Endoplasmic Reticulum Staining: Principles, Dye Selection, and Workflow Design
Cell staining of the endoplasmic reticulum is widely used in fluorescence-based cell analysis because ER organization is tightly linked to intracellular architecture, membrane dynamics, organelle interaction, and image interpretation quality. In many experiments, researchers are not simply trying to confirm that the endoplasmic reticulum is present. They want to understand whether the ER forms a fine reticular network, whether the perinuclear region appears dense or expanded, whether sheet-like and tubular features remain distinguishable, whether a treatment alters ER distribution relative to the nucleus, and whether the ER channel can be combined reliably with other fluorescence readouts. That is why endoplasmic reticulum staining is more demanding than routine nuclear labeling or simple membrane counterstaining. A useful ER staining page therefore needs to do more than list dyes. It should explain what ER fluorescence is actually showing, how probe mechanism influences interpretation, why live-cell and fixed-cell workflows should not be treated as interchangeable, and how to build a staining workflow that supports a biologically credible conclusion rather than a visually attractive but ambiguous image.
What Is Endoplasmic Reticulum Staining?
Endoplasmic reticulum staining refers to the use of fluorescent dyes, organelle-selective small molecules, or fluorescent probes to visualize the endoplasmic reticulum within cells so that ER position, intracellular distribution, reticular organization, perinuclear enrichment, and spatial relationships with other structures can be assessed under fluorescence imaging. In practical research workflows, ER staining is rarely used as a decorative organelle label. It is introduced because the ER is one of the most structurally informative membrane systems in the cell. The network extends from the nuclear envelope into a broad peripheral meshwork, and its appearance can strongly influence how researchers interpret intracellular organization, protein localization, trafficking-related readouts, and multicolor microscopy data. Depending on endoplasmic reticulum probes design, the fluorescent signal may primarily serve as a live-cell localization marker, may be only partially preserved after fixation, or may be better replaced by antibody-based ER labeling in fixed preparations. For that reason, ER staining should be treated as an interpretive imaging workflow rather than a simple staining step.
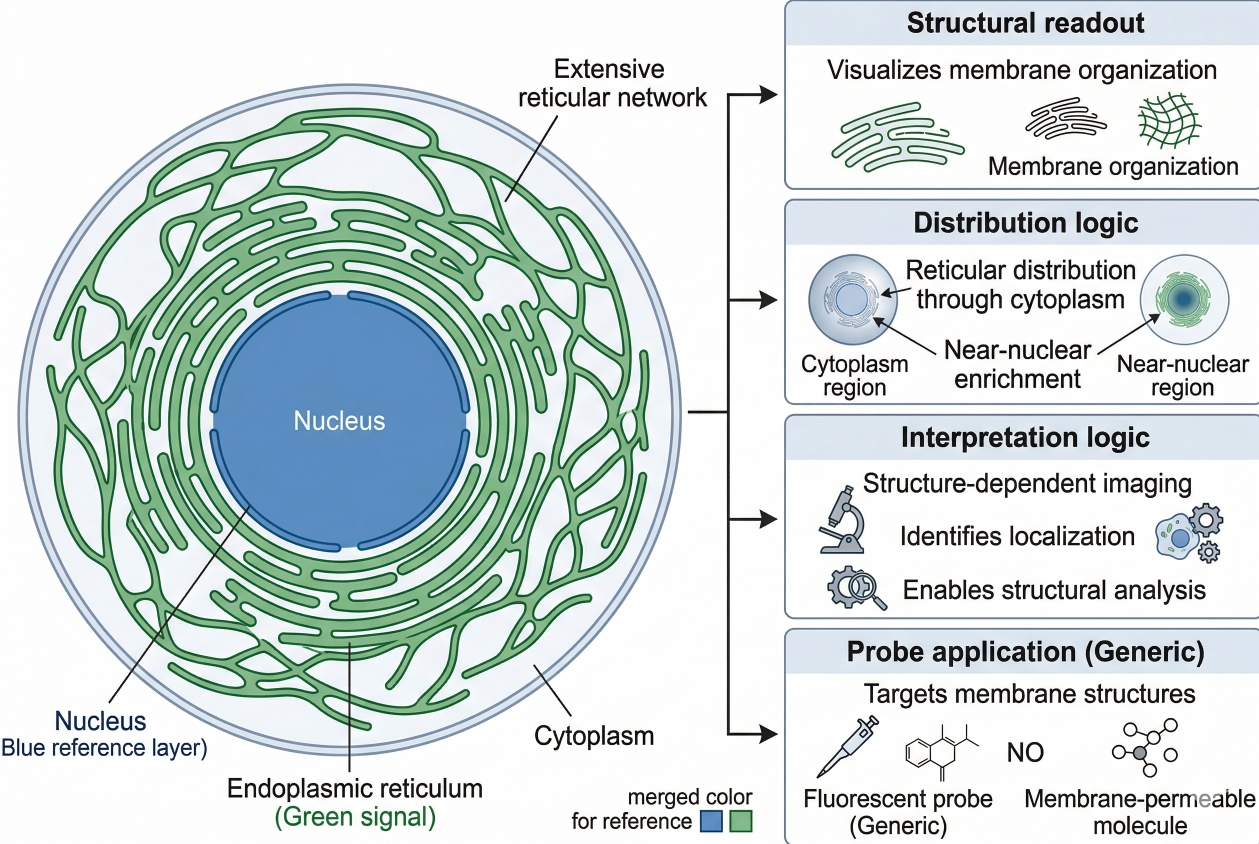 Fig. 1. ER staining reveals reticular intracellular membrane organization (BOC Sciences Authorized).
