Cell Viability Staining: Fluorescent Methods to Distinguish Live and Dead Cells
Cell staining workflows often depend on one basic question before any target-specific analysis begins: are the cells alive, dead, or in a compromised transitional state that could distort the readout? That is why cell viability staining is one of the most important control layers in fluorescence-based experiments. A viability stain does more than separate intact cells from damaged cells. It helps researchers judge sample quality, improve data interpretation, reduce false positives from non-specific fluorescence, and decide whether a downstream image, population analysis, or multicolor panel is biologically meaningful. In practical terms, viability information often determines whether the rest of the experiment can be trusted.
In fluorescence workflows, the distinction between live and dead cells is usually made through membrane integrity, intracellular enzyme activity, nucleic acid accessibility, or reactivity with intracellular amines after damage. Different probes exploit these differences in different ways. Some stains label only cells with disrupted membranes, while others accumulate in viable cells that maintain esterase activity or intact membrane barriers. Some methods are optimized for rapid imaging, while others are better suited for endpoint analysis or compatibility with flow cytometry. Because of these differences, there is no single universal viability dye. The right choice depends on the sample, the detection platform, the panel design, and whether the experiment remains live or transitions into a fixed workflow.
What Is Cell Viability Staining?
Before choosing a fluorescent viability dye, it is important to define what cell viability staining actually means and what it does not mean. In fluorescence experiments, viability staining refers to the use of fluorescent dyes or fluorescent probes that distinguish living cells from dead or severely damaged cells based on biological properties that differ between those states. These properties most commonly include membrane integrity, intracellular esterase activity, nucleic acid accessibility, or differential chemical reactivity under live versus damaged conditions. The central purpose is not simply to create contrast, but to classify cells according to whether they still maintain essential structural and functional integrity at the time of analysis.
Basic Definition of Cell Viability Staining
