Cell and Organelle Stains
- Apoptosis Fluorescent Probes
- Cell membrane Fluorescent Probes
- Cell Proliferation Tracer Fluorescent Probes
- Cytoskeleton Fluorescent Probes
- Endoplasmic Reticulum Fluorescent Probes
- Golgi Fluorescent Probes
- Lipid Fluorescent Probes
- Lysosomal Fluorescent Probes
- Mitochondrial Fluorescent Probes
- Nerve Terminal Probes
- Nuclear Fluorescent Probes
- Other Cell Fluorescent Probes
Customized Fluorescent Reagents
One-stop Solution for Your Research
BOC Sciences offers a one-stop solution for fluorescent reagents, providing custom synthesis, modification, and large-scale production services. Our comprehensive portfolio includes high-purity fluorescent dyes, probes, and labeling reagents for research and industrial applications, ensuring superior performance and reliability.
Explore More

Background
Cell and organelle stains are essential tools for visualizing cellular structures, assessing physiological states, and designing fluorescence-based workflows with greater precision. In practice, this topic extends well beyond simply applying a dye to a sample. Researchers often need to decide whether the experiment requires live-cell compatibility or post-fixation structural preservation, whether the goal is morphology, viability, localization, trafficking, or proliferation analysis, and whether one channel or multiple channels must be coordinated within the same imaging workflow. Because these choices directly influence staining quality, interpretability, and downstream reproducibility, stain selection should connect staining strategy, dye selection, organelle targeting, troubleshooting, and workflow planning rather than treating them as isolated decisions.
At BOC Sciences, we support cell staining projects with a broad portfolio of fluorescent dyes and probes for nuclear staining, mitochondrial imaging, lysosome labeling, membrane visualization, endoplasmic reticulum and Golgi staining, cytoskeleton studies, cell viability analysis, and longitudinal cell tracking. The following content is structured to help users understand what cell and organelle stains are, how to choose them more rationally, and which technical factors should guide the move from general knowledge to practical stain and probe selection.
What Are Cell and Organelle Stains?
Cell and organelle stains are fluorescent or chromogenic agents used to reveal cellular architecture, intracellular localization, or condition-dependent biological signals. Some stains function by binding specific biomolecular targets such as DNA, proteins, lipids, or glycoconjugates, while others accumulate in cellular compartments because of membrane permeability, pH partitioning, membrane potential, or affinity for particular organelle environments. This is why not all stains should be treated as interchangeable. A nuclear stain and a mitochondrial stain may both generate bright fluorescence, but they rely on very different localization logic and support different types of interpretation.
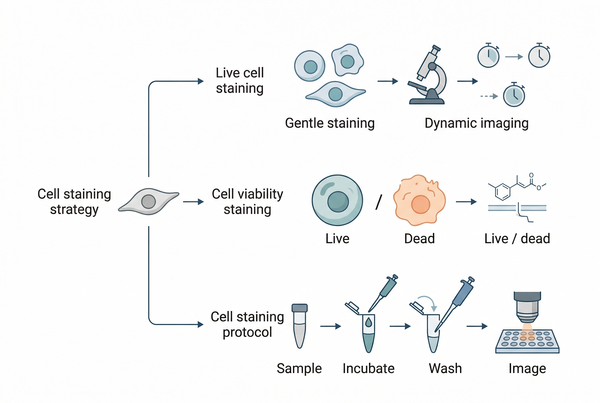 Fig. 1. Cell and organelle staining overview (BOC Sciences Authorized).
Fig. 1. Cell and organelle staining overview (BOC Sciences Authorized).
How Cell and Organelle Stains Support Imaging and Functional Analysis?
Cell staining can support several different readout types within one research area. Structural staining helps reveal morphology, compartment boundaries, and subcellular organization. Localization staining helps determine where a biomolecule, organelle, or pathway-associated signal is enriched. Functional staining is often more conditional, relying on membrane integrity, membrane potential, pH, esterase activity, or intracellular retention. As a result, a successful stain is not defined only by brightness, but also by whether the signal genuinely reflects the biological feature being measured.
Why the Same Stain Is Not Suitable for Every Experiment?
