A Practical Guide to Nuclear Staining in Cells: Principles, Selection, and Applications
Nuclear staining is one of the most widely used techniques in fluorescence-based cell analysis because it solves several experimental needs at the same time. It helps researchers identify cells, define intracellular orientation, support image segmentation, normalize field-to-field comparisons, and interpret how other fluorescent signals are positioned relative to a stable structural reference. In microscopy, a reliable nuclear channel often determines whether downstream organelle or localization data can be interpreted cleanly. In phenotypic screening and comparative imaging, it frequently acts as the analytical anchor that makes multiparameter workflows scalable and reproducible. Even though nuclear staining is common, choosing the right nuclear stain is not a trivial decision. Different dyes vary in permeability, brightness, photostability, background behavior, fixation compatibility, and suitability for multicolor workflows. A stain that works well in fixed-cell endpoint imaging may not be appropriate for live-cell observation. A dye that produces strong nuclear contrast by itself may become a poor option if it occupies a critical channel in a more complex panel. In drug discovery, biotech, and CDMO research settings, where experiments often require strong reproducibility across large datasets or multiple conditions, nuclear staining should be treated as part of workflow design rather than as a routine add-on.
What Is Nuclear Staining?
Nuclear staining is the use of fluorescent dyes or fluorescent probes to label cell nuclei so that nuclear structures can be visualized, counted, segmented, and interpreted within broader cell-based workflows. In practice, it is one of the most foundational layers in fluorescence imaging because the nucleus is usually the most recognizable and analytically stable intracellular structure in the field. For researchers working in pharma, biotech, new drug discovery, and CDMO environments, nuclear staining is not only a visualization tool. It is often part of the measurement logic of the assay itself, supporting image normalization, cell identification, morphology analysis, and interpretation of how other intracellular signals are distributed relative to the nucleus. This is why nuclear staining should not be reduced to the idea of "making nuclei visible." A nuclear stain becomes useful only when the signal is strong enough to define nuclei clearly, specific enough to distinguish them from surrounding regions, and compatible enough with the rest of the workflow to preserve interpretability. In some assays, the nucleus is mainly a structural reference. In others, nuclear morphology is itself part of the biological output. In high-content imaging, the nuclear channel may serve as the starting point for segmentation and feature extraction. In multicolor microscopy, it may function as the central spatial anchor for every other channel in the experiment. The practical meaning of nuclear staining therefore depends on how the nuclear signal will be used, not just on whether fluorescence is present.
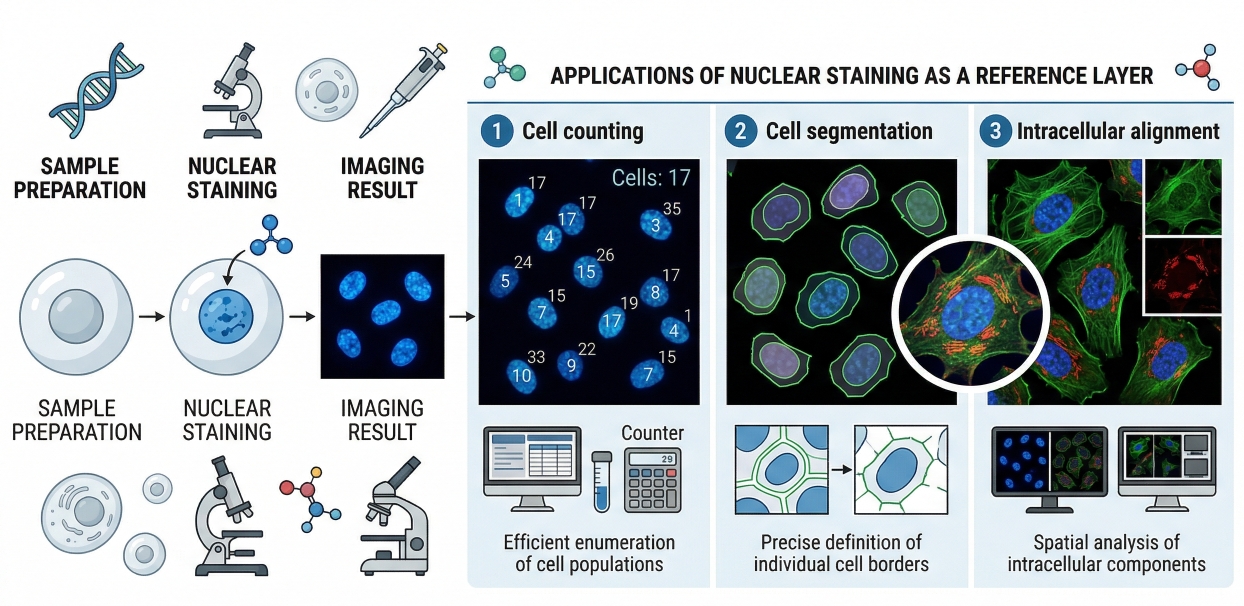 Fig. 1. Nuclear staining provides a clear reference layer for cell counting, segmentation, and intracellular imaging (BOC Sciences Authorized).