Cell viability staining is a fluorescence-based method used to identify whether cells remain intact and functionally viable or whether they have lost membrane integrity and entered a non-viable state. In most workflows, live cells exclude certain impermeant dyes, while dead cells permit those dyes to enter and bind intracellular components such as nucleic acids. Other approaches work in the opposite direction by labeling living cells through active intracellular conversion or retention mechanisms that are no longer effective once cells lose metabolic integrity. In both cases, the fluorescent signal is interpreted in relation to cell status rather than merely target abundance. This definition matters because viability staining is often confused with broader measures of cell health. A sample may contain cells that are alive but stressed, cells that are alive but metabolically slowed, cells that are apoptotic but not yet fully permeable, and cells that are clearly dead. A useful viability stain helps separate at least some of these categories, but it should not be expected to answer every biological question about cell state on its own. That is why viability readouts are most valuable when interpreted together with sample handling history, treatment conditions, and the rest of the fluorescent panel.
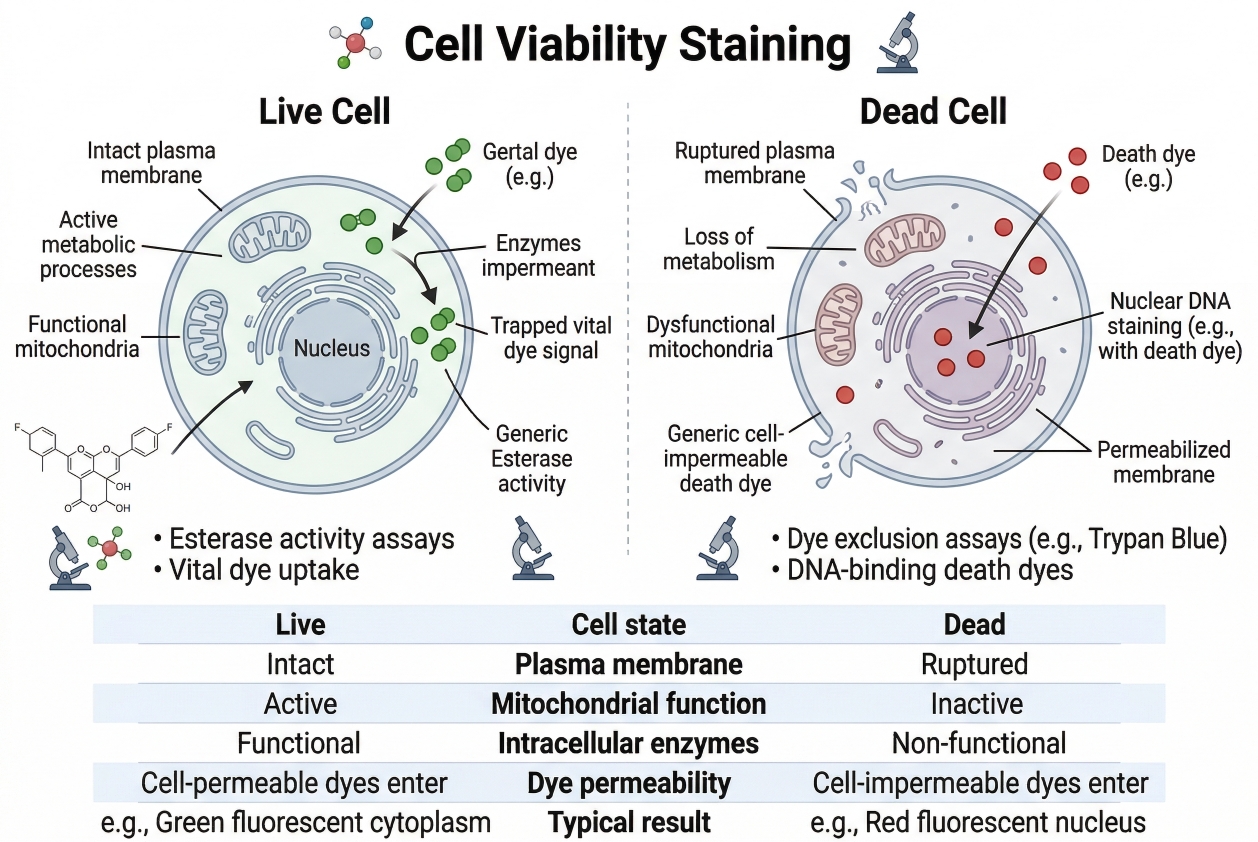 Fig. 1. Fluorescent staining principles for live and dead cell discrimination (BOC Sciences Authorized).
Fig. 1. Fluorescent staining principles for live and dead cell discrimination (BOC Sciences Authorized).
What "Live," "Dead," and "Compromised" Mean in Fluorescence Assays?
In fluorescence assays, a live cell is generally understood as one that maintains membrane integrity, excludes membrane-impermeant dyes appropriately, and preserves enough intracellular function to support the biological process under study. A dead cell is one that has lost the barrier properties and internal integrity required to maintain selective exclusion, leading to accessibility for dyes that should otherwise remain outside. A compromised cell lies between these extremes. It may still appear morphologically intact, yet begin to show altered permeability, abnormal dye uptake, or reduced metabolic activity. These transitional cells are one reason viability analysis requires careful interpretation rather than a simplistic binary assumption.
Why Viability Staining Is Not the Same as Proliferation or Metabolic Activity Measurement?
A common conceptual mistake is to treat viability staining as interchangeable with proliferation assays or broader metabolic activity assays. They are related, but they are not identical. A proliferating cell population may include dead cells, and a non-proliferating population may still be highly viable. Likewise, metabolic activity can decrease without full membrane failure, and some viability dyes respond more directly to membrane integrity than to proliferation status. This is why cell viability staining should be treated as its own analytical layer. It tells you whether the sample remains interpretable in terms of live/dead discrimination, not whether every aspect of cell physiology is unchanged.