Fig. 1. ER staining reveals reticular intracellular membrane organization (BOC Sciences Authorized).
In fluorescence experiments, the ER channel is often expected to answer several questions at once. Researchers may want to determine whether a protein of interest shows ER-associated localization, whether the ER remains as a fine network or collapses into a denser juxtanuclear pattern, whether the selected probe can coexist with nuclear and mitochondrial channels, and whether the staining remains meaningful after post-staining handling. This practical complexity is why endoplasmic reticulum staining should not be reduced to "which dye is brightest." A bright signal is not automatically a selective signal, and a visible ER-like pattern is not automatically a workflow-compatible readout. Some stains are most useful in live-cell imaging windows, some are selected because they fit multicolor designs, and others are better interpreted as conventional lipophilic membrane probes that require caution. The right choice depends on what the experiment needs to show and what the chosen probe mechanism can realistically support.
What ER staining is designed to reveal?
ER staining is designed to reveal the position and morphology of one of the cell's most extensive membrane systems. In imaging terms, that usually means showing a reticular pattern that extends through the cytoplasm, often with stronger organization around the nucleus and continuity with the outer nuclear membrane. In morphology-oriented experiments, ER staining can help users assess whether the network appears continuous or fragmented, whether the peripheral meshwork is preserved, whether a perinuclear region dominates the signal, and whether ER distribution remains spatially coordinated with the rest of the cell. In colocalization-oriented work, the ER channel is used as a contextual reference for proteins, vesicles, or other organelles. In panel-based imaging, the ER signal can add a membrane-system layer that complements nuclear morphology, mitochondrial distribution, or acidic compartment labeling. The point is not merely to "see ER," but to create a trustworthy spatial framework for interpreting where other fluorescent events are happening.
Why ER staining is more interpretive than it looks?
ER staining looks straightforward because many successful images display an elegant green, red, or blue-white reticular network. However, the interpretive challenge begins once researchers assume that every apparent ER pattern has the same meaning. Some live-cell ER probes are selective because of their affinity for ER-associated targets, whereas other conventional membrane dyes label intracellular membranes more broadly and depend on morphology for interpretation. Some stains are useful immediately after loading into live cells but are not appropriate for staining cells after fixation. Others may remain partially visible after formaldehyde treatment but at reduced intensity, which means persistence of signal should not be confused with preservation of original staining logic. As a result, ER staining is not only about image appearance. It is about matching probe mechanism, sample state, handling sequence, and imaging goal before drawing conclusions from the pattern.
When researchers need ER staining instead of general membrane labeling?
Researchers need ER staining rather than general membrane labeling when the experimental question requires a specifically interpretable ER readout. General lipophilic membrane dyes may distribute across multiple intracellular membranes, and in some cases the ER can only be inferred from characteristic morphology. That can be acceptable for some exploratory imaging tasks, but it is much less suitable when the ER channel needs to serve as a structured reference in multicolor analysis, co-localization imaging, or treatment comparison. In those cases, users usually benefit from ER-selective live-cell probes such as ER-Tracker Blue-White DPX, LumiTracker ER Green, or LumiTracker ER Red, or from fixed-cell ER labeling routes based on ER-associated markers. The more important the ER channel is for interpretation, the less appropriate it is to rely on broad membrane staining alone.
Choosing between live-cell ER dyes, fixation-compatible workflows, and multicolor panel options?
We can help match ER probe selectivity, spectral placement, and staining workflow to your imaging goals, sample handling strategy, and co-localization design.
Why Endoplasmic Reticulum Staining Matters in Cell Imaging?