Stain suitability depends on sample type, treatment conditions, imaging duration, fixation status, spectral design, and interpretation goals. A dye that performs well in endpoint fixed-cell imaging may not remain selective in live cells. Likewise, an organelle probe that works in healthy cells may shift localization when membrane potential collapses, lysosomal acidity changes, or membrane trafficking is perturbed. This is why a topic-level framework is useful: it helps users choose stains according to mechanism, workflow fit, and application boundary rather than by fluorescence color alone.
Choosing the Right Cell Staining Strategy for Your Experiment
Before selecting a specific dye, it is usually more productive to define the staining strategy. In most cases, the highest-value decision is not "Which fluorophore is brightest?" but "What type of biological information should the staining workflow preserve?" Strategy choices influence permeability, wash conditions, incubation timing, fixation compatibility, image acquisition settings, and whether a signal can be interpreted as static structure or dynamic function.
Live Cell Staining vs Fixed Cell Staining Strategies
The choice between live-cell and fixed-cell staining should be made early because it changes nearly every downstream decision. Live-cell staining is more suitable when researchers need dynamic observation, physiological monitoring, trafficking analysis, or time-course imaging. Fixed-cell staining is generally preferred when structural preservation, stronger permeabilization control, extended imaging, or higher compatibility with antibody-based workflows is required. The trade-off is that live-cell staining better preserves temporal biology, whereas fixed-cell staining often improves consistency of localization and access to intracellular targets.
Cell Viability Staining Methods and Selection Factors
Viability staining is not simply a subtype of general cell staining. It is a readout-driven method designed to distinguish intact, damaged, dying, or dead cells on the basis of membrane integrity, esterase conversion, exclusion behavior, or apoptotic membrane changes. Selection should depend on whether the experiment requires rapid live/dead discrimination, multiparameter analysis, endpoint microscopy, or compatibility with follow-up tracking or flow-based analysis. A good viability stain must be interpreted in the context of assay timing, cell stress, and signal reversibility.
Cell Staining Protocol Design for Reliable Results
Even when the correct stain is selected, staining outcomes can fail if the protocol design is not matched to the assay. Reliable workflows usually depend on controlling concentration ranges, incubation time, solvent exposure, wash stringency, temperature, serum conditions, imaging delay, and the order of multistep staining. Protocol design should reduce background without stripping weak but real signals, and should preserve enough consistency that differences between samples can still be interpreted biologically rather than procedurally.
Multicolor Cell Staining and Panel Design Basics
Multicolor staining requires more than assigning different colors to different targets. Panel design should consider spectral separation, expected signal intensity, channel bleed-through, organelle overlap, acquisition order, and the likelihood that one stain changes the performance of another. A strong panel balances fluorophore brightness with target abundance and avoids using physiologically sensitive dyes in combinations that complicate interpretation. The best multicolor workflow is usually the one that minimizes ambiguity, not the one that uses the most channels.
Fluorescent Dye Selection for Cell Staining Applications
After the workflow type is defined, fluorescent dye selection becomes more rational. Dye choice should link target accessibility, subcellular localization mechanism, spectral compatibility, photostability, background behavior, and retention characteristics. Researchers often focus heavily on excitation and emission wavelengths, but spectral fit is only one part of a broader selection framework. In many staining workflows, permeability, organelle selectivity, quenching sensitivity, and resistance to washout are equally important. A practical dye selection workflow usually starts with the biological target, then moves to sample state, imaging instrument, and assay duration. For example, if the signal must be retained in living cells over time, dye retention and cytotoxicity become more important than maximal brightness. If the main objective is fixed-cell structural contrast, permeability becomes less restrictive, while fixation stability and low off-target staining become more important. Matching the stain to the experiment means balancing specificity, usability, and interpretive reliability rather than optimizing one property in isolation.