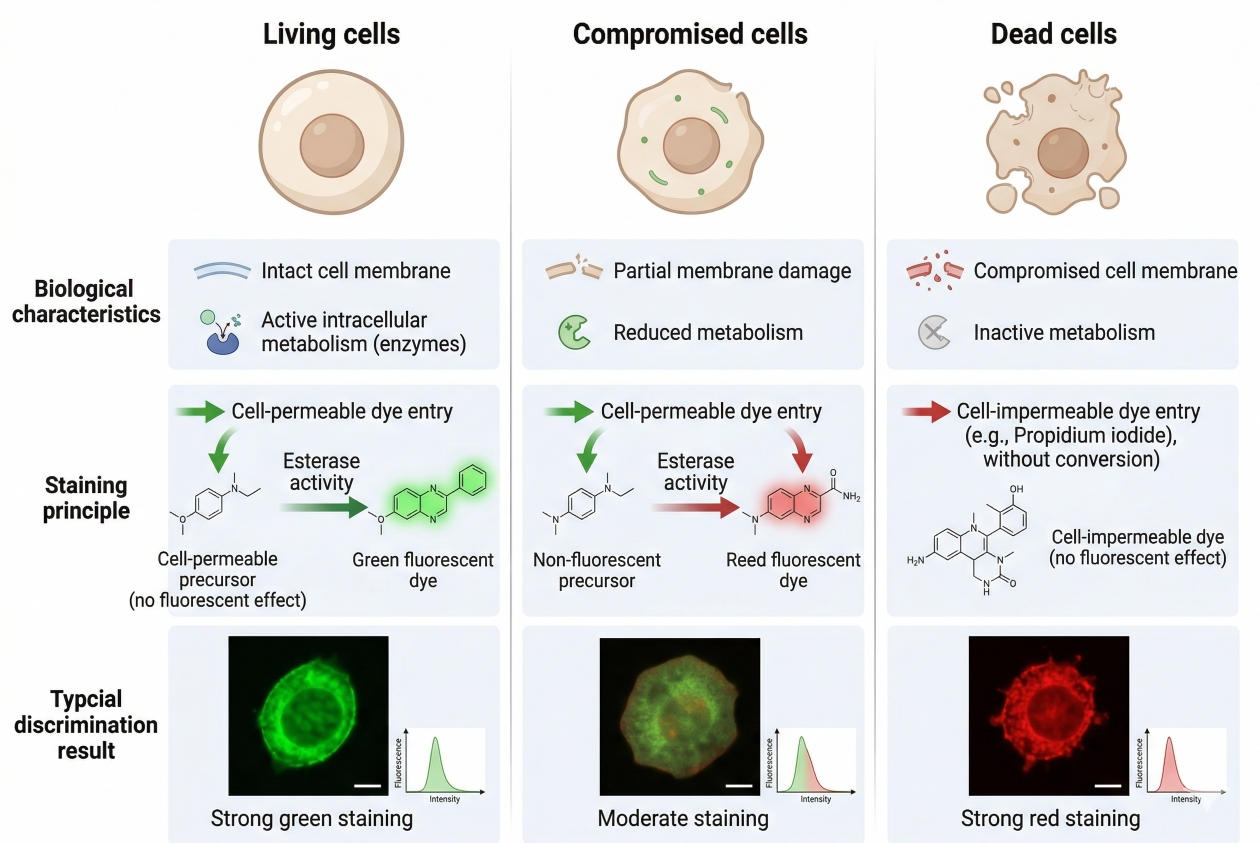 Fig. 2. Cell viability staining distinguishes live, dead, and compromised cells based on fluorescence-linked biological differences such as membrane integrity and intracellular activity (BOC Sciences Authorized).
Fig. 2. Cell viability staining distinguishes live, dead, and compromised cells based on fluorescence-linked biological differences such as membrane integrity and intracellular activity (BOC Sciences Authorized).
Why Cell Viability Staining Matters in Cell Analysis?
Viability staining is often treated as a routine control, but in many experiments it is much more than that. It can determine whether image quality is interpretable, whether a cell population has been damaged during preparation, whether treatment effects are biologically meaningful, and whether downstream quantification is being distorted by dead-cell-associated signal. In fluorescence-based experiments, non-viable cells frequently bind dyes in abnormal ways, generate elevated background, or accumulate probes that were not intended to report on the target of interest. This makes viability staining fundamental to both data quality and biological credibility.
Why Viability Is a Critical Control in Fluorescence Experiments?
In multistep experiments, dead cells often behave differently from viable cells in ways that extend beyond simple loss of function. They may become more permeable to reagents, expose intracellular binding sites unexpectedly, or retain fluorescent molecules non-specifically. Without viability discrimination, these cells can appear highly positive in a way that falsely suggests target enrichment. As a result, the absence of a viability control can exaggerate treatment effects, inflate apparent signal intensity, and make population-level comparisons much less reliable. This issue is especially important when the experimental outcome depends on accurate single-cell interpretation, quantitative image analysis, or population gating. Dead cells can contribute disproportionate fluorescence simply because they no longer regulate uptake or exclusion normally. In these settings, a viability stain is not an optional addition. It is a filter for deciding what should count as biologically relevant data.
How Dead Cells Distort Imaging and Quantitative Results?