Fig. 1. Nuclear staining provides a clear reference layer for cell counting, segmentation, and intracellular imaging (BOC Sciences Authorized).
At the technical level, nuclear staining refers to the introduction of a dye or fluorescent probe that preferentially associates with nuclear material, most often through interaction with DNA-rich structures, to generate contrast between the nucleus and the rest of the cell. This contrast allows nuclei to be identified as discrete intracellular objects rather than as part of a general fluorescent background. The analytical value of this is substantial. A well-defined nuclear signal can support cell counting, define object boundaries for image processing, improve comparison between treated and untreated samples, and provide a reliable internal landmark for interpreting the position of organelles, proteins, or other fluorescent features. Because of this, nuclear staining is often one of the first channels planned in assay development, not one of the last.
What Nuclear Staining Is Designed to Reveal in Cells?
Nuclear staining is designed to reveal more than nuclear presence alone. In many workflows it is used to define how many intact cells are present, where each cell is positioned in the field, whether nuclei show changes in size, condensation, or shape, and how other fluorescent signals relate spatially to the nuclear region. In imaging-based assays, this may help distinguish whether a signal is truly nuclear, merely perinuclear, or broadly cytoplasmic. In screening applications, nuclear staining can provide the reference layer required for automated segmentation and per-cell measurement. In morphology-driven studies, nuclear shape or distribution may also become part of the biological interpretation. This is why the question "What should the nuclear channel reveal?" is so important during workflow design. Some projects need a simple and stable reference signal, while others need a channel that supports more nuanced interpretation of nuclear phenotype.
Why Nuclear Staining Is More Than Just a Counterstain?
Nuclear staining is often described casually as a counterstain, but in many fluorescence-based experiments that description understates its real analytical role. A counterstain is often thought of as supplementary visual context, whereas the nuclear channel frequently functions as a core structural and computational layer. In multicolor experiments, it can determine how the rest of the panel is interpreted by anchoring spatial relationships between intracellular signals. In high-content imaging, it is often the first object class identified by image analysis software and may determine how every downstream measurement is assigned on a per-cell basis. In comparative assays, it helps distinguish changes in cell abundance from changes in target-specific fluorescence. For these reasons, nuclear staining should be selected with the same level of care used for any biologically important channel. It affects not only what the image looks like, but how reliably the full experiment can be analyzed and reproduced.
Need a nuclear staining solution for segmentation, phenotypic imaging, or multichannel assay design?
We can help align nuclear signal quality, fluorescence compatibility, and staining workflow design with your experimental objectives.
Why Nuclear Staining Matters in Fluorescence-Based Cell Analysis?
The importance of nuclear staining becomes clear when a workflow moves beyond simple visualization and begins to rely on reproducible interpretation. In fluorescence-based cell analysis, the nucleus is often the most stable and most easily recognized intracellular structure. That stability makes it valuable across microscopy, high-content imaging, phenotypic screening, and multicolor analysis. A strong nuclear channel helps reduce ambiguity in both qualitative and quantitative workflows, especially when multiple signals must be compared across treatment groups or time points.
