Mitochondrial Staining in Cells: Dyes, Workflow, and Interpretation
Cell staining of mitochondria is widely used in fluorescence-based cell analysis because mitochondrial organization is closely tied to cellular state, intracellular distribution, and imaging readout quality. In many experiments, researchers do not only want to know whether mitochondria are present. They want to understand how mitochondria are arranged in the cytoplasm, whether the network appears fragmented or elongated, whether signal intensity changes after treatment, and whether those changes should be interpreted as structural redistribution or as a shift in mitochondrial condition. That is why mitochondrial staining is often more demanding than routine labeling of nuclei or plasma membrane. The same experiment may contain both localization information and function-linked signal behavior, and these two layers are not always easy to separate. A dye can appear to "lose mitochondrial staining" even when mitochondria are still present, or it can produce bright signal that looks convincing while actually reflecting non-ideal handling, overload, or interpretation bias. A useful mitochondrial staining page therefore needs to do more than list dyes. It should help researchers understand the logic behind signal generation, the consequences of sample handling, and the workflow choices that determine whether mitochondrial fluorescence will support a reliable biological conclusion.
What Is Mitochondrial Staining?
Mitochondrial staining refers to the use of fluorescent dyes or fluorescent probes to visualize mitochondria within cells so that mitochondrial position, intracellular distribution, morphology, network organization, and in some workflows mitochondrial condition-related behavior can be assessed under fluorescence imaging. In practical research settings, mitochondrial fluorescent probe is rarely used as a purely decorative organelle label. It is usually introduced because investigators need a mitochondrial readout that can support comparative analysis across treatment groups, process development conditions, cell states, or imaging workflows. Depending on probe design, the resulting fluorescence may primarily report mitochondrial localization, may be influenced by mitochondrial membrane potential, or may reflect a mixture of structural and condition-linked information. This is why mitochondrial staining should be treated as an interpretive workflow rather than a simple staining step. For teams building robust cell-based assays, the key question is not only whether mitochondria become visible, but what the observed mitochondrial signal actually means under the chosen sample handling and imaging conditions.
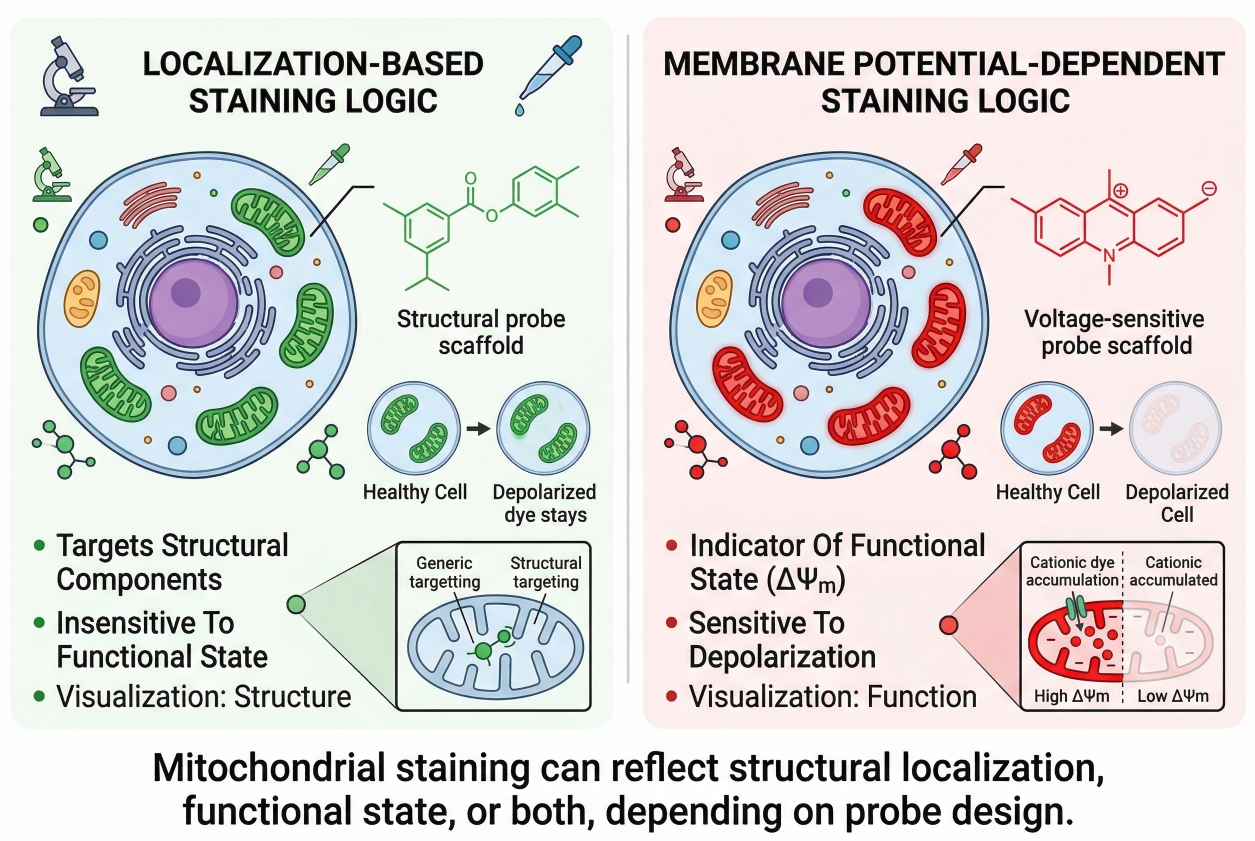 Fig. 1. Mitochondrial staining can reflect structural localization, functional state, or both, depending on probe design (BOC Sciences Authorized).