Endoplasmic reticulum staining matters because the ER is not a minor intracellular background structure. It is a large, dynamic membrane system involved in protein processing, lipid-related organization, and intracellular spatial coordination. In fluorescence imaging, this makes the ER one of the most informative structural channels available. A well-resolved ER stain can help users understand whether a treatment alters cell-wide membrane organization, whether a protein of interest follows an ER-like distribution, whether a multicolor assay is spatially coherent, and whether organelle relationships remain interpretable across samples. The value of ER staining increases when researchers define the intended readout in advance and choose a workflow that preserves that meaning from loading to image acquisition.
- ER as a structural readout in fluorescence microscopy: In fluorescence microscopy, the ER provides a rich structural pattern that is more informative than a simple diffuse cytoplasmic label. Because the ER extends from the nuclear envelope into a peripheral membrane network, the staining pattern offers a practical way to map intracellular organization. This is particularly useful when the imaging question is about where signals occur in relation to a membrane framework instead of merely whether fluorescence is present.
- Why ER staining is useful in co-localization and organelle context analysis: Many users search for ER staining because they want to know whether a protein, vesicle population, or fluorescent construct appears ER-associated or ER-adjacent. In these workflows, the ER channel serves as a contextual reference rather than a standalone endpoint. The image becomes more meaningful because the location of the target can be interpreted relative to a defined intracellular membrane system instead of against a visually empty cytoplasm.
- The value of ER staining in multicolor workflow design: ER staining is commonly integrated into broader cell imaging panels together with nuclear fluorescent probes, mitochondrial fluorescent probes, lysosomal fluorescent probes, membrane fluorescent probes, or antibody-based channels. In these workflows, the ER channel must do more than look good by itself. It has to occupy a sensible spectral position, retain adequate contrast under the actual acquisition settings, and remain compatible with the rest of the panel without overwhelming weaker channels.
- Why ER staining supports more reliable image interpretation: A defined ER stain can reduce ambiguity when users are comparing untreated and treated samples, validating intracellular localization, or building image-analysis workflows. When the ER pattern is clear and the mechanism is understood, downstream interpretation is less dependent on subjective visual impression and more grounded in consistent staining logic.
Core Principles Behind Endoplasmic Reticulum Staining
The most important principle in ER staining is that not all ER-related dyes report the same thing in the same way. Some probes are designed for selective live-cell ER labeling, some are better understood as broader lipophilic membrane dyes that can include ER among other internal membranes, and some workflows are fundamentally better served by fixed-cell antibody labeling rather than by attempting to extend live-cell dyes beyond their intended use. A strong ER page therefore needs to explain not only what stains are available, but how signal selectivity is achieved, when the signal remains meaningful, and where the workflow boundaries are.
- Live-cell ER-selective dyes and how they work: A major category of ER stains is the live-cell, cell-permeant, ER-selective probe class represented by ER-Tracker-type reagents and products such as LumiTracker ER Green and LumiTracker ER Red. These probes are useful because they produce a relatively clean ER pattern in living cells and fit naturally into organelle imaging workflows that prioritize direct visualization over complex staining sequences. In practice, they are selected when users need a live-cell ER channel that can be added with a simple incubation and then imaged promptly. Their value lies in the combination of accessibility, selectivity, and panel compatibility. At the same time, they should not be treated as universal ER solutions for every sample state and every downstream handling step.
- Why fixation compatibility is a major decision point: One of the most common sources of error in ER staining is assuming that a probe that performs well in live cells will remain equally meaningful after fixation. This is not a safe assumption. Some ER probes show partial pattern retention after formaldehyde treatment, while others lose signal quality or are not suitable for use after fixation at all. The practical consequence is important: if the experiment truly requires a fixed-cell endpoint, users should decide that early and choose a workflow consistent with that endpoint rather than performing live staining first and hoping the pattern survives later processing. For fixed-cell ER analysis, antibody-based labeling of ER-associated markers is often the more reliable route because it is designed around fixation and permeabilization rather than working against them.
- Specificity limits: ER signal vs non-ER labeling: ER selectivity is never a reason to stop thinking critically about staining behavior. Probe-target interactions, cell-type-specific expression patterns, concentration, incubation timing, and wash conditions can all influence how clean the final image looks. Some ER-selective live-cell stains can show non-ideal localization in specialized cell types or under non-optimized handling. Conventional membrane dyes add another layer of caution, because they may label intracellular membranes more broadly and require users to identify ER by morphology rather than by high intrinsic selectivity. That means the question is not simply "Does this dye produce a reticular pattern?" but "Does this pattern remain consistent with the intended ER interpretation under the actual conditions being used?"
