Live Cell Staining vs Fixed Cell Staining: How to Choose the Right Strategy
Choosing between live cell staining and fixed cell staining is one of the most important early decisions in a fluorescence experiment because it affects what kind of biology you can observe, which probes or antibodies will work, how stable the signal will be, and how reproducible the final data will be. Live cell staining is designed for researchers who need to follow dynamic processes in real time, such as membrane trafficking, mitochondrial behavior, organelle movement, or short-term changes in cell state. Fixed cell staining, by contrast, is usually the better fit when the priority is structural preservation, long acquisition windows, intracellular target accessibility, or a more controlled endpoint workflow. In practice, this is not simply a technical preference. It is a strategy choice that shapes sample handling, dye selection, imaging setup, control design, and downstream interpretation.
Many staining problems begin before the first reagent is added because the sample strategy and the detection goal are not aligned. A researcher may select a bright live-cell probe, then later decide to fix the sample and lose signal retention. Another experiment may rely on fixation to preserve morphology but unintentionally mask a sensitive target or introduce extra background during permeabilization. That is why the most reliable approach is to start with the biological question and then work backward through sample condition, staining chemistry, signal requirements, and imaging workflow. If your broader project also involves cell staining strategy selection, protocol planning, or dye comparison, this decision page should function as the first branch point in that process.
Basic Definitions of Live Cell Staining and Fixed Cell Staining
Before comparing the strengths and limitations of each approach, it is important to define what live cell staining and fixed cell staining actually mean in practical laboratory terms. Although both belong to the broader field of fluorescent cell staining, they are built around different assumptions about sample condition, target accessibility, and what kind of information the researcher wants to capture. A clear definition helps avoid a common planning error: selecting a stain based on color or familiarity before deciding whether the sample must remain alive or can be chemically preserved.
 Fig. 1. Key applications of live and fixed cell staining strategies (BOC Sciences Authorized).
Fig. 1. Key applications of live and fixed cell staining strategies (BOC Sciences Authorized).
What Is Live Cell Staining?
Live cell staining refers to the process of labeling cells while they remain viable and metabolically active. In this approach, fluorescent probes or dyes are introduced under conditions that preserve cell integrity as much as possible, allowing researchers to observe biological events in real time or within a short experimental window. The goal is not only to generate fluorescence, but to do so without disrupting the behavior being measured. Because the cells are still alive, live cell staining is especially useful for tracking dynamic processes such as organelle movement, membrane trafficking, probe uptake, changes in viability, and time-dependent cellular responses.
In practical terms, live cell staining requires careful attention to probe toxicity, incubation time, media compatibility, temperature control, and light exposure. The dyes used in these workflows must either enter cells without damaging them or interact with extracellular or membrane-associated targets in a minimally disruptive way. This makes live staining highly valuable for live cell imaging, but also more sensitive to workflow design than many endpoint assays.
What Is Fixed Cell Staining?
Fixed cell staining refers to labeling cells after they have been chemically preserved, typically through a fixation step that stabilizes morphology and halts biological activity. Once fixed, cells are no longer living samples, so the experiment shifts from observing ongoing behavior to analyzing a defined endpoint. This strategy is commonly used when the research priority is structural preservation, reproducible imaging, intracellular target accessibility, or multi-step staining workflows that would be too disruptive for live cells.
Fixed cell staining often includes additional processing steps such as permeabilization, blocking, repeated washing, and antibody-based labeling. These steps make it easier to access intracellular structures and generate stable endpoint images, but they also mean the observed signal reflects a preserved state rather than an ongoing biological process. For this reason, fixed staining is often preferred for detailed localization analysis, nuclear and cytoskeletal staining, and experiments where timing flexibility and image consistency are more important than real-time observation.
Deciding between live cell and fixed cell staining for your experiment?
We can help you compare staining strategies based on target accessibility, temporal resolution, sample preparation, and downstream analysis needs.
Why the Choice Between Live and Fixed Cell Staining Shapes the Entire Experiment?