In fluorescence microscopy and related imaging workflows, non-viable cells often generate bright but misleading fluorescence due to non-specific dye entry, abnormal morphology, condensed nucleic acid staining, or uneven accumulation of probe. These effects can create false impressions of localization, false increases in average intensity, or apparent structures that are really artifacts of membrane breakdown. In image-based quantification, even a modest proportion of dead cells can skew mean fluorescence measurements or distort segmentation quality if they are not identified and handled appropriately. In population-based analyses, the same problem appears as broad background, unexpected tails in signal distribution, or unstable gating boundaries. Cells that should have been excluded may appear as strongly stained outliers or contribute misleading fluorescence spill into other channels. The result is not simply more noise. It is a structural change in how the data are interpreted.
Why Viability Assessment Should Be Planned Before Panel Design?
Viability stains should be chosen at the beginning of panel design, not added at the end as an afterthought. They occupy spectral space, influence sample handling, and may constrain whether the workflow remains fully live or transitions into fixation. For example, a live/dead dye that works well in imaging may not be suitable for post-fix analysis, while a fixable viability dye may introduce different handling requirements. Planning viability from the start improves channel allocation, helps prevent overlap, and reduces the chance that the viability control itself interferes with the rest of the experiment.
Comparing live/dead cell staining methods for your assay or imaging workflow?
We can help you evaluate viability dyes based on membrane integrity, esterase activity, signal clarity, and compatibility with your experimental conditions.
How Fluorescent Cell Viability Staining Works?
Fluorescent viability methods are built around differences between intact and damaged cells. Although the specific chemistries vary, most viability stains use one of three major principles: selective membrane exclusion, intracellular conversion in viable cells, or chemical labeling that becomes possible once membrane integrity is lost or once cells are processed for downstream fixed workflows. Understanding which principle a dye relies on is essential, because interpretation depends on the mechanism rather than on fluorescence color alone.
Membrane Integrity-Based Staining Logic
Many live/dead methods depend on whether the plasma membrane still acts as a selective barrier. In viable cells, impermeant dyes are excluded or enter only minimally. In damaged or dead cells, the membrane barrier fails, allowing these dyes to access intracellular components and generate a strong fluorescent signal. This is one of the most direct and widely used approaches because it creates an intuitive distinction between intact and non-intact cells. It is also the basis of many nucleic-acid-binding dead-cell stains used in imaging and suspension analysis. The advantage of this logic is simplicity, but it also has limitations. Cells in early or partial damage states may not behave as fully live or fully dead. Some may show partial permeability or inconsistent signal depending on incubation time and concentration. For that reason, membrane integrity stains should be optimized carefully and interpreted in the context of how the sample was treated before staining.
Intracellular Conversion and Esterase-Dependent Fluorescent Readouts
A second major strategy uses fluorogenic compounds that become fluorescent after intracellular conversion in viable cells, often through esterase activity. Under this model, living cells actively convert or retain the fluorescent form, while dead cells do not support the same transformation or retention behavior. This creates a signal that highlights viable cells directly rather than labeling dead cells. Such methods can be particularly useful when the experimental emphasis is on identifying viable cells in situ rather than excluding non-viable cells only. These methods can produce clear live-cell signals, but they are not simply indicators of membrane integrity. They reflect aspects of intracellular function and retention, which means they can be influenced by treatment conditions, incubation length, or metabolic changes that do not necessarily correspond to complete loss of viability. This is one reason they are often most powerful when combined with a complementary dead-cell indicator.
Amine-Reactive Fixable Viability Dye Strategies
In workflows that require fixation after initial viability assessment, amine-reactive viability dyes provide another approach. These reagents exploit the increased accessibility of intracellular amines in membrane-compromised cells. Because dead or heavily damaged cells allow more extensive labeling, they become distinguishable from live cells even after subsequent processing. This makes such dyes particularly useful when viability information must be preserved into downstream fixed analysis. However, their value depends on appropriate timing, controlled labeling conditions, and compatibility with the rest of the fluorescent panel. The central lesson across all three strategies is that the meaning of the signal depends on the biology the dye is designed to exploit. A green signal does not automatically mean "live," and a red signal does not automatically mean "dead." Interpretation must always begin with the staining mechanism.