Fig. 1. Mitochondrial staining can reflect structural localization, functional state, or both, depending on probe design (BOC Sciences Authorized).
In cell analysis, mitochondrial staining is often used to answer several different questions at once. Researchers may want to compare whether mitochondria remain filamentous or become fragmented, whether the network appears broadly distributed or more perinuclear, whether organelle organization changes after compound exposure, or whether a multicolor panel can capture mitochondrial pattern together with nuclear and membrane-level features. This practical complexity is why mitochondrial staining is more demanding than many routine organelle staining workflows. A bright signal is not automatically a reliable signal, and a mitochondrial dye that performs well in one use case may be poorly matched to another. For example, a probe selected for live-cell mitochondrial imaging may provide convincing organelle contrast during short acquisition windows but may not preserve the same interpretive meaning after additional handling or delayed imaging. Likewise, dyes such as Rhodamine-type mitochondrial probes can be highly useful in fluorescence workflows, but their value depends on whether the assay is intended to emphasize structural visualization, intensity comparison, or condition-sensitive behavior.
What mitochondrial staining is designed to reveal?
Mitochondrial staining is designed to reveal much more than mitochondrial presence alone. In morphology-oriented imaging, it can show whether mitochondria appear elongated, punctate, reticular, clustered, or redistributed within the cytoplasm. In comparative treatment workflows, it can support side-by-side evaluation of mitochondrial organization under matched acquisition settings. In multiparameter assays, it can contribute one organelle channel within a larger fluorescence panel that also includes nuclear, membrane, or lysosomal markers. In some live-cell assays, the mitochondrial signal is also used as part of a broader interpretation of cell condition, but this only becomes reliable when the dye's signal logic is understood in advance. For instance, JC-1 chloride is often referenced in mitochondrial fluorescence discussions because its staining behavior is associated with mitochondrial condition-dependent signal changes rather than serving as a simple static localization label. That does not make it universally better; it makes it appropriate for certain readout objectives and less appropriate for others. A well-designed mitochondrial staining workflow therefore begins with deciding whether the experiment is mainly about structure, comparison, dynamic imaging, or condition-related interpretation.
Why mitochondrial staining is not just another organelle label?
Mitochondrial staining is not just another organelle label because mitochondrial fluorescence is unusually easy to misread when the staining mechanism is not aligned with the biological question. A nucleus stain is usually interpreted as a positional marker. A membrane stain is often interpreted as a boundary marker. Mitochondrial dyes, however, can sit at the intersection of localization, retention, and mitochondrial condition. This means that apparent loss of mitochondrial signal does not always indicate mitochondrial disappearance, and stronger fluorescence does not always indicate greater mitochondrial abundance. In practice, this distinction becomes critical in assay development, treatment comparison, and multicolor imaging where teams need the mitochondrial channel to remain analytically trustworthy across batches and decision points. Probes such as MitoMark Red I or MitoHunt Red CM-H2Xros may be attractive in red-channel imaging workflows because they can help place the mitochondrial signal in a more convenient spectral region, especially when green channels are already occupied by other assay markers. Even then, the right decision depends on whether the assay needs a structural mitochondrial readout, compatibility with short live imaging, or a channel that integrates cleanly into a larger fluorescence panel.
Why Mitochondrial Staining Matters in Cell Analysis?
Mitochondrial staining matters because mitochondria are not passive background structures. Their placement, network pattern, and image behavior often reflect how a cell is organized and how it responds under different experimental conditions. In comparative imaging studies, mitochondrial signal is frequently used to interpret whether cellular architecture remains stable, whether organelle organization appears altered, and whether the mitochondrial channel can be integrated into larger multicolor workflows. The value of this readout increases when researchers understand exactly what the stain can and cannot tell them.