Live and fixed staining do not simply represent two versions of the same protocol. They answer different kinds of questions. The difference becomes especially important in experiments that combine multiple readout layers. For example, a panel intended for real-time organelle observation may require live-compatible dyes, low-light imaging, and rapid acquisition. A structurally detailed endpoint panel may need fixation, permeabilization, and fluorescent antibodies or counterstains selected for stability rather than speed. Once that distinction is clear, it becomes easier to define the right workflow, avoid incompatible reagents.
- Live cell staining prioritizes temporal information, minimal perturbation, and compatibility with living systems. It is especially useful when the biological value lies in observing movement, redistribution, accumulation, or signal change over time. In live workflows, probe permeability, cytotoxicity, phototoxicity, incubation speed, media compatibility, and environmental control become central design concerns. The success of the experiment often depends on keeping cells healthy while preserving enough fluorescence intensity for repeated imaging.
- Fixed cell staining is different in purpose and logic. Once cells are chemically fixed, the experiment shifts from dynamic observation to stabilized endpoint analysis. This creates major advantages for imaging consistency, timing flexibility, and target access. Investigators can wash more thoroughly, use permeabilization to reach intracellular epitopes, apply antibody-based detection, and acquire images over a longer window without worrying about keeping cells viable. That is why fixed staining remains common in workflows focused on localization, morphology, structural comparison, and multiplex endpoint readouts.
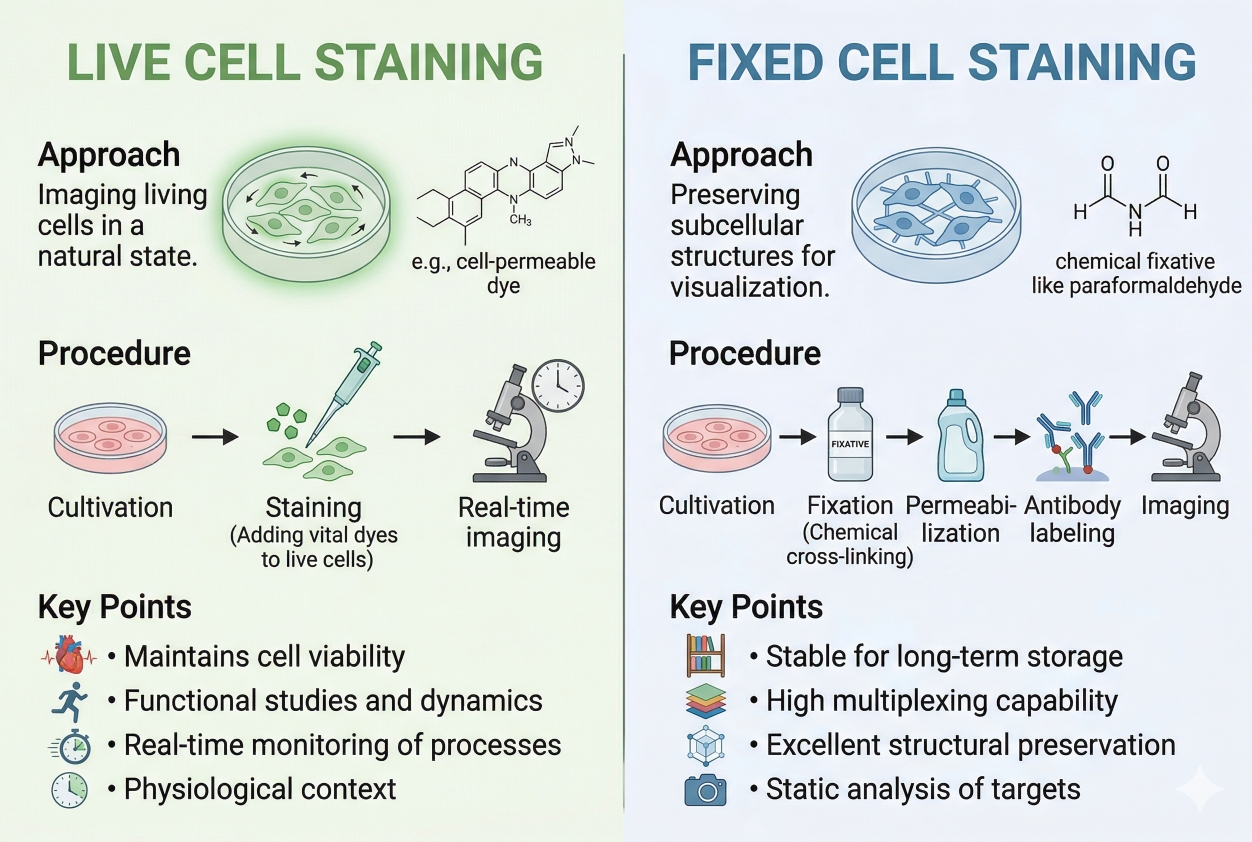 Fig. 2. Live cell staining and fixed cell staining differ in purpose, workflow, and signal behavior (BOC Sciences Authorized).