| Selection Dimension | Why It Matters | Questions to Ask |
| Target mechanism | Determines whether staining reflects binding, partitioning, accumulation, or physiological state | Does the signal represent a structure, a condition, or both? |
| Cell permeability | Influences whether live-cell access is possible without fixation | Can the dye enter intact cells under workable conditions? |
| Retention and washout | Affects long-term imaging and signal stability | Will the dye remain localized during washing and imaging? |
| Photostability | Matters in repeated acquisition and multichannel imaging | Will the signal decay too quickly under illumination? |
| Background tendency | Shapes contrast and interpretability | Is nonspecific signal likely in the cytoplasm or membranes? |
| Spectral compatibility | Essential for co-staining and panel design | Can the dye fit the instrument and other fluorophores? |
Matching Stains to Experimental Goals Improves Workflow Quality
The most effective cell staining workflows begin with the biological question. Whether the goal is morphology, organelle localization, viability, multicolor colocalization, proliferation analysis, or long-term tracking, stain selection should follow the experimental endpoint rather than the other way around. Aligning assay goals with stain mechanism, imaging duration, and interpretation limits helps reduce background, avoid misleading signals, and improve reproducibility across samples.
Matching Cell and Organelle Stains to Experimental Goals
Many users begin by asking which stain is best, but a more accurate starting point is to ask which readout the experiment must support. Stains that are effective for morphology may not be suitable for functional monitoring, and dyes that perform well in a short endpoint assay may not remain interpretable in longitudinal imaging. Defining the goal first helps narrow the stain class, the protocol style, and the level of interpretation that can be defended from the data.
Cell Morphology and Localization Staining Goals
If the primary need is to visualize shape, boundaries, nuclear distribution, or the relative position of intracellular compartments, the most useful stains are those that provide crisp structural contrast and minimal positional ambiguity. In this context, fixation-compatible stains and strongly localized probes often outperform more physiologically sensitive dyes because they simplify interpretation.
Functional Readouts and Organelle State Assessment
When the experiment depends on changes in membrane potential, acidity, enzymatic conversion, or intracellular stress responses, the stain becomes part of the assay logic rather than a passive marker. These workflows demand extra care because the fluorescence signal may shift when the organelle or cell state changes, which means signal intensity and localization must be interpreted together.
Endpoint Imaging vs Dynamic Monitoring Needs
Endpoint imaging usually tolerates more aggressive staining conditions and longer incubations because the sample is read once. Dynamic monitoring requires gentler conditions, higher retention stability, lower toxicity, and imaging-compatible signal persistence. A dye that is acceptable for one time point may be too disruptive or unstable for repeated imaging.
Single-Target Staining vs Multicolor Colocalization Design
Single-target staining can prioritize maximal contrast, while multicolor designs must reserve spectral space, manage brightness hierarchy, and avoid overlap between structurally adjacent signals. This is especially important when membrane, cytoskeletal, and organelle probes are used in the same sample, because apparent colocalization may arise from resolution limits or bleed-through rather than true biological overlap.
Organelle Cell Staining for Major Intracellular Targets
Organelle staining depends on how accurately a dye can distinguish one intracellular environment from another. Some probes rely on target abundance or binding affinity, while others depend on compartmental properties such as membrane potential, lipid composition, pH gradients, or trafficking dynamics. The following target-specific overview helps users understand where stain choice must be adapted rather than generalized across all organelles.
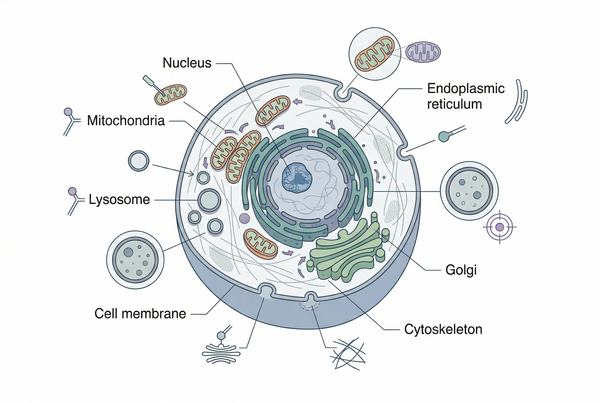 Fig. 2. Major organelles require different staining strategies (BOC Sciences Authorized).
Fig. 2. Major organelles require different staining strategies (BOC Sciences Authorized).