- Mitochondria as a spatial and functional readout: Mitochondria are distributed throughout the cytoplasm in patterns that can appear filamentous, punctate, perinuclear, peripheral, or network-like depending on cell type and experimental context. Because many mitochondrial dyes are influenced by both localization and mitochondrial condition, the signal can serve as a combined spatial and state-related readout. That makes mitochondrial staining informative, but it also means interpretation should be tied to dye mechanism rather than appearance alone.
- Why mitochondrial imaging is important in comparative cell studies: In treatment comparison experiments, mitochondrial staining can help reveal whether intracellular organization differs between groups, whether mitochondrial pattern becomes more diffuse or more condensed, and whether overall signal behavior changes with the same imaging settings. These comparisons are especially useful when cells are analyzed under a shared acquisition workflow and when controls are included to distinguish real biology from staining drift.
- The value of mitochondrial staining in multicolor and organelle interaction workflows: Mitochondrial staining is often combined with nuclear stains, membrane labels, lysosomal dyes, and broader multicolor cell staining workflows. In these panel-based experiments, mitochondria provide one of the most informative organelle channels because they add intracellular context to cell shape, organelle proximity, and treatment-induced redistribution. This is particularly useful in assays where organelle relationships are part of the interpretation rather than a separate follow-up experiment.
Core Principles Behind Mitochondrial Staining
The most important principle in mitochondrial staining is that not all mitochondrial dyes report the same thing in the same way. Some probes are used mainly to visualize mitochondrial localization, while others show signal behavior that is more strongly influenced by membrane potential or related mitochondrial condition. Even when two dyes both produce a bright mitochondrial pattern, their interpretation can differ substantially. Understanding this distinction is the foundation for choosing the right dye and avoiding false conclusions.
- Localization-driven vs membrane potential-dependent staining logic: Localization-oriented probes are generally chosen when the main goal is to visualize where mitochondria are in the cell and how they are arranged. Membrane potential-sensitive dyes, by contrast, rely more strongly on mitochondrial condition for accumulation or signal behavior. In real experiments, this means a loss of signal may reflect reduced dye accumulation efficiency rather than disappearance of mitochondria as structures.
- Why mitochondrial signal can reflect both structure and condition: Mitochondrial fluorescence often looks like a structural image, but signal intensity and retention can still be shaped by mitochondrial state, dye concentration, incubation conditions, and imaging timing. Researchers should therefore treat mitochondrial signal as a readout with mixed meaning unless the staining logic has been carefully defined in advance.
- How sample handling influences mitochondrial staining behavior: Cell density, culture condition, incubation temperature, medium composition, exposure time, wash intensity, and delay before imaging can all influence mitochondrial staining quality. Because mitochondria are responsive organelles, small workflow differences may change signal appearance enough to complicate comparison across experiments.
- Why not all mitochondrial dyes mean the same thing: A mitochondrial dye should never be selected only because it is popular or bright. The real question is whether its signal logic matches the intended readout. A dye that works well for live-cell tracking may not be ideal for post-staining fixation. A dye that is useful in short imaging windows may be less suitable for extended acquisition. The mechanism behind the stain is therefore part of the interpretation, not just part of the reagent description.
Common Types of Mitochondrial Dyes Used in Cell Experiments
Mitochondrial dyes are best classified by staining logic and workflow role rather than by chemical name alone. This is the most practical way to select them in real cell experiments, because researchers are usually making decisions around assay purpose, sample state, imaging duration, spectral allocation, and interpretation risk. A flat list of product names is rarely enough to guide that choice. What matters more is whether a dye is better suited to structural mitochondrial visualization, membrane potential-associated readout, live-cell imaging, endpoint comparison, or multicolor integration. When mitochondrial staining is used in environments that prioritize reproducibility and interpretive consistency, the classification should always start from what the assay needs to measure and only then move toward specific dye examples.
Membrane potential-sensitive mitochondrial dyes
Membrane potential-sensitive mitochondrial dyes are commonly used when the staining readout is expected to carry information related to mitochondrial condition in addition to mitochondrial localization. These dyes often accumulate in ways that are influenced by the electrochemical state of mitochondria, which makes them useful in workflows where changes in mitochondrial signal are part of the intended interpretation. At the same time, this is exactly why they require caution. A decrease in fluorescence may reflect reduced accumulation efficiency rather than disappearance of mitochondria as physical structures, and an altered signal pattern may reflect mitochondrial condition-related change rather than redistribution alone. Rhodamine-123 is a representative example frequently associated with this category, and JC-1 chloride is another widely used example in workflows where mitochondrial signal behavior is considered together with condition-sensitive interpretation. Rhodamine 6G may also appear in mitochondrial staining discussions because rhodamine-based scaffolds are often relevant to mitochondrial accumulation behavior. These dyes can be highly informative, but only when the user is explicit that the mitochondrial channel is not being treated as a purely static morphology marker.