Fig. 2. Live cell staining and fixed cell staining differ in purpose, workflow, and signal behavior (BOC Sciences Authorized).
What Live Cell Staining Is Designed to Measure?
Live cell staining is most useful when the experiment depends on preserving biological activity during observation. Instead of asking only where a structure is located at the end of the assay, live staining asks what that structure is doing during the experiment. This makes it highly valuable for time-lapse imaging, short-term tracking, organelle behavior studies, membrane dynamics, viability readouts, and functional assays where the signal depends on membrane integrity, metabolic status, ion flux, or intracellular environment.
Dynamic Processes and Real-Time Observation
One of the clearest strengths of live cell staining is its ability to capture dynamic events that would be lost after fixation. Membrane internalization, mitochondrial redistribution, lysosomal trafficking, cell movement, probe uptake, and transient changes in morphology are all examples where real-time observation provides more insight than a single endpoint image. In these cases, the staining reagent is not just a marker but part of a live experimental system. Its behavior must therefore be evaluated in terms of speed, retention, compatibility with imaging media, and the extent to which it alters the cells being studied.
Because cells remain metabolically active, live staining also places more responsibility on environmental control. Temperature, CO₂ balance, osmolarity, and light exposure can influence signal quality and cell health. The imaging method matters as much as the stain itself. A dye that performs well in a brief endpoint readout may perform poorly under repeated illumination if it bleaches quickly or causes phototoxic stress. For experiments connected to live cell imaging, stain choice and image acquisition settings should therefore be planned together rather than separately.
Viability, Probe Toxicity, and Experimental Disturbance
The most common mistake in live cell staining is assuming that a probe is suitable simply because it can enter the cell and fluoresce. A useful live-cell probe must do more than generate signal. It should also preserve enough cell health and native behavior for the intended observation window. Some dyes accumulate rapidly but change membrane properties or organelle function when used at excessive concentrations. Others produce acceptable images in a short assay but become problematic in longer time-course experiments. This is why optimization should focus on the minimum concentration and shortest incubation time that still produce reliable discrimination.
Live staining is also the natural setting for many viability and membrane integrity assays. In these workflows, the signal often depends on whether a dye can cross intact membranes or bind accessible intracellular targets under defined conditions. That logic is very different from fixed workflows, where the membrane barrier may later be altered deliberately. If your experiment depends on distinguishing living and compromised cells, it is often helpful to coordinate live/fixed decisions with cell viability staining strategy early in panel design.
What Fixed Cell Staining Does Better?
Fixed cell staining becomes the better strategy when the main goal is to preserve morphology, stabilize a moment in time, or gain reliable access to intracellular targets that are difficult to stain in living cells. Fixation generally reduces the urgency of immediate imaging, supports more controlled processing steps, and allows the use of additional reagents such as permeabilization buffers, blocking systems, and fluorescent antibodies. In other words, fixed staining trades real-time biology for structural control and workflow flexibility.