Nuclear Staining in Cells: Selection and Use Cases
Nuclear stains are widely used because the nucleus provides a stable and interpretable positional landmark. However, the most appropriate dye still depends on whether live-cell compatibility, DNA selectivity, chromatin contrast, or compatibility with other channels is more important. In multicolor workflows, nuclear stains often anchor segmentation and localization analysis, so their channel placement and brightness should be planned carefully.
Mitochondrial Staining in Cells: Dye and Workflow Considerations
Mitochondrial staining is especially sensitive to assay context because many common probes accumulate according to membrane potential. This makes them valuable for functional studies, but also means signal changes may reflect altered physiology rather than simple changes in organelle abundance. Workflow design should therefore distinguish structural visualization from functional interpretation whenever possible.
Lysosome Staining in Cells: Probe Choice and Signal Interpretation
Lysosome-associated probes are often selected for their ability to accumulate in acidic compartments, but this same mechanism can create interpretive challenges when lysosomal pH shifts or when staining spills into related vesicular structures. Strong lysosomal imaging depends on understanding both localization and the biological conditions that support or disrupt probe retention.
Cell Membrane Staining: Dye Options and Key Considerations
Membrane stains are useful for outlining cells, tracking morphology, studying vesiculation, and supporting segmentation in complex samples. Selection should account for how tightly the dye associates with lipid bilayers, whether membrane internalization is acceptable, how long the label needs to persist, and whether the workflow includes fixation or long-term imaging.
Endoplasmic Reticulum Staining: Probe Selection and Workflow Design
ER staining typically requires probes that provide good reticular contrast without overwhelming the cytoplasm. Because the ER spans broad intracellular regions and interacts with multiple organelles, background control and imaging resolution become especially important. Probe choice should also account for whether the study focuses on general architecture or stress-related ER changes.
Golgi Fluorescent Staining: Probe Selection and Troubleshooting
Golgi staining can be informative but is often challenging because compact juxtanuclear localization may be disrupted by cell type, fixation conditions, trafficking perturbations, or overloading of nonspecific membrane-associated dyes. Good Golgi imaging usually requires careful balance between selectivity, resolution, and acquisition settings.
Cytoskeleton Staining: Probe Selection and Workflow Planning
Cytoskeletal staining supports analysis of morphology, motility, polarity, and intracellular organization. Because actin- and tubulin-associated structures can be highly dynamic and mechanically sensitive, probe selection should reflect whether the experiment prioritizes stable structural imaging or more condition-sensitive observations. Fixation, extraction, and imaging order may all influence the final pattern.
Lipid Staining: Principles, Methods, Dyes, and Uses
Lipid staining is especially useful when the experiment needs to visualize lipid-rich structures, neutral lipid accumulation, or intracellular lipid droplets that are difficult to resolve with conventional hydrophilic stains. Because lipid labeling depends on the lipophilic behavior of the dye and the hydrophobic nature of the target, stain choice should account for whether the workflow emphasizes general lipid distribution, lipid droplet visualization, or fluorescence-based detection in more dynamic cell imaging settings.
Cell Tracking and Proliferation Staining for Dynamic Studies
Dynamic studies introduce a different set of priorities from static staining. Instead of maximizing short-term contrast, the workflow must preserve detectable signal through time, dilution, or cell movement while minimizing toxicity and false redistribution. This is why proliferation tracers and cell tracking dyes should be viewed as purpose-built tools rather than extensions of general morphology stains.
Cell Proliferation Tracer Selection and Staining Principles
Proliferation tracers are designed to support lineage expansion and division-related signal analysis rather than simple localization. They are most useful when the workflow needs to distinguish actively dividing populations, compare proliferation history, or integrate growth behavior into a broader fluorescence assay. Selection should consider signal dilution behavior, readout window, and compatibility with viability and phenotyping strategies.
Cell Tracking Dyes for Longitudinal Cell Labeling
Cell tracking dyes are more appropriate when the aim is to follow movement, migration, distribution, or persistence across time. The ideal dye should remain associated with the labeled population for the required observation window without producing major physiological disturbance. Signal retention, transfer risk, and dilution behavior should all be evaluated before the workflow is finalized.