Common Fluorescent Methods to Distinguish Live and Dead Cells
Most viability workflows use one of a small number of practical staining formats, each with its own strengths. Some favor speed and simplicity, some support direct imaging, and some fit better into complex multicolor panels. Choosing among them requires understanding how each method behaves in real experiments rather than only how it appears in a product description.
Impermeant Dyes for Dead-Cell Identification
One widely used format relies on membrane-impermeant dyes that become strongly fluorescent only in cells that have lost membrane integrity. These dyes are especially useful when the goal is to identify and exclude dead cells rapidly. They are often chosen for straightforward live/dead discrimination because the biological logic is clear and the signal tends to be easy to interpret when the workflow is well controlled. Some nucleic-acid-binding probes used in this space align naturally with categories such as DNA stains or nuclear fluorescent probes, depending on how they are positioned in the broader staining workflow.
Calcein-Based Live-Cell Viability Staining
Calcein-type strategies are common when researchers want to highlight viable cells directly. These methods generally rely on intracellular processing and retention in intact cells, making viable cells appear fluorescent while non-viable cells remain dim or negative. This can be useful in imaging-based workflows where identifying the living portion of the sample is visually informative. However, since the readout depends on more than membrane exclusion alone, these methods should not be treated as perfect substitutes for direct dead-cell stains in every context.
Dual-Color Live/Dead Approaches
Dual-color methods combine a viable-cell signal with a dead-cell signal, creating more explicit separation and often improving interpretability. This format is attractive in microscopy and quantitative imaging because it allows simultaneous positive marking of viable cells and non-viable cells within the same field. When carefully designed, dual-color strategies can also improve confidence in borderline cases where one signal alone would be ambiguous. The main tradeoff is that they consume more spectral space and therefore require more careful planning in multicolor experiments.
Fixable Viability Dyes for Downstream Processed Workflows
Fixable viability dyes are best suited for workflows where live/dead discrimination must be captured before fixation and then preserved into later analysis. This is particularly useful when the experiment includes additional fluorescent labels, antibody-based steps, or endpoint analysis after the viability decision has already been made. The strength of this method is workflow continuity. The challenge is that labeling conditions, compensation or overlap, and processing order must be carefully matched to the rest of the panel. In practice, researchers often choose between these methods not because one is universally superior, but because one aligns better with the experimental sequence. Short, live readouts often prioritize simple dead-cell exclusion. Imaging-based assays may benefit from dual-color logic. Processed panels may require fixable viability solutions. The decision should be functional, not fashionable.
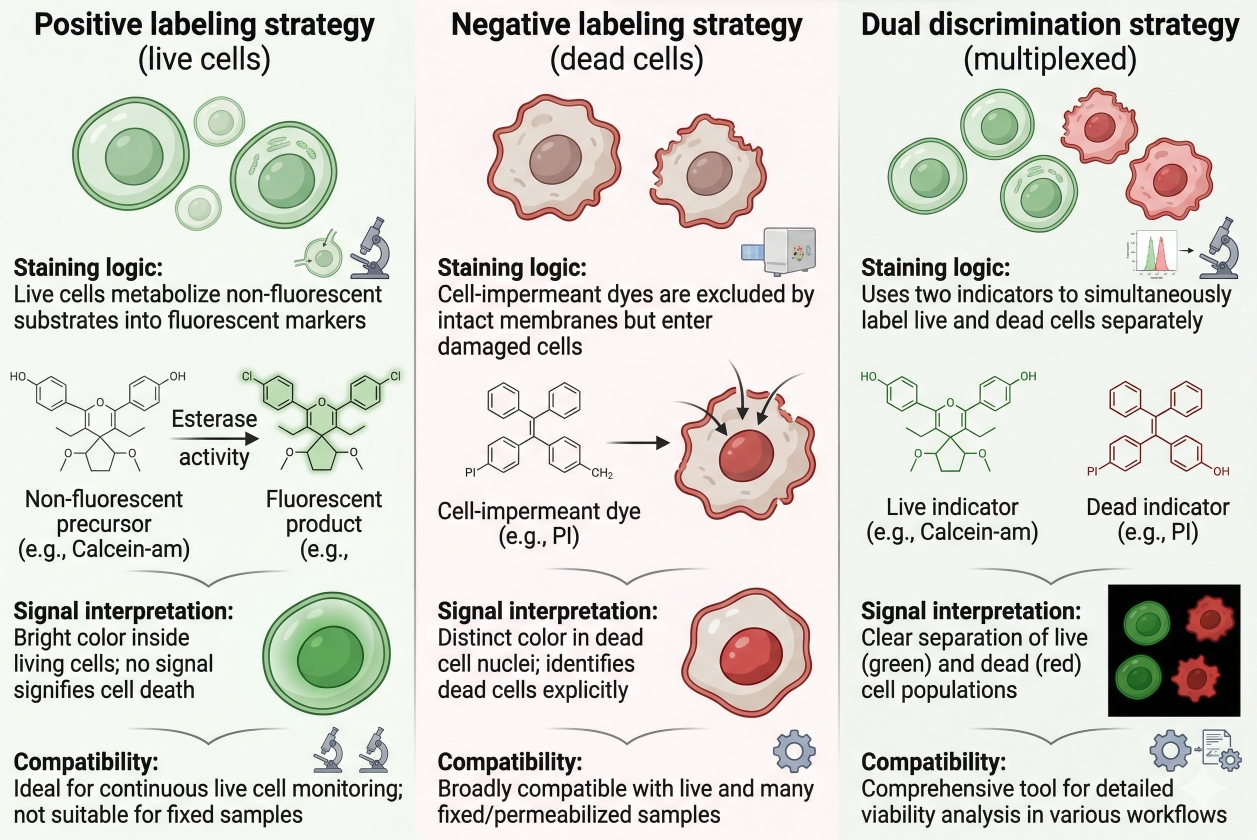 Fig. 3. Fluorescent viability methods differ in staining logic, signal interpretation, and compatibility with live versus processed workflows (BOC Sciences Authorized).