Structural Preservation and Endpoint Consistency
A major advantage of fixed staining is that it can preserve cellular architecture long enough to support careful washing, multiple incubation steps, and more consistent image acquisition across samples. When the research question centers on comparative morphology, subcellular localization at an endpoint, or multiplex structural analysis, fixation often produces cleaner and more reproducible results than a live protocol. This is particularly true when experiments are processed in batches, when many fields must be imaged with the same settings, or when the workflow requires pauses between staining and acquisition.
Fixed staining is also more forgiving in scheduling. Live-cell experiments demand real-time attention because the sample continues to change. Fixed samples allow the operator to standardize timing and reduce variation introduced by staggered acquisition or changing environmental conditions. This advantage becomes especially meaningful in larger projects where multiple replicates, multiple conditions, or sequential staining steps must be coordinated.
Access to Intracellular Targets Through Permeabilization
Another core reason to choose fixed staining is access. Many intracellular proteins, nuclear epitopes, cytoskeletal components, and organelle-associated targets are difficult or impossible to reach in living cells without unacceptable disturbance. Fixation followed by permeabilization solves that problem by stabilizing cellular structure and then opening the membrane barrier in a controlled way. This enables a far broader staining repertoire, especially for antibody-based workflows and endpoint fluorescence microscopy.
That advantage is particularly relevant when the assay needs fluorescent antibodies, nuclear counterstains, or multi-step labeling sequences that are impractical in living cells. Researchers moving toward fixed workflows often combine this strategy with pages such as nuclear staining in cells or organelle-specific cell staining to refine target-specific choices. The key is to remember that fixation improves access and stability, but it can also alter epitope exposure, probe retention, and apparent localization if the staining chemistry is not compatible with the processing steps.
Key Differences Between Live and Fixed Cell Staining That Matter in Practice
The decision between live and fixed staining becomes easier when it is broken down into a few practical variables: permeability, retention after processing, signal stability, background behavior, and compatibility with the intended readout. These variables influence not only image quality but also whether the biological interpretation remains valid. A stain that is perfect for rapid live imaging may perform poorly after fixation, while a structurally reliable fixed stain may be unusable in dynamic assays because it requires permeabilization or multiple washes that living cells cannot tolerate.
Permeability and Target Access
Live staining depends on the probe's natural ability to access the intended target without destroying cell viability or function. This makes membrane permeability a decisive factor. Some dyes readily enter living cells and accumulate based on charge, lipophilicity, membrane potential, or intracellular environment. Others remain excluded unless the membrane is intentionally disrupted. Fixed staining removes much of this limitation because permeabilization can be introduced after fixation, allowing a broader range of intracellular targets to be labeled.
Retention After Fixation
One of the most overlooked questions in mixed workflows is whether a live-cell signal will survive fixation. Some live-cell probes remain associated with their target or become trapped sufficiently to retain useful fluorescence after processing. Others diffuse, redistribute, or disappear once the cells are fixed and washed. This is why it is risky to assume that a stain developed for live observation can automatically be converted into a live-then-fix workflow. If post-fix analysis is likely, the stain should be selected specifically for retention behavior rather than convenience.
Signal Stability and Imaging Window
Fixed cell staining typically supports a longer and more controlled imaging window because the sample is no longer changing in real time and repeated acquisition does not threaten viability. Live staining has the opposite challenge. The cells remain responsive to light, temperature, and media conditions, so image quality and biological integrity must be balanced continuously. Long exposure, repeated scanning, or aggressive illumination can reduce signal quality or change the biology being observed. This difference is one of the main reasons live protocols often require gentler acquisition settings and shorter imaging cycles.
Background, Reproducibility, and Workflow Control
Fixed workflows often allow stronger washing, more deliberate blocking, and more standardized reagent exposure, which can improve reproducibility. Live workflows can be more variable because the cells themselves remain active and sensitive. At the same time, fixation can also introduce its own problems, including altered morphology, extraction of weakly associated signals, or increased background after permeabilization. In short, fixed staining usually offers more procedural control, while live staining offers more biological immediacy. Choosing correctly means deciding which kind of uncertainty is more acceptable for the specific experimental objective.