Localization-oriented mitochondrial dyes for structural imaging
Localization-oriented mitochondrial dyes are more appropriate when the primary goal is to visualize mitochondrial placement, intracellular patterning, and network-like structure with minimal emphasis on condition-linked signal interpretation. These dyes are often favored in imaging workflows where researchers want to compare mitochondrial distribution across treatment groups, monitor organelle arrangement in relation to cell shape, or combine the mitochondrial channel with other organelle markers in a more structurally balanced assay. In this context, a probe such as MitoMark Red I can be introduced as a practical example of a mitochondrial-targeted red fluorescent option that may be useful when the assay design benefits from moving mitochondria into a red channel. Likewise, MHI-148 may be considered in fluorescence workflows where far-red spectral placement is advantageous for panel organization. The point is not that any one product should automatically be chosen for all structural imaging applications, but that localization-oriented mitochondrial dyes are generally selected when interpretability of mitochondrial pattern is more important than extracting a condition-sensitive intensity readout.
Mitochondrial probes for live-cell imaging vs endpoint analysis
Live-cell mitochondrial staining and endpoint mitochondrial staining place different demands on dye behavior. In live-cell imaging, the dye needs to support organelle visualization without introducing unnecessary workflow instability during the acquisition window. It should produce interpretable mitochondrial signal within the practical timing constraints of the experiment and fit the broader panel if multiple channels are being used. In endpoint analysis, researchers may care more about reproducible side-by-side comparison across conditions than about extended temporal observation. Some probes perform best when imaged soon after loading into live cells, whereas others are selected because they fit a narrower but more controlled endpoint workflow. MitoHunt Red CM-H2Xros can serve as a useful example in discussions of red fluorescent mitochondrial probes for live-cell related workflows, while JC-1 chloride is better understood as a condition-associated mitochondrial probe whose interpretation depends heavily on the experimental design. Distinguishing these use cases early helps prevent the common mistake of choosing a dye based on familiarity rather than on workflow fit.
Choosing spectral regions for multicolor mitochondrial staining
In multicolor experiments, mitochondrial dye selection is strongly influenced by spectral planning. The mitochondrial channel must be assigned to a region that does not compromise the most critical markers in the assay. If the green channel is already reserved for a high-priority reporter, selecting a green-emitting mitochondrial dye may create unnecessary channel burden. In those cases, red or far-red mitochondrial probes can be more practical. MitoMark Red I and MitoHunt Red CM-H2Xros are examples that may be considered when researchers want a red-shifted mitochondrial signal, while MHI-148 may become relevant where longer-wavelength channel placement supports cleaner panel separation. Conversely, if a workflow is relatively simple and the green channel is available, Rhodamine-123 may still be a reasonable candidate depending on the assay objective. The best spectral choice is therefore not just about fluorescence color. It depends on instrument filters, channel hierarchy, autofluorescence burden, target brightness balance, and whether the mitochondrial readout is mainly structural or partly condition-related.
Choosing between structural mitochondrial dyes and potential-sensitive probes?
We can help align mitochondrial staining chemistry with your imaging purpose, interpretation goals, and multicolor assay design.
How to Choose the Right Mitochondrial Dye for Your Workflow?
Choosing the right mitochondrial dye starts with the experimental question, not with a product list. Researchers should decide whether they need a mainly structural readout, a more condition-related readout, a short live-cell imaging stain, a post-treatment endpoint label, or a multicolor-compatible mitochondrial channel. From there, dye behavior can be matched to workflow needs. This selection process is more reliable than asking which mitochondrial dye is "best" in general, because no single dye is best for every assay design.