- Nuclear Staining as a Reference Layer for Imaging and Segmentation: In imaging analysis, nuclear staining is frequently the starting point for segmentation because nuclei are usually discrete, high-contrast objects that can be recognized more consistently than the whole-cell boundary. This makes the nuclear signal especially useful for automated image analysis and high-throughput workflows, where consistent object definition is essential. When nuclei are reliably segmented, downstream measurements such as cell count, per-cell fluorescence, spatial distribution, and treatment-associated morphology become more robust. This is why many high-content platforms treat the nuclear channel not as a decorative stain, but as the analytical core of image-based quantification.
- Why Nuclear Signals Improve Interpretability in Multicolor Workflows: In multicolor workflows, nuclear staining improves interpretability by giving other channels a spatial frame of reference. A membrane or organelle signal becomes much easier to interpret when its location relative to the nucleus is clear. This is especially important in multicolor cell staining, where several channels compete for attention and false conclusions can arise if structural context is weak. A well-placed nuclear channel helps distinguish nuclear localization from cytoplasmic accumulation, supports perinuclear pattern recognition, and reduces ambiguity in co-localization analysis. In practical terms, it often improves not only the clarity of the final image, but also the reliability of the biological conclusion drawn from it.
- The Role of Nuclear Staining in Cell Counting, Morphology, and Comparative Analysis: Nuclear staining is also important because it supports several of the most common readouts in cell-based assays: cell count, nuclear morphology, and condition-to-condition comparison. Changes in nuclear number can indicate altered cell abundance or loss of intact cells, while changes in nuclear size, condensation, or shape can provide useful contextual clues about treatment-related cell state. In comparative imaging, the nuclear layer helps distinguish whether a decrease in total signal arises from fewer cells, altered morphology, or genuine target-specific biology. This makes nuclear staining especially useful in drug discovery and mechanism-oriented workflows where the same assay may need to support both descriptive imaging and more standardized quantitative interpretation.
Core Principles Behind Fluorescent Nuclear Staining
A successful nuclear staining workflow depends on more than choosing a dye with a familiar name. Different nuclear stains behave differently because their performance is shaped by nuclear access, fluorescence mechanism, sample state, and compatibility with the rest of the imaging system. Understanding these core principles helps research teams choose stains more rationally and avoid treating all nuclear dyes as interchangeable.
- DNA-Associated Staining Logic and Nuclear Signal Generation: Most fluorescent nuclear stains generate useful signal by associating with DNA-rich nuclear material, creating higher contrast in the nucleus than in surrounding cellular regions. This association is what makes the nucleus such a dependable fluorescence target. However, DNA-associated signal generation is not identical across all dyes. Some reagents provide very strong endpoint staining in fixed-cell workflows, while others are more suitable for live-cell nuclear labeling because of their permeability and lower workflow disruption. The critical point is that nuclear signal should be evaluated not only by its intensity, but by whether it creates crisp nuclear contrast that remains analytically useful under the chosen conditions.
- Cell Permeability and Accessibility in Live vs Fixed Workflows: Permeability is one of the main variables that separates live-cell and fixed-cell nuclear staining strategies. In fixed cells, nuclear accessibility is usually easier to manage because processing can increase reagent access to intracellular structures. In live cells, the stain must reach the nucleus under conditions that preserve cell integrity and still generate a usable signal. This difference has direct workflow consequences. A dye that produces excellent fixed-cell nuclear contrast may not cross intact cell barriers in a way that is useful for live imaging, while a live-compatible stain may be selected specifically because it balances access with lower disturbance. For this reason, nuclear stain selection should always be matched to whether the workflow emphasizes live observation, endpoint imaging, or a transition between the two.