Fig. 3. Fluorescent viability methods differ in staining logic, signal interpretation, and compatibility with live versus processed workflows (BOC Sciences Authorized).
Applications of Cell Viability Staining
Cell viability staining is used across a wide range of fluorescence-based workflows because live/dead discrimination improves both analytical confidence and experimental efficiency. Although the underlying principles are shared, the practical role of viability staining changes depending on whether the experiment emphasizes imaging, population analysis, treatment response, or multicolor assay quality control.
Fluorescence Microscopy and Imaging-Based Viability Evaluation
In imaging workflows, viability staining helps determine whether fluorescent patterns arise from living cellular organization or from damaged-cell artifacts. This is especially important in time-sensitive live assays, high-content imaging, and experiments where cell shape or subcellular fluorescence distribution is part of the interpretation. When dead cells are not identified, they can appear unusually bright, abnormally condensed, or morphologically distorted in ways that confuse downstream image analysis. Viability staining therefore acts as both a biological control and an image quality control in fluorescence imaging workflows.
Flow Cytometry and Population-Level Live/Dead Discrimination
In flow-based assays, viability staining helps researchers exclude dead cells before interpreting marker-positive populations. This is critical because dead cells often bind reagents non-specifically and can distort fluorescence distributions across multiple channels. A well-chosen viability dye improves gating quality, makes population structure more stable, and reduces the risk of overestimating target-positive events. This is one reason viability control is widely treated as a foundational part of well-designed flow cytometry panels.
Drug Response, Cytotoxicity, and Treatment Comparison Studies
Viability staining is also highly relevant in treatment-response experiments where the central question is whether exposure changes the proportion of live and dead cells. In these assays, fluorescence-based viability methods support rapid comparison between untreated and treated groups, help define dose-related effects, and reveal whether observed biological responses reflect selective target modulation or simply widespread cell damage. This makes viability staining useful not only for endpoint discrimination but also for experimental context: it tells you whether the rest of the fluorescence data should be interpreted as biology or as the consequence of sample collapse.
Viability Control in Multicolor Staining and Sorting Workflows
In multicolor assays, viability staining protects the integrity of the panel. It prevents damaged cells from introducing false-positive fluorescence into channels assigned to other targets and improves interpretation of complex panels that include structural or functional labels. It also helps ensure that downstream isolation or sorting strategies focus on intact cells rather than debris-rich or dying populations. In this sense, viability staining supports the reliability of the entire panel rather than functioning as an isolated measurement.
Need help choosing viability stains for flow cytometry, microscopy, or cell-based assays?
We can help match fluorescent viability probes to your detection platform, sample type, and multicolor workflow for accurate live/dead discrimination.
How to Choose the Right Viability Dye for Your Experiment?