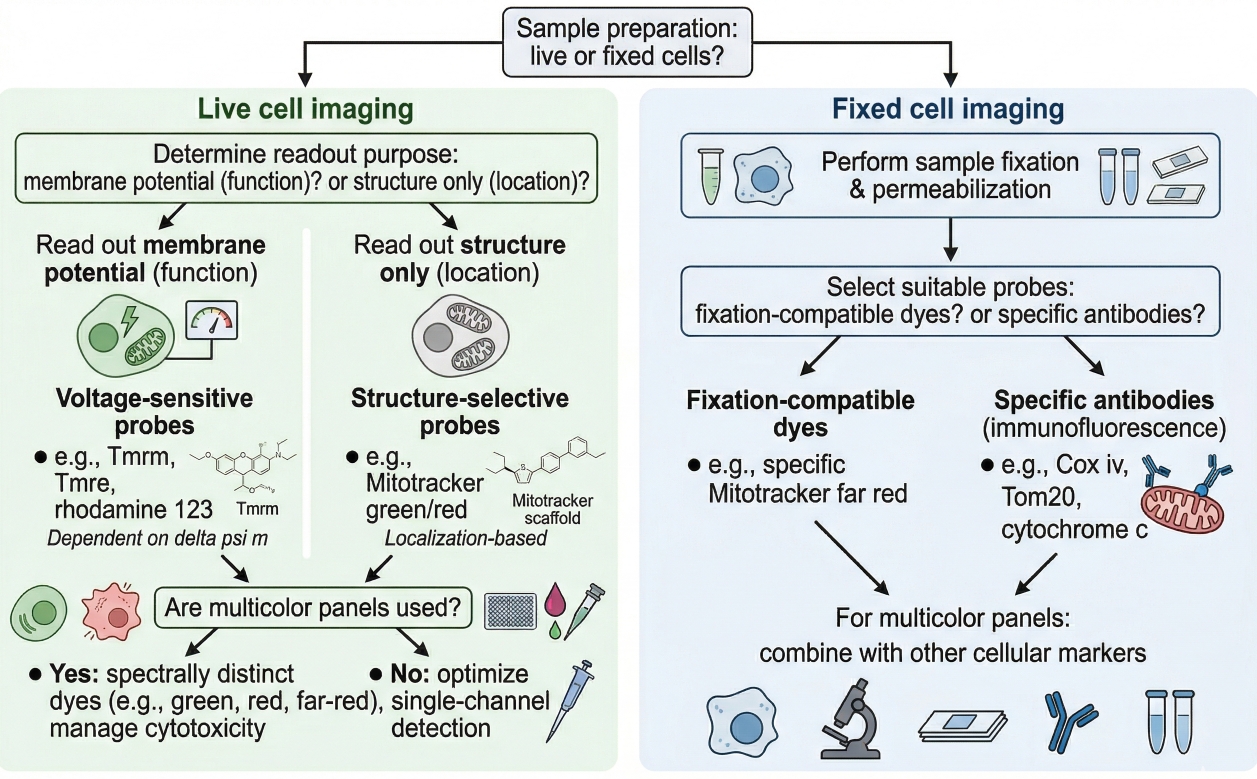 Fig. 2. Choosing a mitochondrial dye depends on sample state, readout purpose, and imaging workflow design (BOC Sciences Authorized).
Fig. 2. Choosing a mitochondrial dye depends on sample state, readout purpose, and imaging workflow design (BOC Sciences Authorized).
- Choosing dyes for live-cell mitochondrial imaging: For live-cell imaging, prioritize dyes that give interpretable mitochondrial localization under the actual acquisition duration you plan to use. Consider whether the stain needs to support short endpoint capture or repeated imaging, whether cells remain healthy throughout the observation window, and whether signal is stable enough to compare across time or treatment groups.
- Choosing dyes for fixed-cell or endpoint workflows: For endpoint workflows, the main question is whether the stain still provides meaningful mitochondrial information after the planned handling sequence. Some mitochondrial dyes are better suited to direct imaging soon after staining, while others may be more practical when a short endpoint readout is needed. Researchers should be cautious about assuming that staining signal will remain unchanged after post-staining processing.
- Matching dye behavior to structural vs functional readout needs: If the aim is to compare mitochondrial shape or intracellular distribution, select dyes whose use is more consistent with structural interpretation. If the aim includes condition-related signal behavior, then a membrane potential-sensitive option may be appropriate, but only if controls are included to separate structural conclusions from signal mechanism. In other words, the stain should reflect the question, not the other way around.
- Balancing brightness, photostability, and multicolor compatibility: A mitochondrial dye should be bright enough to visualize the network clearly without overwhelming nearby channels. In multicolor imaging, the mitochondrial channel has to be compatible with the rest of the panel in terms of excitation source, emission spacing, and relative intensity. A very bright dye can be helpful, but not if it creates bleed-through, dominates the image, or complicates comparison with dimmer channels. When needed, researchers can also compare broader how to choose the right fluorescent dye for cell staining principles before finalizing a mitochondrial stain.
Practical Workflow for Mitochondrial Staining in Cells
A good mitochondrial staining workflow is not only about reagent incubation. It is about preserving an interpretable relationship between the cells, the dye, and the imaging schedule. Many apparent mitochondrial staining problems arise because the workflow was optimized for convenience rather than for signal meaning. The most reliable approach is to control the steps that most strongly affect uptake, background, retention, and interpretation.
- Preparing cells for mitochondrial staining: Begin with cells that are healthy, appropriately plated for imaging, and compatible with the planned medium and temperature conditions. Overconfluent, stressed, or poorly attached cells often yield harder-to-interpret mitochondrial patterns. Cell preparation should also be aligned with the intended imaging platform so that mitochondrial staining is evaluated under realistic acquisition conditions rather than under a temporary pilot setup.