- Why ER staining should not be reduced to signal intensity alone: ER staining is usually more powerful as a structural and positional readout than as a simple intensity measurement. A stronger fluorescent signal does not automatically mean more ER, better ER integrity, or better biological relevance. It may reflect higher loading, slower washout, greater membrane partition, different optical settings, or spectral imbalance relative to other channels. Conversely, a weaker signal may still be more interpretable if it preserves fine reticular detail with lower background. The practical goal should therefore be signal quality that supports correct spatial interpretation, not maximum brightness at any cost.
Common Types of ER Dyes and How to Choose Between Them?
ER dyes are best classified by staining logic and workflow role rather than by chemical name alone. This is how researchers actually make decisions in real imaging projects. They are not usually asking whether a single product is universally "best." They are asking whether the stain fits live-cell imaging, whether it can coexist with GFP, whether a UV-excited channel would help panel design, whether a conventional membrane dye is acceptable for morphology-only work, and whether the assay should move instead toward fixed-cell ER labeling. The following categories are therefore more useful than a flat product list.
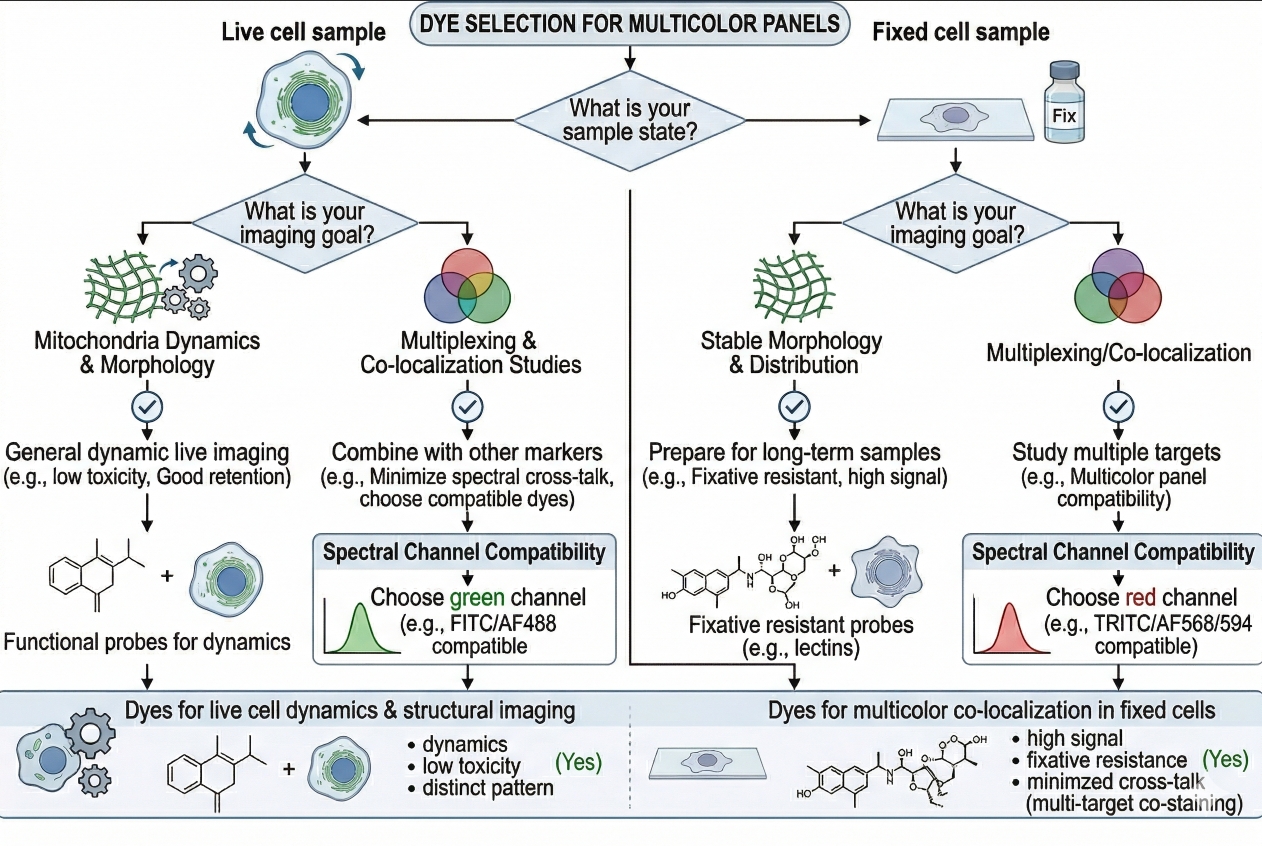 Fig. 2. Choose ER dyes by workflow and imaging goals (BOC Sciences Authorized).