Choosing the right viability dye requires matching the staining mechanism to the workflow rather than starting from fluorescence color alone. Researchers should ask a sequence of practical questions: Will the assay remain live or proceed into fixation? Is the readout based on microscopy or suspension analysis? Does the panel already occupy key channels? Is the main need rapid dead-cell exclusion, positive identification of viable cells, or preservation of viability information into later steps? These questions usually narrow the field more effectively than generic brightness comparisons.
- Live-Cell Versus Fixed-Cell Workflow Requirements: The first major decision is whether viability will be measured only in live cells or whether the information must survive a later fixed workflow. If the sample remains live through acquisition, simple live/dead strategies may be sufficient. If viability labeling must be preserved into later processed analysis, a fixable approach is often more appropriate. This decision should be made before the rest of the panel is finalized because it affects processing order and channel planning.
- Microscopy Versus Flow Cytometry Platform Considerations: Platform matters. Imaging workflows often benefit from stains that create visually clear discrimination and fit naturally into segmentation or field-level interpretation. Flow-based workflows often prioritize rapid, robust exclusion of dead cells with minimal ambiguity and easy gating behavior. A dye that looks excellent in a microscope may not be the most efficient choice for high-throughput population analysis, just as a stain chosen for rapid suspension gating may not provide the most informative spatial context in microscopy.
- Spectral Compatibility with Other Fluorescent Channels: Viability dyes consume spectral space and therefore compete with the rest of the panel. This is especially important when the assay already includes multiple fluorescent probes, structural stains, or fluorophore families such as Cyanine, Rhodamine, or Coumarin. The right dye is one that fits the biological question and the optical layout at the same time. Channel conflict is one of the easiest ways to undermine an otherwise strong viability strategy.
- Signal Stability, Convenience, and Interpretation Needs: Practical factors matter as well. Some experiments need a fast stain with minimal preparation. Others need strong signal separation for quantitative analysis. Some samples are fragile and require low-disturbance handling. Others can tolerate longer processing if it yields more reliable classification. The best viability dye is therefore not simply the brightest or most commonly used one. It is the one that produces interpretable results under the actual experimental conditions.
Common Pitfalls in Cell Viability Staining and How to Avoid Them
Viability staining is conceptually simple, but in practice it is highly sensitive to timing, concentration, sample condition, and panel design. Many misleading results come not from the dye itself, but from mismatches between staining mechanism and workflow. Avoiding these errors is essential because viability data are often used to decide which cells should be included in the rest of the analysis.
- Misclassifying Stressed Cells as Dead Cells: One common problem is treating every abnormal viability signal as proof of complete cell death. In reality, stressed or partially compromised cells may display intermediate behavior. They can show altered uptake, partial permeability, or reduced conversion of viability reagents without behaving like fully dead cells. This is why borderline populations should be interpreted carefully, especially after aggressive treatment or harsh sample preparation.
- High Background and Nonspecific Fluorescence: Background fluorescence can arise from excessive dye concentration, insufficient washing, abnormal sample debris, or high proportions of damaged cells. In these situations, the viability dye may appear less selective than expected and can obscure the rest of the panel. Optimization should focus on concentration, incubation control, and overall sample quality rather than assuming that more dye will create better separation.
- Overlap with Other Dyes in Multicolor Experiments: A viability stain that works well on its own may become problematic in a multicolor experiment if it occupies a critical channel or creates unnecessary overlap. This can lead to false interpretation of target expression, poor compensation, or loss of useful marker resolution. The safest approach is to allocate the viability channel early and evaluate it against the whole panel rather than selecting it at the end.
- Fixation Mismatch and Loss of Staining Relevance: Another frequent mistake is using a live-only viability stain in a workflow that later includes fixation and expecting the signal meaning to remain unchanged. If the stain is not designed for that transition, the biological interpretation may no longer be reliable after processing. A viability signal should be trusted only within the workflow context for which it was designed.
How BOC Sciences Supports Cell Viability Staining Projects?
BOC Sciences supports cell viability staining projects through integrated capabilities in viability dye supply, custom probe development, assay optimization, and quality-focused supply support. Rather than treating viability assessment as a simple live/dead labeling step, we help researchers align probe mechanism, fluorescence properties, detection platform, and experimental workflow with the actual purpose of the study. This is especially important in cell viability assays, where membrane integrity, esterase activity, metabolic state, staining sequence, and multicolor compatibility can all influence whether the final readout is accurate and interpretable. Our support is therefore structured to cover both routine viability evaluation needs and more specialized fluorescence-based cell health analysis applications.