Table 1. Practical differences between live cell staining and fixed cell staining.
| Feature | Live Cell Staining | Fixed Cell Staining |
|---|---|---|
| Main Purpose | Observe dynamic processes and preserve biological activity during imaging. | Stabilize structure and analyze endpoint localization or morphology. |
| Cell Status | Cells remain viable and responsive during the assay. | Cells are chemically preserved and no longer biologically active. |
| Target Accessibility | Limited to probes that can access targets without disruptive processing. | Broad intracellular access after fixation and permeabilization. |
| Signal Retention | Must be optimized for live observation; may not survive fixation. | Generally more stable for endpoint imaging and repeated acquisition. |
| Workflow Sensitivity | Highly sensitive to media, temperature, light, and incubation stress. | More tolerant of washing, multiple incubation steps, and scheduling flexibility. |
| Best Fit | Time-lapse imaging, viability readouts, dynamic organelle behavior, cell tracking. | Structural imaging, intracellular staining, antibody-based labeling, endpoint comparison. |
| Common Risks | Cytotoxicity, phototoxicity, signal loss over time, weak retention after fixation. | Epitope masking, extraction artifacts, fixation-induced background, over-permeabilization. |
How to Match the Strategy to Your Readout and Experimental Goal?
The best way to choose between live and fixed staining is to define the readout first. What exactly needs to be measured, and what kind of sample handling is required before that measurement becomes reliable? Once that question is answered, the appropriate staining path usually becomes much clearer. The strategy should be driven by the biology, not by habit.
Microscopy Workflows
For microscopy, live staining is typically the right choice when the experiment requires time-lapse observation, rapid response measurement, or preservation of native movement. This includes monitoring organelle dynamics, uptake behavior, membrane events, and transient changes in signal intensity. In these settings, dyes should be selected for low perturbation, appropriate brightness under gentle imaging conditions, and compatibility with the environmental controls available on the microscope.
Fixed staining is often superior when the microscopy goal is detailed structural imaging, comparative localization across conditions, or robust multiplex endpoint analysis. Because the sample is stabilized, the operator can spend more time optimizing focus, channel order, and exposure settings without worrying about the sample changing during acquisition. Fixed workflows are therefore particularly effective for detailed localization studies, intracellular targets, and panels that need repeated washing or antibody-based amplification.
Flow Cytometry and Endpoint Population Analysis
In flow-based workflows, live staining is commonly used for rapid viability discrimination, membrane integrity assays, and some direct probe-based analyses where the cells are read shortly after staining. Fixed workflows become more attractive when intracellular targets must be measured or when sample stabilization is needed before acquisition. In those cases, fixation and permeabilization can create access to intracellular markers, but the staining chemistry has to be compatible with those processing steps. That is especially important when combining viability discrimination with later fixation, because only certain viability reagents retain useful signal under those conditions.
Endpoint Versus Time-Course Designs
A useful rule of thumb is simple. If the question depends on what happens over time, start from a live-cell design. If the question depends on what is present at a defined endpoint and where it is located after full processing, start from a fixed-cell design. There are mixed workflows where live staining is followed by fixation, but those should be treated as a separate strategy rather than as an automatic extension of a live protocol. Probe retention, redistribution risk, and post-fix signal behavior all need to be checked before that hybrid approach can be trusted.
Researchers often benefit from approaching this decision through a short hierarchy: first define whether the signal is dynamic or endpoint-based, then determine whether target accessibility requires permeabilization, then choose the stain family, and finally refine imaging settings. This sequence also reduces confusion later when designing multicolor cell staining panels or troubleshooting inconsistent results.
Need help selecting dyes and protocols for live or fixed cell workflows?
We can help match probe compatibility, fixation conditions, and imaging requirements to ensure optimal signal and experimental reliability.
Common Mistakes When Switching Between Live and Fixed Cell Staining
Some of the most persistent staining failures occur when a workflow drifts from one strategy into the other without re-optimizing the design. A live stain may be applied, followed by fixation because the investigator wants longer imaging time. A fixed protocol may be simplified by omitting a step, even though the omitted step was the reason the target was accessible in the first place. These changes often seem small, but they can completely alter the meaning of the signal.