Fig. 2. Choose ER dyes by workflow and imaging goals (BOC Sciences Authorized).
ER-selective live-cell probes for direct ER visualization
ER-selective live-cell probes are the most practical starting point when the main goal is to visualize the ER in living cells with a relatively straightforward workflow. LumiTracker ER Green and LumiTracker ER Red fit this category well because they are intended for selective ER labeling in live-cell imaging and can be used to generate a clear reticular pattern when staining conditions are optimized. A probe in this class is generally the right choice when users need a structurally interpretable ER channel without relying on transfection or antibody staining. It is especially useful for routine live imaging, short-term comparison across conditions, and panel-based organelle visualization where the ER channel has to be readable and operationally simple.
Blue-white ER probes for UV or DAPI-region channel allocation
ER-Tracker Blue-White DPX is especially useful when the visible channels are already crowded and the assay would benefit from placing ER in a UV-excited or DAPI-compatible region. This can be strategically valuable in multicolor imaging because it may free green and red channels for other higher-priority markers. Blue-white ER probes are not automatically better than green or red options, but they are highly relevant when the panel architecture is the limiting factor. Researchers should still remember that even when a stain remains partially visible after fixation-related handling, substantial signal reduction may occur, so the channel assignment decision should be made together with the sample-state decision rather than independently.
Conventional lipophilic dyes that can include ER labeling
3,3'-Dihexyloxacarbocyanine iodide, commonly discussed as DiOC6(3)-type staining, represents a more classical membrane-labeling route that can be used to visualize intracellular membranes including the ER under some conditions. However, it should be approached with more caution than ER-selective probes. At low concentrations, this class is often associated more strongly with mitochondrial staining, whereas at higher concentrations it may stain other internal membranes such as the ER. That means users should not interpret it as functionally equivalent to an ER-selective tracker dye. It may still be useful in exploratory membrane imaging, structure-oriented experiments, or comparative morphology work where the ER can be recognized by its characteristic reticular appearance, but it is less ideal when the ER channel needs maximum specificity.
Red-shifted ER options for GFP-compatible imaging
When a workflow already includes GFP, FITC-like reporters, or green-emitting organelle probes, red-shifted ER staining becomes especially valuable. LumiTracker ER Red is a natural candidate in this context because it helps place the ER signal into a more panel-friendly spectral region. ReZolve-ER™ may also be considered when users are comparing ER-focused options and want flexibility in optical planning. The central decision is not color alone, but whether the red ER channel will improve panel separation, protect the green channel for the most critical marker, and reduce interpretation problems caused by spectral crowding.
Alternative probes and complementary reagents in broader imaging workflows
Not every useful product in an ER imaging project is itself an ER stain. For example, C-Laurdan can support membrane-environment studies, Toto-1 and Oxazole yellow can serve nucleic-acid-related or counterstaining roles in selected workflows, and LysoTracker Red DND-99 may be useful when the project includes ER-lysosome spatial context rather than ER alone. IraZolve-L1™ may enter broader intracellular imaging designs when users are comparing alternative live-cell fluorescent tools. These reagents are not substitutes for ER-selective staining, but they can become relevant when ER is one layer within a larger multicolor experiment.
Need an ER staining strategy for live-cell imaging, fixed-cell analysis, or multicolor organelle studies?
We can help align probe choice, staining conditions, and channel planning with your experimental timeline and fluorescence workflow.
Practical Workflow for Endoplasmic Reticulum Staining in Cells
A reliable ER staining workflow is not only about adding dye and collecting images. It is about preserving an interpretable relationship between the cells, the stain, the imaging medium, and the acquisition timing. Many staining failures happen because the workflow was built around convenience rather than signal meaning. The most robust approach is to control cell condition, define whether the imaging is live or endpoint-based, optimize concentration and timing in small pilot runs, and standardize acquisition settings before comparisons are made. Researchers who want a more general framework for assay consistency can also review broader cell staining protocol principles before locking in an ER-specific workflow.
1. Preparing cells for ER staining
Start with healthy cells that are appropriately plated for the imaging modality. Overconfluent cultures, uneven attachment, excessive debris, or stressed cells can make ER morphology harder to interpret because the reticular pattern becomes less distinct and background rises relative to useful structural detail. The imaging vessel should support the optical resolution required for the project, and the working medium should be chosen with fluorescence performance in mind. In many live-cell workflows, users benefit from prewarmed buffer or imaging-compatible medium and from minimizing unnecessary handling immediately before staining. The practical aim is to preserve a stable cell state so that the ER channel reflects intracellular organization rather than last-minute workflow disturbance.