- Incubation, washing, and imaging timing considerations: Mitochondrial staining quality depends strongly on dye concentration, incubation duration, wash stringency, and the delay between staining and acquisition. Excessive loading can increase background or diffuse cytoplasmic signal, while insufficient loading may produce weak or incomplete labeling. Timing matters as well: signal that looks acceptable immediately after staining may shift if imaging is delayed. This is why a mitochondrial workflow benefits from early pilot runs focused on timing rather than on concentration alone.
- Live imaging vs post-staining fixation decisions: Researchers should decide early whether mitochondrial signal must be interpreted in living cells or whether a short endpoint readout is sufficient. Some stains are best used for immediate live imaging and should not be assumed to preserve the same localization behavior after downstream handling. When post-staining fixation is considered, the workflow should first test whether the observed signal remains meaningful rather than merely visible.
- Controls needed for reliable mitochondrial interpretation: Reliable interpretation typically benefits from unstained cells, matched treatment controls, and workflow-matched comparison groups imaged with the same settings. In multicolor assays, channel controls are also important because mitochondrial dyes can appear to redistribute when in fact the change arises from altered image contrast, spillover, or acquisition settings. Practical staining decisions are therefore tightly linked to cell staining protocol discipline rather than to reagent choice alone.
Need a more reliable mitochondrial staining workflow for live-cell imaging or endpoint analysis?
We can help match probe behavior, channel placement, and staining workflow to your cell model, imaging timeline, and comparison needs.
Applications of Mitochondrial Staining in Research Workflows
Mitochondrial staining is used in a broad range of research workflows because mitochondria provide one of the most informative intracellular readouts in fluorescence-based cell analysis. Their morphology, spatial organization, and signal behavior can add important context to treatment comparison, assay qualification, organelle interaction analysis, and image-based decision making. In advanced workflows, the mitochondrial channel is rarely interpreted alone. It is used together with cell morphology, nuclear information, membrane state, or other organelle markers to support a more integrated understanding of what is happening inside the cell. This is why mitochondrial staining remains highly relevant in environments that require robust imaging logic, careful workflow transferability, and interpretable fluorescent readouts across multiple project stages.
Mitochondrial staining in fluorescence microscopy and cell imaging
In fluorescence microscopy, mitochondrial staining is commonly used to obtain a visually interpretable map of mitochondrial distribution across the cytoplasm so that researchers can compare organelle organization between untreated and treated samples or across different experimental conditions. This is especially useful in assay systems where the mitochondrial channel contributes to phenotypic interpretation rather than serving as an isolated endpoint. Depending on the imaging design, probes such as MitoMark Red I may be attractive because red-shifted mitochondrial signal can integrate more smoothly into microscopy panels that already contain green nuclear or reporter channels. In other settings, Rhodamine-123 may still be selected if the workflow benefits from its mitochondrial staining behavior and the assay does not place excessive burden on the corresponding channel. The practical value of mitochondrial staining in microscopy comes from consistent workflow control: identical staining conditions, matched acquisition settings, and clear understanding of how probe mechanism affects the visual output.
Mitochondrial staining in morphology and intracellular distribution studies
One of the most established uses of mitochondrial staining is the evaluation of mitochondrial morphology and intracellular distribution. Researchers may compare whether mitochondria are elongated versus punctate, widely dispersed versus perinuclear, or organized as an extended network versus a more compact pattern. These studies are especially informative when mitochondrial architecture is part of the observed cellular response. For such work, the preferred dyes are usually those that support clean spatial interpretation instead of encouraging over-reliance on intensity alone. A red-channel option such as MitoHunt Red CM-H2Xros may be useful in morphology-oriented imaging where the mitochondrial channel needs to coexist with other fluorophores, while a far-red example such as MHI-148 may offer value in assays that require additional spectral separation. The central requirement in morphology studies is not maximum signal brightness but trustworthy organelle patterning under standardized imaging conditions.
Mitochondrial staining in multicolor organelle interaction assays
Mitochondrial staining is highly valuable in multicolor organelle interaction assays because mitochondria often need to be interpreted alongside nuclei, lysosomes, plasma membrane, cytoskeleton, or vesicular compartments within the same cell. In these workflows, the mitochondrial channel acts as one organelle layer in a larger spatial framework, helping researchers evaluate whether intracellular organization appears coordinated or disrupted under specific conditions. Spectral placement becomes particularly important here. If the green channel is already occupied by another critical readout, a red mitochondrial dye such as MitoMark Red I or MitoHunt Red CM-H2Xros may be more practical. If the panel is already crowded in the visible range, longer-wavelength options such as MHI-148 may deserve consideration. The value of mitochondrial staining in this context lies in its ability to preserve organelle context across multiple simultaneous readouts, but that value depends on selecting a dye whose channel placement and staining logic fit the entire assay architecture.