Factors That Influence Cell Staining Performance
Staining quality is determined not only by the stain itself but by the conditions under which it is used. Two users can apply the same dye to the same nominal target and still obtain very different results because concentration, incubation time, buffer composition, wash steps, imaging delay, and sample health alter how fluorescence is generated and retained. Understanding these variables before problems arise is one of the most effective ways to improve reproducibility.
| Performance Factor | Typical Effect on Results | Optimization Priority |
| Dye concentration | Too low reduces contrast; too high raises background and aggregation risk | Titrate around the minimal effective range |
| Incubation time | Insufficient loading weakens signal; excessive loading can distort localization | Balance uptake with specificity |
| Wash conditions | Poor washing leaves background; over-washing can strip weak true signal | Adjust wash number and buffer strength |
| Cell condition | Stress alters permeability, organelle state, and retention | Assess viability and treatment burden |
| Imaging settings | Overexposure and repeated illumination accelerate photobleaching | Use conservative acquisition settings |
| Assay timing | Delay between staining and imaging may change localization or intensity | Standardize readout timing |
Dye Concentration, Incubation, and Background Control
Staining conditions should be optimized to maximize meaningful contrast rather than absolute intensity. High concentration and extended incubation can create bright images, but they may also increase nonspecific binding, cytoplasmic haze, membrane overloading, or abnormal compartment accumulation. Titration is therefore a central step in robust workflow design.
Permeability, Retention, and Localization Behavior
Some probes enter cells rapidly but wash out easily, while others remain stable only in certain physiological states. Interpreting localization requires understanding whether the stain is directly bound to a target or merely enriched in a favorable environment. This difference affects how confidently users can relate fluorescence patterns to real subcellular distribution.
Imaging Settings, Photobleaching, and Reproducibility
A stain that appears weak may actually be suffering from acquisition-related loss rather than poor target engagement. Illumination intensity, detector gain, exposure length, and repeated channel cycling all influence signal quality. Reproducibility improves when imaging parameters are standardized alongside staining parameters rather than treated as independent steps.
Sample State, Treatment Effects, and Signal Interpretation
Cell staining results are strongly influenced by the biological state of the sample before and during staining. Drug treatment, metabolic stress, apoptosis, altered membrane integrity, or organelle dysfunction can all change dye uptake, retention, redistribution, or apparent target selectivity. For this reason, fluorescence patterns should always be interpreted together with sample condition, especially when using condition-sensitive stains for mitochondria, lysosomes, viability, or membrane-associated readouts.
Looking for Expert Guidance?
BOC Sciences supports cell and organelle staining projects with fluorescent dye options, target-oriented probe selection, multicolor workflow planning, and staining optimization guidance for imaging-based studies.
Common Problems in Cell Staining and Troubleshooting Priorities
Troubleshooting works best when problems are classified according to the type of failure rather than by repeating the full protocol without a diagnosis. Weak signal, high background, poor compartment contrast, signal drift, crosstalk, and misleading localization do not usually arise from the same root cause. A practical troubleshooting sequence starts by separating staining failure from imaging failure, then evaluates concentration, timing, sample condition, and dye-target compatibility in that order.
Weak Signal and Poor Contrast
Weak signal often results from insufficient loading, premature washout, low target abundance, or overly conservative acquisition settings. It can also occur when a live-cell stain is used under conditions that suppress the physiological state needed for accumulation. The right fix depends on whether the problem is dye delivery, target engagement, or imaging loss.
High Background and Misleading Localization
Background problems are commonly caused by excess stain, incomplete washing, nonspecific membrane association, or channel bleed-through. Apparent localization can also become misleading when organelle state changes alter dye partitioning. In these cases, stronger signal is not necessarily better signal, and cleaner contrast often requires milder staining rather than more stain.
Crosstalk, Channel Overlap, and Multicolor Interference
In multicolor workflows, apparent signal overlap may come from spectral bleed-through, imbalanced brightness, or unsuitable acquisition order rather than true colocalization. Troubleshooting should check filter compatibility, fluorophore separation, detector settings, and whether highly abundant targets are overwhelming weaker channels. Cleaner panel design often improves interpretability more effectively than simply increasing exposure or staining intensity.