Cell Viability Dyes and Detection Reagent Supply
- Access to a range of fluorescent viability and cell death staining reagents relevant to live/dead discrimination, including products comparable to or aligned with commonly used assay logic such as Calcein AM, PI, and 7-AAD-type workflows.
- Support for selecting dyes associated with membrane integrity assessment, esterase activity evaluation, and metabolic activity-related fluorescence readouts according to experimental purpose.
- Availability of viability reagents across multiple fluorescence regions to support fluorescence microscopy, flow cytometry, and high-content imaging workflows.
- Product options adapted to different cell systems, including adherent cells and suspension cells, helping improve assay fit under different handling and detection conditions.
Customized Viability Probe Development
- Custom development of cell viability probes according to specific research needs, including mechanism-oriented probes linked to ROS response, mitochondrial function, or other cell health-related fluorescence logic.
- Structural optimization support to improve probe sensitivity, selectivity, and photostability for more demanding viability and cytotoxicity analysis workflows.
- Design support for multiparameter viability probes, including strategies that combine viability-related readout with apoptosis-associated or other functional fluorescence markers.
- One-stop custom services covering molecular design, synthesis, and performance validation for tailored viability detection probes.
Assay Design and Technical Support
- Practical guidance for optimizing cell viability assays, including dye concentration, incubation duration, staining order, and detection conditions based on the actual workflow.
- Assistance with selecting more suitable assay formats, including Live/Dead staining routes, metabolism-related methods, and fluorescence-based viability strategies matched to the experimental objective.
- Support for multichannel detection design and combined staining strategies when viability readouts need to be integrated with additional cellular or organelle markers.
- Troubleshooting help for common assay challenges, including false-positive or false-negative signals, high background, staining-related toxicity, and reduced interpretability under real sample conditions.
Quality Control and Stable Supply System
- Viability staining reagents supported by strict quality testing using analytical methods such as HPLC and LC-MS where applicable.
- Availability of complete quality documentation, including COA and batch analysis reports, to support evaluation, internal review, and project use.
- Flexible supply support suitable for research-stage use as well as broader pilot-scale purchasing needs across different project phases.
- Attention to batch consistency and long-term supply continuity to help support ongoing cell viability studies and repeated assay workflows over time.
Do You Need A Consultation?
BOC Sciences integrates cutting-edge fluorescence technologies to accelerate your research, driving next-generation solutions for drug discovery and diagnostics.
Transform Your Studies with Cutting-Edge Fluorescent Products
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0087 | RH 795 | 172807-13-5 | Bulk Inquiry |
| A16-0156 | Speed DiI | 278173-35-6 | Bulk Inquiry |
| A16-0084 | Speed DiO | 164472-75-7 | Bulk Inquiry |
| A16-0147 | APTAB | 86727-71-1 | Bulk Inquiry |
| A16-0222 | YOYO 1 | 143413-85-8 | Bulk Inquiry |
| A16-0223 | YO-PRO 3 | 157199-62-7 | Bulk Inquiry |
| A16-0220 | TOTO 3 | 166196-17-4 | Bulk Inquiry |
| A16-0111 | BF 594 Phalloidin | 330626-83-0 | Bulk Inquiry |
| A16-0175 | Lucifer Yellow CH Dipotassium Salt | 71206-95-6 | Bulk Inquiry |
High-Performance Fluorescent Tools for Your Research
- Cell membrane Fluorescent Probes Surface-labeling dyes for membrane dynamics studies.
- Mitochondrial Fluorescent Probes Targeted dyes for mitochondrial structure and function.
- Nerve Terminal Probes Fluorescent tracers for synaptic activity analysis.
- Golgi Fluorescent Probes Targeted probes for Golgi apparatus visualization.
- Lipid Fluorescent Probes Dyes for lipid droplets and membranes.
- Cytoskeleton Fluorescent Probes Probes for actin and microtubule visualization.
- Lysosomal Fluorescent Probes Acidic organelle markers for lysosome tracking.
- Metal Fluorescent Probes Selective sensors for intracellular metal ions.
Explore More Topics
Online Inquiry