Mitochondrial staining in screening and comparative treatment analysis
In screening and comparative treatment analysis, mitochondrial staining can provide a sensitive organelle-level layer of evidence that complements broader phenotypic observations. Researchers may use the mitochondrial channel to compare signal pattern consistency, organelle redistribution, or mitochondria-associated fluorescence behavior across control and treated groups under standardized acquisition conditions. In this type of workflow, the goal is often not to make a single absolute statement about mitochondria, but to integrate mitochondrial information into a larger decision framework that includes morphology, viability-related indicators, and other fluorescent markers. For example, JC-1 chloride may be introduced in assays where a condition-sensitive mitochondrial readout is relevant to comparison logic, while Rhodamine-123 may be considered when a mitochondrial-associated fluorescent signal is needed in a more conventional live-cell comparison workflow. The key is that mitochondrial stains in screening environments should be chosen for interpretive reliability and workflow compatibility, not simply because they generate visually strong images.
Common Problems in Mitochondrial Staining and How to Avoid?
Mitochondrial staining can fail in obvious ways, such as weak signal or high background, but the bigger risk is often interpretive rather than visual. A mitochondrial image may look convincing and still support the wrong conclusion if signal mechanism is misunderstood. Troubleshooting therefore has two goals: improving staining quality and preventing mistaken biological interpretation.
- Weak or diffuse mitochondrial signal: Weak signal may result from insufficient loading, suboptimal incubation, poor cell condition, or dye behavior that does not match the sample. Diffuse signal often indicates that mitochondrial accumulation is not well resolved from cytoplasmic background or that staining conditions were too aggressive or too weak to produce clear organelle contrast. Before changing the biological interpretation, researchers should first test concentration, incubation time, cell health, and imaging timing.
- High background and non-mitochondrial fluorescence: Background fluorescence can arise from excessive dye concentration, incomplete washing, non-specific retention, or sample-associated autofluorescence. In multicolor assays, the problem becomes more serious because background in one channel can make mitochondrial localization look broader or more redistributed than it really is. Channel-aware optimization is therefore essential in mitochondrial staining, especially when several organelles are labeled together.
- Signal loss or redistribution after fixation: One of the most common errors is assuming that a dye used successfully in live cells will retain the same spatial meaning after post-staining handling. In reality, signal may weaken, redistribute, or become less informative after fixation-related steps. A visible fluorescent pattern after fixation does not automatically prove that the stain still reports the same mitochondrial feature it reported in the live state.
- Misreading functional signal changes as structural changes: This is the most important interpretive problem in mitochondrial staining. A change in fluorescence intensity or pattern may reflect altered dye accumulation behavior rather than a genuine change in mitochondrial number, mass, or localization. Researchers should therefore avoid treating every decrease as mitochondrial loss or every punctate increase as structural fragmentation unless the workflow includes appropriate controls and a dye choice consistent with that conclusion.
Best Practices for Building Reliable Mitochondrial Staining Workflows
The most reliable mitochondrial staining workflows are built by defining the question first, choosing a stain whose behavior matches that question, and validating the workflow before large-scale data generation begins. This approach reduces avoidable troubleshooting and makes downstream interpretation more robust. Mitochondrial staining becomes far more useful when it is planned as a logic-driven measurement rather than as a decorative imaging step.
- Start from the biological question before choosing the dye: Decide whether the experiment is primarily about localization, morphology, intracellular distribution, treatment comparison, or a condition-related mitochondrial signal. Once the intended readout is clear, dye selection becomes more rational and false assumptions become easier to avoid.
- Validate live-cell compatibility and imaging timing early: Short pilot experiments should test whether the stain gives interpretable signal under the exact timing and handling conditions planned for the real experiment. This is especially important for live-cell workflows, where acceptable staining at one time point does not guarantee stability across the full imaging period.
- Use controls to separate localization from functional changes: Controls should be designed to answer not only whether the stain is bright enough, but whether the observed difference is likely structural, condition-related, or workflow-induced. This is often the decisive step that prevents over-interpretation of mitochondrial fluorescence data.
- Document dye logic and workflow conditions for reproducibility: Mitochondrial staining is more reproducible when dye type, concentration range, incubation timing, wash conditions, imaging delay, acquisition settings, and interpretation criteria are documented together. This kind of structured workflow record is particularly important when the assay may later be transferred, scaled, or incorporated into broader imaging studies.
How BOC Sciences Supports Mitochondrial Staining?