2. Incubation, wash, and imaging timing considerations
For live-cell ER-selective probes, concentration and incubation time should be treated as optimization variables rather than fixed truths. A sensible starting window for products such as LumiTracker ER Green and LumiTracker ER Red is often in the low-nanomolar to low-micromolar range, with short incubations at physiological temperature followed by gentle washing and prompt imaging. In practical terms, too little loading may yield incomplete network visualization, while excessive loading can increase background or non-ideal intracellular fluorescence. Timing matters as much as concentration. A signal that looks excellent immediately after staining may no longer have the same contrast or interpretive quality after unnecessary delay. This is why pilot experiments should test not just whether the cells become fluorescent, but whether the ER pattern remains clean across the actual acquisition window planned for the study.
3. Live imaging vs post-staining fixation decisions
Users should decide early whether the ER staining result must be collected in live cells or whether a fixed endpoint is truly required. This decision should not be postponed until after staining, because live-cell ER probes and fixed-cell ER workflows are not simply interchangeable versions of the same assay. If the study depends on live-cell ER organization, then the workflow should be built around immediate imaging and consistent handling. If the project requires fixation, permeabilization, and additional antibody channels, then antibody-based ER labeling or other fixed-compatible approaches may be more appropriate than stretching a live-cell tracker dye beyond its intended boundaries. When users try to combine both goals without planning for them, the resulting signal is often visible but interpretively unstable.
4. Controls needed for reliable ER interpretation
ER staining becomes far more reliable when the workflow includes the right controls. At minimum, users should include unstained cells to identify background and autofluorescence, single-color controls to assess spectral bleed-through, and matched comparison samples imaged under identical settings. If the ER channel will be used for co-localization claims, a reference condition or positive localization control is highly valuable. If a conventional lipophilic dye is being evaluated, direct comparison with an ER-selective probe can clarify whether the apparent reticular pattern is truly usable for the intended purpose. In multicolor imaging, controls are essential because what looks like an altered ER pattern may actually reflect spectral conflict, poor filter separation, or display scaling rather than a real change in intracellular organization.
Applications of Endoplasmic Reticulum Staining in Research Workflows
Endoplasmic reticulum staining is used across a wide range of research workflows because the ER provides an unusually rich structural and contextual readout. Its broad intracellular reach makes it valuable in microscopy, localization analysis, panel-based organelle imaging, and workflow development. In many projects, the ER channel is not interpreted alone. Instead, it becomes one layer within a broader decision framework that may also include nuclear position, mitochondrial pattern, acidic compartment distribution, membrane order, or fluorescently labeled proteins. The more integrated the assay becomes, the more important it is to select an ER staining strategy that supports that broader architecture.
ER staining in fluorescence microscopy and image-based morphology analysis
In fluorescence microscopy, ER staining is often used to generate an interpretable map of intracellular membrane organization. Researchers may compare whether the ER remains broadly reticular, whether the perinuclear zone becomes more dominant, or whether the peripheral network remains evident at the cell edge. These applications favor stains that preserve structural clarity rather than merely producing high total fluorescence. LumiTracker ER Green, LumiTracker ER Red, and ER-Tracker Blue-White DPX are relevant here because each can place the ER into a different spectral region while still supporting organelle-level visualization.
ER staining in protein localization and co-localization workflows
A common use of ER staining is to determine whether a protein or fluorescent construct shows an ER-associated pattern. In this setting, the ER channel functions as a localization reference. The stain does not prove biochemical association by itself, but it provides essential spatial context. Red-shifted ER options are often especially useful here because many localization studies already rely on GFP or FITC-like reporters. When the green channel is reserved for the target, a red ER stain can reduce channel competition and make the image easier to interpret.
ER staining in multicolor organelle interaction assays
ER staining is highly valuable in multicolor organelle interaction assays because the ER often needs to be interpreted alongside mitochondria, nuclei, lysosomes, or membrane-environment probes. For example, an assay may combine ER visualization with a nuclear counterstain, a mitochondrial channel, and LysoTracker Red DND-99 or C-Laurdan depending on the imaging goal. In these workflows, the ER channel must be selected with panel design in mind. A probe that looks impressive alone may still be a poor choice if it occupies the same spectral space as the assay's most important marker or generates a level of brightness that distorts image balance.
ER staining in workflow development and comparative treatment analysis
Comparative imaging workflows often rely on ER staining as one structural benchmark for judging whether cell organization changes across conditions. In these contexts, the most useful ER stain is not necessarily the most intense one. It is the one that remains interpretable, reproducible, and compatible with the rest of the assay. Live-cell ER probes are often favored for short comparative imaging runs, while fixed-cell ER labeling may be preferred when longer processing steps or antibody panels are required. The important point is that ER staining can support treatment comparison only when the staining logic remains stable across all groups being analyzed.