Inconsistent Staining Between Samples
Variability across samples is often caused by differences in cell density, treatment response, incubation timing, wash consistency, or sample health before staining begins. Even small shifts in handling can alter permeability, uptake, and retention, especially for condition-sensitive dyes. Standardizing workflow order, timing, and imaging conditions is essential when staining results must be compared across groups.
How BOC Sciences Supports Cell and Organelle Staining Workflows?
BOC Sciences supports cell and organelle staining workflows through a combination of standard stain and probe supply, custom development options, workflow-oriented technical guidance, and documentation support. Because these experiments often depend on target specificity, live- or fixed-cell compatibility, multicolor design, and reliable signal interpretation, effective support should address both reagent selection and experimental workflow planning. Our goal is to help researchers choose and apply cell and organelle stains in ways that fit the biological target, imaging strategy, and downstream readout rather than treating all fluorescent stains as interchangeable.
Standard Cell and Organelle Stain Supply
- Supply of fluorescent stains and probes relevant to nuclear staining, mitochondrial staining, lysosome labeling, membrane visualization, endoplasmic reticulum staining, Golgi staining, cytoskeleton imaging, cell viability analysis, and dynamic cell labeling workflows.
- Product options can be matched to different staining purposes such as morphology visualization, intracellular localization, organelle targeting, live/dead discrimination, proliferation analysis, and longitudinal tracking studies.
- Coverage of multiple spectral regions to support single-color assays, multicolor staining panels, and instrument-specific channel planning.
- Product availability suited to exploratory studies, comparative staining evaluation, workflow optimization, and broader fluorescence-based cell analysis applications.
Custom Stain and Probe Development
- Support for customizing fluorophore choice, probe structure, and related design elements according to target localization, retention behavior, and assay-specific workflow needs.
- Flexible development routes for projects requiring alternative spectral placement, modified signal properties, or more tailored compatibility with specific cell systems and imaging conditions.
- Practical support for aligning stain design with organelle targeting, multicolor imaging, viability workflows, or dynamic monitoring studies.
- Development approaches that connect reagent chemistry with the intended biological application rather than optimizing only for brightness or nominal staining efficiency.
Workflow Guidance for Cell Staining Studies
- Guidance on stain selection, incubation strategy, wash conditions, imaging timing, and workflow order to improve staining specificity and signal interpretability.
- Support for matching stain behavior to live-cell imaging, fixed-cell analysis, multicolor staining, organelle-specific workflows, and follow-up fluorescence readouts.
- Assistance with reducing background ambiguity, improving channel separation, and strengthening reproducibility in comparative staining experiments.
- More structured planning for workflows that combine cell and organelle staining with broader fluorescence imaging, tracking, or assay development strategies.
Quality and Documentation Support
- Quality-related documentation support to help researchers evaluate reagent consistency and suitability for cell and organelle staining studies.
- Assistance with specifications relevant to purity, storage, handling, and fluorescence-related use considerations for staining reagents.
- Availability of structured documentation support such as COA- and SDS-related materials where needed.
- Better traceability for laboratories that require organized documentation alongside technical workflow planning and reagent selection.
Explore More Topics
Resources

- Hoechst Dyes: Definition, Structure, Mechanism and Applications
- Mastering the Spectrum: A Comprehensive Guide to Cy3 and Cy5 Dyes
- Fluorescent Probes: Definition, Structure, Types and Application
- Fluorescent Dyes: Definition, Mechanism, Types and Application
- Coumarin Dyes: Definition, Structure, Benefits, Synthesis and Uses
- Unlocking the Power of Fluorescence Imaging: A Comprehensive Guide
- Cell Imaging: Definitions, Systems, Protocols, Dyes, and Applications
- Lipid Staining: Definition, Principles, Methods, Dyes, and Uses
- Flow Cytometry: Definition, Principles, Protocols, Dyes, and Uses
- Nucleic Acid Staining: Definition, Principles, Dyes, Procedures, and Uses
Online Inquiry