- Assuming live-cell probes are automatically fixation compatible: many live stains do not retain the same localization or intensity after fixation and washing, so the signal may weaken, redistribute, or disappear.
- Using fixation without considering target sensitivity: chemical fixation preserves structure, but it can also alter epitope exposure, increase background, or change apparent morphology if conditions are too harsh.
- Ignoring permeabilization effects: permeabilization is essential for many intracellular targets, but excessive treatment can extract weakly retained components or distort delicate structures.
- Transferring imaging settings directly from fixed to live samples: exposure conditions that are acceptable for fixed specimens may be too intense for living cells and can lead to photobleaching or phototoxic stress.
- Building the panel around color instead of compatibility: dye choice should reflect sample state, retention behavior, brightness needs, and acquisition method, not just spectral preference.
- Skipping control redesign after changing strategy: once the workflow shifts from live to fixed, or vice versa, control conditions should also be reevaluated because background and signal behavior can change substantially.
Most of these errors are avoidable when the experiment is treated as a coherent workflow rather than a collection of interchangeable steps. That is why it helps to coordinate this decision with a dedicated fluorescent dye selection guide and a deeper troubleshooting page whenever the first round of results is inconsistent.
A Practical Decision Framework for Choosing the Right Strategy
The most effective decision framework begins with a biological question, not with a dye catalog. Start by asking whether you need to observe a living process or preserve a defined endpoint. Then determine whether your target is accessible in living cells or requires fixation and permeabilization. Next consider how much signal stability is needed, how long the imaging or acquisition window will be, and whether the workflow must support multicolor readouts, antibody staining, or downstream analysis on a fixed sample.
If the experiment depends on dynamic behavior, rapid response, or maintaining a near-native cellular state, a live staining workflow is usually the stronger choice. In that case, prioritize live-compatible probe chemistry, low perturbation, shorter incubations, environmental control, and acquisition settings that minimize phototoxicity. If the experiment depends on structural preservation, intracellular target access, high-content endpoint analysis, or flexible timing, fixed staining is usually more reliable. In that case, prioritize fixation compatibility, permeabilization logic, washing robustness, and dyes or antibodies selected for stable signal after processing.
In mixed projects, a two-stage logic often works best. First, use live staining when the primary value lies in dynamic observation or viability-sensitive measurement. Second, use a separate fixed-cell workflow for structural validation or intracellular endpoint analysis rather than forcing one protocol to do both jobs poorly. This separation often produces cleaner data and more defensible interpretation than attempting to create a compromise workflow that is only partially suitable for each objective.
For researchers building a broader workflow, the next useful pages are usually multicolor cell staining, organelle cell staining strategy, and step-by-step cell staining protocol. Together, these pages help translate the live-versus-fixed decision into a workable experimental design.
How BOC Sciences Supports Live Cell Staining and Fixed Cell Staining Workflows?
BOC Sciences supports live cell staining and fixed cell staining workflows through integrated capabilities in fluorescent dye supply, custom probe development, staining strategy optimization, and quality-focused supply support. Rather than treating live-cell and fixed-cell staining as interchangeable applications, we help researchers align probe behavior, sample handling sequence, fluorescence properties, and imaging goals with the actual requirements of the experiment. This is especially important because dyes suitable for live-cell imaging often need high cell permeability and low disturbance, while fixed-cell workflows place greater emphasis on signal retention, fixation tolerance, and compatibility with permeabilization and downstream staining steps. Our support is therefore structured to cover both routine staining needs and more specialized workflow development requirements.
Fluorescent Dye Supply for Live-Cell and Fixed-Cell Applications
- Access to fluorescent dyes suited to both live-cell and fixed-cell staining workflows, including high-permeability, lower-disturbance probes for live-cell use and more stable dyes compatible with fixation and permeabilization steps.
- Support for selecting fluorophores across multiple wavelength regions to improve compatibility with multichannel imaging, co-localization analysis, and broader fluorescence workflow design.
- Availability of target-oriented staining reagents for nuclei, membrane structures, cytoskeleton, and a variety of subcellular organelles commonly used in fluorescence-based cell analysis.
