.
Dyes for Multicolor Cell Staining
In multicolor staining, the selected dye must work not only as an individual stain but as part of an integrated fluorescence panel. This changes the selection logic substantially. A dye that looks excellent in a single-color test may become a poor choice if it occupies a crowded channel, overwhelms weaker markers, or creates unnecessary bleed-through in adjacent detection windows. For this reason, multicolor dye selection should always be approached at the panel level rather than on a stain-by-stain basis. One of the first considerations in multicolor design is spectral spacing. Each dye should be assigned to a channel that can be excited and detected cleanly without causing preventable overlap with the rest of the panel. But channel separation alone is not enough. Relative brightness matters as well. Highly abundant targets can often be assigned to dimmer or more moderate fluorophores, while low-abundance targets may require brighter channels with less background competition. If this logic is ignored, the panel may become unbalanced, with one stain dominating the image while another becomes too weak to interpret confidently.
Researchers also need to consider staining function within the multicolor layout. A nuclear counterstain, for example, is often useful as a reference channel, but it should not occupy a channel needed for a weak organelle marker or a critical viability readout. Membrane stains, organelle dyes, and live/dead markers must all be evaluated not only for their own merit, but for how they influence the interpretability of the full image or population profile. In complex workflows, panel performance often depends more on overall coordination than on the theoretical quality of any single fluorophore. The most effective multicolor dye selections are usually built through hierarchy: define the essential targets first, assign the most sensitive or lowest-abundance readouts to the most favorable channels, then fit reference stains and broader structural dyes into the remaining spectral space. Controls are especially important here because multicolor panels can fail silently. A stain may appear acceptable in isolation but distort the whole experiment through overlap, compensation burden, or channel competition. This is why multicolor dye choice should always be validated with a panel-aware mindset rather than a catalog-first mindset.
Dyes for Organelle Visualization
Organelle visualization requires dyes whose targeting logic is closely matched to the biology of the compartment being studied. Unlike general structural stains, organelle dyes must do more than create intracellular fluorescence. They need to localize with sufficient selectivity that the observed signal can reasonably be interpreted as reporting on a specific compartment such as mitochondria, lysosomes, endoplasmic reticulum, Golgi structures, lipid-rich domains, or the nucleus. Because different organelles differ in membrane composition, pH environment, electrochemical properties, and trafficking behavior, the choice of dye must be guided by how the fluorophore reaches and remains associated with the target compartment.
This makes organelle dye selection particularly sensitive to workflow context. Some organelle-targeted dyes are best suited to live-cell use because their localization depends on active physiological properties such as membrane potential or acidic compartment accumulation. Others perform better in fixed or endpoint analysis because the experiment prioritizes structural preservation over dynamic function. A mitochondrial dye, for example, may provide strong localization in live cells but lose interpretive value after fixation if its signal depends on a functional state rather than a permanent structural anchor. Similarly, a lysosome-oriented probe may highlight acidic compartments well in living cells but become less informative if the assay design changes the underlying environment it depends on.
Dyes for Viability Assessment and Cell Tracking
Viability and tracking workflows require especially careful alignment between dye mechanism and experimental purpose because, in both cases, the signal is expected to support interpretation over time or across changing cell states. For viability staining, the dye should reflect the biological criterion that defines whether a cell is considered live, dead, or compromised in the context of the assay. For cell tracking, the dye should persist long enough to remain informative without transferring excessively to neighboring cells or becoming so diluted that the signal no longer reflects lineage or movement meaningfully. In viability assessment, dye choice depends on whether the workflow needs to identify dead cells directly, label viable cells positively, or preserve viability information into later processing. Some experiments only need rapid dead-cell exclusion, while others need more nuanced interpretation of cell integrity under treatment or stress. In these cases, the best dye is the one whose staining logic matches the decision the researcher needs to make. A membrane-impermeant dead-cell dye may be ideal for quick quality control, but a different strategy may be required if the experiment includes fixation, multiparameter analysis, or dynamic live-cell observation. This is why viability dyes should never be selected simply because they are widely used; they must fit the workflow definition of interpretable cell status.
Both viability and tracking applications also interact strongly with multicolor design. A viability stain should not consume a critical channel needed for a weak biological marker unless that tradeoff is justified. A tracking dye should be placed in a channel that remains interpretable over time and does not interfere excessively with downstream stains. In both cases, the dye must be evaluated not just for optical properties, but for how its biological behavior supports the intended analytical conclusion. That is why these scenarios benefit particularly from pilot validation before the full experiment is scaled.
How BOC Sciences Supports Fluorescent Dyes for Cell Staining?