Common Problems in ER Staining and How to Avoid?
ER staining can fail in obvious ways such as weak signal, high background, or loss of network detail, but the more serious problem is often interpretive. A pattern can look convincingly "ER-like" and still be poorly matched to the actual question being asked. Troubleshooting therefore needs to address both image quality and biological meaning. The best troubleshooting approach is to separate issues of cell state, dye behavior, optical setup, and workflow logic rather than changing everything at once.
- Weak or incomplete ER network signal: Weak signal may arise from insufficient loading, short incubation, low cell health, or imaging settings that are too conservative for the selected fluorophore. Before concluding that the stain is unsuitable, users should test concentration, incubation time, wash intensity, and acquisition parameters in a small matrix. In many cases, the problem is not that the ER is unstained, but that the network detail is being under-sampled or underexposed.
- High background or diffuse cytoplasmic fluorescence: Background can result from excessive dye concentration, incomplete washing, overexposure, or the use of a conventional membrane dye in a workflow that really requires an ER-selective probe. Diffuse fluorescence is especially problematic because it reduces the visual distinction between a genuine ER network and nonspecific intracellular labeling. When this occurs, users should first reduce loading burden and then reassess whether the chosen probe class is appropriate for the desired level of specificity.
- Signal loss after fixation or post-staining handling: This is one of the most common ER staining failures. Even when some pattern is retained after formaldehyde treatment, intensity may drop enough to compromise interpretation, and some live-cell ER dyes are not appropriate for staining cells after fixation. The most reliable solution is to choose a fixed-compatible ER strategy at the start rather than retrofitting a live-cell staining route into a fixed-cell assay.
- Confusing ER with Golgi or other internal membranes: Broad membrane dyes can stain multiple intracellular membrane systems, which means the ER must often be recognized from morphology. If the assay depends on strong organelle specificity, users should not rely on morphological inference alone. Comparing a conventional membrane stain against an ER-selective tracker in the same cell type can help determine whether the broader stain is acceptable for that use case.
- Multicolor spectral conflict: In panel-based imaging, the apparent ER signal may be distorted by bleed-through, filter mismatch, or dominance of a brighter neighboring channel. This is especially likely when green and red channels are both heavily loaded or when display settings are adjusted unevenly between samples. Spectral planning should therefore be considered part of ER troubleshooting, not merely an imaging afterthought.
- Probe behavior affecting interpretation: Some ER-selective live-cell probes rely on molecular interactions that make them very useful, but those same properties also mean their behavior is not completely independent of cell type or workflow context. When users need highly conservative interpretation, it is wise to validate the stain in the exact cell system of interest before scaling the assay.
Best Practices for Building Reliable Endoplasmic Reticulum Staining Workflows
The most reliable ER staining workflows are built by defining the imaging objective first, matching the stain to that objective, and validating the workflow before large-scale data collection begins. This approach reduces avoidable troubleshooting and makes the ER channel far more informative. Endoplasmic reticulum staining becomes most useful when it is planned as a logic-driven measurement rather than an attractive add-on to a microscopy experiment.
- Start from the imaging question before choosing the dye: Decide whether the main purpose is ER morphology, colocalization context, multicolor panel balance, or fixed-cell endpoint analysis. Once this is defined, dye selection becomes much more rational.
- Validate the workflow in the actual cell system: Pilot tests should be run in the same cell type, plating density, medium condition, and imaging setup intended for the final experiment. This is particularly important for live-cell ER probes because signal behavior can vary with workflow details.
- Use structural interpretability as the primary criterion: Prefer a stain that preserves fine reticular detail with manageable background over one that simply produces the brightest field. A clean ER pattern is usually more valuable than maximal fluorescence.
- Plan spectral allocation early: Review broader how to choose the right fluorescent dye for cell staining principles before finalizing the ER channel. This is especially important when GFP, red organelle probes, or multiple antibody fluorophores are already part of the panel.
- Do not force live-cell stains into fixed-cell logic: If the project truly depends on post-fixation imaging, choose a workflow designed for that purpose rather than assuming a live-cell ER stain will remain equivalent after processing.
- Document the full staining logic for reproducibility: Record probe class, concentration range, incubation duration, wash conditions, imaging medium, delay before acquisition, exposure settings, and interpretation criteria together. This turns ER staining from a one-off success into a transferable workflow.
How BOC Sciences Supports Endoplasmic Reticulum Staining?