- Product supply based on high-purity, high-stability standardized materials to help improve consistency and reproducibility across repeated staining experiments.
Customized Dye and Probe Development Services
- Custom design of fluorescent probes according to the distinct requirements of live-cell imaging or fixed-cell staining applications, including organelle-targeted and biomolecule-directed probe development.
- Structural optimization of dye molecules to improve cell permeability, reduce workflow disturbance, or enhance post-fixation signal stability depending on the intended staining route.
- Support for fluorescent conjugation strategies involving antibodies, proteins, peptides, and related targeting components for immunofluorescence and other specialized staining applications.
- Development of responsive probe systems, including pH-, ROS-, or enzyme-related fluorescent probes, supported by one-stop services from molecular design and synthesis to preparation and validation.
Staining Strategy Optimization and Application Support
- Practical guidance for designing and optimizing both live-cell and fixed-cell staining workflows, including concentration range, incubation time, staining order, fixation compatibility, and imaging parameter considerations.
- Assistance with selecting more suitable dyes according to whether cell permeability, rapid live-cell uptake, fixation tolerance, or downstream processing compatibility is the higher priority.
- Support for multicolor staining and channel allocation strategies that reduce spectral overlap and improve compatibility between structural, organelle, and functional readouts.
- Troubleshooting help for common staining challenges, including photobleaching, high background, uneven labeling, and reduced interpretability caused by workflow mismatch or non-ideal dye behavior.
Quality Control and Stable Supply Assurance
- Fluorescent staining products supported by strict quality testing using analytical methods such as HPLC, NMR, and MS where applicable.
- Availability of detailed quality documentation, including COA and batch-consistency support, to facilitate evaluation, protocol standardization, and repeated use.
- Flexible production and supply support suitable for laboratory-scale studies as well as broader project purchasing needs.
- Attention to long-term supply continuity and batch stability to help support sustained research programs and ongoing staining workflow development.
Do You Need A Consultation?
BOC Sciences integrates cutting-edge fluorescence technologies to accelerate your research, driving next-generation solutions for drug discovery and diagnostics.
Transform Your Studies with Cutting-Edge Fluorescent Products
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0201 | DAPI dihydrochloride | 28718-90-3 | Bulk Inquiry |
| A16-0144 | Nile Red | 7385-67-3 | Bulk Inquiry |
| A16-0033 | 6-Carboxyfluorescein | 3301-79-9 | Bulk Inquiry |
| A16-0186 | NBD C6-Ceramide | 86701-10-2 | Bulk Inquiry |
| A16-0044 | C6 NBD Sphingomyelin | 94885-04-8 | Bulk Inquiry |
| A16-0059 | C6 NBD Ceramide | 94885-02-6 | Bulk Inquiry |
| A16-0188 | 16:0-DPH PC | 98014-38-1 | Bulk Inquiry |
| A16-0036 | Calcein Blue | 54375-47-2 | Bulk Inquiry |
| A16-0089 | CellHunt CM-DiI | 180854-97-1 | Bulk Inquiry |
| A16-0082 | DiOC16(3) | 161433-32-1 | Bulk Inquiry |
High-Performance Fluorescent Tools for Your Research
- Calcium, Chloride and Other indicators Fluorescent indicators for intracellular ion flux monitoring.
- Endoplasmic Reticulum Fluorescent Probes ER-targeted dyes for organelle structure analysis.
- Nitric Oxide (NO) & Reactive Oxygen Species (ROS) Probes for oxidative stress and signaling detection.
- Cytoskeleton Fluorescent Probes Probes for actin and microtubule visualization.
- Nuclear Fluorescent Probes DNA-binding dyes for nucleus visualization.
- Cell Proliferation Tracer Fluorescent Probes Long-term tracking of cell division processes.
- Mitochondrial Fluorescent Probes Targeted dyes for mitochondrial structure and function.
- Metal Fluorescent Probes Selective sensors for intracellular metal ions.
Explore More Topics
Online Inquiry