BOC Sciences supports fluorescent dye-based cell staining projects through integrated capabilities in product supply, custom probe development, application support, and quality-oriented production services. Rather than treating fluorescent dyes as simple labeling tools, we help researchers align dye type, spectral properties, targeting behavior, and workflow requirements with the actual goals of the experiment. This is especially important in cell staining workflows, where signal quality, dye selectivity, cell compatibility, and long-term supply reliability all influence whether the final staining strategy remains practical and reproducible. Our support is therefore designed to cover both routine dye selection needs and more advanced fluorescence development and application requirements.
Broad Fluorescent Dye Product Supply
- Access to a broad range of fluorescent dye types, including organic small-molecule dyes, quantum-dot-related fluorescent materials, and functional fluorescent probes relevant to diverse cell staining applications.
- Support for selecting fluorophores across multiple wavelength regions, including UV, visible, and near-infrared fluorescence, to better match different imaging systems and analytical requirements.
- Availability of dyes designed for specific cellular targets, including nuclei, mitochondria, lysosomes, and other intracellular structures commonly used in fluorescence-based cell studies.
- Product supply supported by high-purity standards and attention to batch consistency, helping improve reproducibility in repeated staining and imaging workflows.
Customized Fluorescent Probe Development Services
- Custom design of fluorescent probes with specific targeting properties according to research needs, including probes directed toward proteins, nucleic acids, or defined cellular compartments.
- Structural modification and functional optimization of dye molecules to improve properties such as stability, brightness, selectivity, and broader workflow compatibility.
- Support for fluorescent conjugation services, including coupling fluorescent dyes to antibodies, peptides, small-molecule ligands, or other targeting components used in specialized staining and detection strategies.
- Integrated custom development services covering molecular design, synthesis, modification, purification, and related preparation steps for tailored fluorescent probe solutions.
Application Support and Technical Consultation
- Practical guidance for designing and optimizing cell staining workflows according to experimental purpose, target structure, and imaging platform.
- Support for fluorescent dye selection based on key factors such as excitation and emission matching, cell permeability, staining logic, and compatibility with live-cell or fixed-cell workflows.
- Troubleshooting assistance for common fluorescence challenges, including signal quenching, high background, weak contrast, or reduced localization clarity in imaging experiments.
- Availability of product use guidance and related technical documentation to support evaluation, implementation, and more efficient workflow planning.
Quality Control and Scalable Production Support
- Fluorescent dye products supported by strict quality control systems, with analytical methods such as HPLC, NMR, and MS used where applicable to verify quality and composition.
- Availability of detailed analytical reports and quality-related documentation, including COA and related supporting materials, to facilitate product evaluation and project use.
- Production support spanning from early-stage research quantities to larger-scale supply needs, helping match different phases of fluorescence development and application.
- Long-term supply capabilities designed to support project continuity, batch stability, and more reliable execution of ongoing fluorescence programs.
Do You Need A Consultation?
BOC Sciences integrates cutting-edge fluorescence technologies to accelerate your research, driving next-generation solutions for drug discovery and diagnostics.
Transform Your Studies with Cutting-Edge Fluorescent Products
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0052 | C6 NBD Lactosylceramide | 474943-04-9 | Bulk Inquiry |
| A16-0051 | C12 NBD Lactosylceramide | 474943-06-1 | Bulk Inquiry |
| A16-0064 | ReZolve-ER™ | 1404104-40-0 | Bulk Inquiry |
| A16-0150 | 3,3'-Diethyloxacarbocyanine iodide | 905-96-4 | Bulk Inquiry |
| A16-0172 | DiBAC4(3) | 70363-83-6 | Bulk Inquiry |
| A16-0031 | DISBAC10 | 169211-45-4 | Bulk Inquiry |
| A16-0221 | TO-PRO 1 | 157199-59-2 | Bulk Inquiry |
| A16-0083 | Lucifer yellow ethylenediamine | 161578-11-6 | Bulk Inquiry |
| A16-0022 | Fluorescein-5-thiosemicarbazide | 76863-28-0 | Bulk Inquiry |
High-Performance Fluorescent Tools for Your Research
- Nitric Oxide (NO) & Reactive Oxygen Species (ROS) Probes for oxidative stress and signaling detection.
- Cytoskeleton Fluorescent Probes Probes for actin and microtubule visualization.
- Nerve Terminal Probes Fluorescent tracers for synaptic activity analysis.
- Mitochondrial Fluorescent Probes Targeted dyes for mitochondrial structure and function.
- Nuclear Fluorescent Probes DNA-binding dyes for nucleus visualization.
- Ion Fluorescent Probes Indicators for real-time ion concentration imaging.
- Endoplasmic Reticulum Fluorescent Probes ER-targeted dyes for organelle structure analysis.
- Fluorescent Probes Versatile tools for biomolecular and cellular imaging.
Explore More Topics
Online Inquiry