BOC Sciences supports endoplasmic reticulum staining projects through a combination of fluorescent product supply, custom probe development, staining workflow optimization, and technical service support. Rather than treating ER staining as a simple reagent selection task, we help researchers align probe choice, staining conditions, and imaging objectives with the actual needs of their cell system and fluorescence workflow. This is especially important in ER staining, where localization clarity, spectral compatibility, probe selectivity, and experimental handling conditions all affect whether the final image is truly useful. Our support is therefore designed to cover both standard ER staining needs and more specialized development-oriented applications.
Fluorescent Dye and Probe Supply for ER Staining
- Access to a range of validated endoplasmic reticulum-targeting fluorescent dyes and probes, including ER-selective staining options suitable for routine organelle imaging workflows.
- Support for probe selection across different spectral regions, from visible-range fluorescence to longer-wavelength imaging needs, helping researchers build more flexible multicolor staining panels.
- Availability of fluorescent probes with strong selectivity and practical photostability for ER visualization in live-cell imaging workflows, with relevant options also considered for workflows involving fixed-cell staining strategies.
- Product support spanning multiple reagent formats, including small-molecule dyes, fluorescent labels, and functionalized probes adapted to different staining and imaging requirements.
Customized Fluorescent Probe Development
- Custom design of ER-targeted fluorescent probes according to specific research objectives, imaging platforms, and desired localization behavior.
- Structural modification support for fluorescent dyes, including adjustment of hydrophobicity, charge characteristics, and targeting groups to improve ER localization performance and workflow fit.
- Development support for multifunctional probe designs where ER targeting needs to be combined with responsive readout logic, such as pH-sensitive, ROS-responsive, or Ca²⁺-associated fluorescence systems.
- End-to-end custom service covering molecular design, synthesis, purification, and characterization to support probe development from concept to usable material.
Staining Workflow Optimization and Application Support
- Practical guidance for designing and optimizing ER staining protocols, including concentration range, incubation time, staining conditions, and imaging timing considerations.
- Application support for different cell systems, including adherent cells, suspension cells, and primary cells, with attention to how cell state and handling influence ER signal quality.
- Suggestions for multicolor staining strategies involving ER together with other intracellular targets such as mitochondria, nuclei, or additional organelle markers.
- Troubleshooting assistance for common ER staining challenges, including fluorescence quenching, high background, weak signal contrast, or unclear intracellular localization.
Quality Control and Technical Service Support
- Supply of high-purity fluorescent products supported by strict quality control processes, with analytical characterization such as HPLC, NMR, and MS used where applicable.
- Attention to batch-to-batch consistency and reproducibility to help improve reliability in repeated staining and imaging workflows.
- Availability of technical documentation including COA, MSDS, and relevant product application information to support evaluation and implementation.
- Access to technical support from pre-sale consultation through post-sale troubleshooting, helping researchers address both product selection questions and workflow-specific issues.
Do You Need A Consultation?
BOC Sciences integrates cutting-edge fluorescence technologies to accelerate your research, helping build more reliable ER staining and multicolor imaging workflows.
Transform Your Studies with Cutting-Edge Fluorescent Products
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0048 | 3,3'-Dihexyloxacarbocyanine iodide | 53213-82-4 | Bulk Inquiry |
| A16-0064 | ReZolve-ER™ | 1404104-40-0 | Bulk Inquiry |
| A16-0096 | ER-Tracker Blue-White DPX | N/A | Bulk Inquiry |
| A16-0062 | IraZolve-L1™ | 2169684-98-2 | Bulk Inquiry |
| A16-0214 | LumiTracker ER Green | N/A | Bulk Inquiry |
| A16-0215 | LumiTracker ER Red | N/A | Bulk Inquiry |
| A16-0219 | Toto-1 | 143413-84-7 | Bulk Inquiry |
| A16-0204 | Oxazole yellow | 152068-09-2 | Bulk Inquiry |
| A16-0206 | C-Laurdan | 959839-06-6 | Bulk Inquiry |
| A16-0194 | LysoTracker Red DND-99 | 231946-72-8 | Bulk Inquiry |
High-Performance Fluorescent Tools for Your Research
- Lipid Fluorescent Probes Dyes for lipid droplets and membranes.
- pH Indicators Fluorescent sensors for intracellular pH monitoring.
- Nuclear Fluorescent Probes DNA-binding dyes for nucleus visualization.
- Fluorescent Probes Versatile tools for biomolecular and cellular imaging.
- Other Cell Fluorescent Probes Functional probes for diverse cellular imaging studies.
- Cell Proliferation Tracer Fluorescent Probes Long-term tracking of cell division processes.
- Calcium, Chloride and Other indicators Fluorescent indicators for intracellular ion flux monitoring.
- Metal Fluorescent Probes Selective sensors for intracellular metal ions.
Explore More Topics
Online Inquiry