BOC Sciences supports mitochondrial staining projects through integrated capabilities in fluorescent product supply, custom probe development, staining and imaging workflow optimization, and technical service support. Rather than viewing mitochondrial staining as a simple reagent selection step, we help researchers align probe type, spectral properties, staining logic, and imaging conditions with the actual goals of their experiment. This is especially important in mitochondrial workflows, where probe behavior may reflect structural localization, membrane potential dependence, or broader functional response. Our support is therefore designed to cover both standard mitochondrial visualization needs and more specialized assay development requirements.
Mitochondrial Fluorescent Dye and Probe Supply
- Access to a wide range of classic and emerging mitochondrial-targeted fluorescent dyes and probes, including commonly used mitochondrial staining options relevant to organelle imaging workflows.
- Support for mitochondrial probe selection across multiple emission regions, including blue, green, red, and near-infrared fluorescence, to better fit multicolor imaging experiments.
- Availability of highly selective mitochondrial dyes suitable for both live-cell and fixed-cell staining strategies, depending on workflow design and interpretation needs.
- Product support spanning both membrane potential-dependent and membrane potential-independent mitochondrial probes, helping researchers choose more appropriate tools for different study objectives.
Customized Mitochondrial Probe Development
- Custom design of mitochondrial probes with targeted localization properties according to specific research goals, imaging systems, and assay requirements.
- Development support for responsive probes based on mitochondrial membrane potential (ΔΨm), enabling more tailored probe solutions for condition-sensitive imaging workflows.
- Functional probe customization for applications involving mitochondrial ROS detection, ATP-associated readout, Ca²⁺ dynamics monitoring, and related fluorescence-based analysis.
- One-stop development support covering molecular design, synthesis, structural modification, purification, and characterization for customized mitochondrial fluorescent probes.
Staining and Imaging Workflow Optimization
- Practical guidance for optimizing mitochondrial staining conditions, including dye concentration, incubation time, temperature, and workflow timing.
- Application support for different experimental systems, including live cells, fixed cells, and tissue sections, with attention to how sample state influences mitochondrial signal interpretation.
- Suggestions for multicolor staining strategies involving mitochondria together with other intracellular targets such as the endoplasmic reticulum, lysosomes, or nuclei.
- Assistance with imaging condition optimization to improve signal-to-noise ratio, reduce background interference, and enhance localization accuracy in mitochondrial fluorescence readouts.
Quality Control and Technical Support Services
- Supply of high-purity mitochondrial fluorescent products supported by strict quality control processes, with analytical methods such as HPLC, NMR, and MS used where applicable.
- Attention to batch stability and product consistency to support reproducibility across repeated staining and imaging experiments.
- Availability of complete technical documentation, including COA, MSDS, and product use guidance, to support evaluation and implementation.
- Access to professional technical support for both experimental consultation and troubleshooting, helping researchers address issues from probe selection to workflow optimization.
Do You Need A Consultation?
BOC Sciences integrates cutting-edge fluorescence technologies to accelerate your research, driving next-generation solutions for drug discovery and diagnostics.
Transform Your Studies with Cutting-Edge Fluorescent Products
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0159 | HIDC iodide | 36536-22-8 | Bulk Inquiry |
| A16-0170 | Rhodamine-123 | 62669-70-9 | Bulk Inquiry |
| A16-0010 | MitoPY1 | 1041634-69-8 | Bulk Inquiry |
| A16-0005 | MitoMark Green I | 201860-17-5 | Bulk Inquiry |
| A16-0007 | MitoMark Red I | 167095-09-2 | Bulk Inquiry |
| A16-0011 | JC-1 | 3520-43-2 | Bulk Inquiry |
| A16-0093 | Rhodamine 6G | 989-38-8 | Bulk Inquiry |
| A16-0142 | Dihydrorhodamine 6G | 217176-83-5 | Bulk Inquiry |
| A16-0085 | MitoHunt Red CM-H2Xros | 167095-08-1 | Bulk Inquiry |
| A16-0104 | JC-9 Dye | 522592-13-8 | Bulk Inquiry |
High-Performance Fluorescent Tools for Your Research
- Golgi Fluorescent Probes Targeted probes for Golgi apparatus visualization.
- Nerve Terminal Probes Fluorescent tracers for synaptic activity analysis.
- pH Indicators Fluorescent sensors for intracellular pH monitoring.
- Nitric Oxide (NO) & Reactive Oxygen Species (ROS) Probes for oxidative stress and signaling detection.
- Lipid Fluorescent Probes Dyes for lipid droplets and membranes.
- Lysosomal Fluorescent Probes Acidic organelle markers for lysosome tracking.
- Metal Fluorescent Probes Selective sensors for intracellular metal ions.
- Cytoskeleton Fluorescent Probes Probes for actin and microtubule visualization.
Explore More Topics
Online Inquiry