- Signal Intensity, Background, and Nuclear Contrast: The best nuclear stain is not simply the brightest one. What matters is the balance between signal intensity, nuclear contrast, and background control. A very strong dye may oversaturate the nucleus and conceal useful internal detail, while a weaker dye may fail to create enough contrast for reliable segmentation or field-to-field comparison. Background can also compromise nuclear interpretation if non-nuclear fluorescence is elevated or if the signal spills into neighboring regions of the image. In practice, a useful nuclear channel is one that produces consistent nucleus-to-cytoplasm contrast without overwhelming the rest of the panel or creating avoidable image analysis problems.
- Why Not All Nuclear Dyes Behave the Same Way: Nuclear dyes differ in more than color. They vary in permeability, brightness, photostability, suitability for live versus fixed samples, spectral position, and how well they coexist with other fluorophores. Some work especially well as simple structural references, while others are chosen because they fit better into a specific multicolor layout. This is why nuclear stain selection should not be reduced to "blue dye versus red dye." In real workflows, the question is which nuclear stain best supports the analytical purpose of the experiment while remaining compatible with sample handling and panel design.
Common Types of Nuclear Stains Used in Cell Experiments
Nuclear stains are often grouped together under one broad label, but in practical cell experiments they serve different purposes and are selected for very different workflow reasons. In pharma, biotech, and drug discovery imaging workflows, researchers typically do not choose a nuclear stain simply because it can bind nucleic acids. They choose it because they need a nuclear channel that performs well under a defined experimental condition, such as fixed-cell endpoint imaging, live-cell observation, automated segmentation, or multicolor fluorescence analysis. A useful way to approach nuclear stain selection is to classify reagents according to how they are used in real workflows. Some stains are best suited to fixed-cell structural visualization, where high nuclear contrast and reproducible endpoint imaging are the main priorities. Others are more useful in live-cell settings, where permeability and signal usability must be balanced against lower workflow disturbance. Additional nuclear staining options become valuable in multicolor panel design, where spectral position can be just as important as staining behavior. This workflow-based view helps research teams choose more rationally and reduces the risk of using a familiar stain that does not actually fit the assay design.
DNA-Binding Fluorescent Dyes for Fixed-Cell Nuclear Staining
One of the most widely used groups in nuclear staining includes DNA-binding fluorescent dyes that perform especially well in fixed-cell workflows. A representative example is DAPI dihydrochloride, which is commonly used when researchers need strong endpoint nuclear contrast, sharp nucleus-to-cytoplasm separation, and a reliable reference layer for image segmentation or fixed-cell fluorescence microscopy. Another widely recognized example is Hoechst 33258, which is frequently used in nucleus-focused imaging where clear DNA-associated signal and reproducible nuclear visualization are important. In these fixed-cell contexts, the nuclear channel often becomes one of the most stable structural layers in the assay, supporting automated cell counting, morphology comparison, and image-based screening. From a technical standpoint, these dyes are typically favored when the workflow requires crisp nuclear boundaries, consistent endpoint performance, and compatibility with broader fluorescence-based analysis rather than dynamic live-cell observation.
Nuclear Dyes Compatible with Live-Cell Imaging
A second important category includes nuclear dyes selected for workflows in which the nucleus must be visualized while the cells remain viable enough for live observation or time-dependent imaging. In this context, the stain must not only label nuclei effectively, but also remain compatible with the practical conditions of live-cell microscopy, including imaging duration, media conditions, and the need to preserve biologically meaningful cellular behavior. Examples from commonly used nucleic-acid-binding probe families include YOYO 1 and YO-PRO 3, which may be considered in workflows where researchers need strong nucleic-acid-associated fluorescence and are evaluating different spectral or permeability-related options. The main technical challenge in this category is not just whether nuclei can be labeled, but whether the signal remains interpretable throughout the live workflow without introducing excessive background, channel imbalance, or reduced assay usability. This is why live-cell nuclear stain selection should be based on practical imaging performance rather than on endpoint brightness alone.
Blue, Green, and Red Nuclear Staining Options in Multicolor Panels
In multicolor cell imaging, nuclear stains are often chosen according to spectral placement as much as according to DNA-binding performance. Blue-region nuclear dyes such as DAPI dihydrochloride and Hoechst 33258 are commonly used because they often leave green, orange, red, and near-infrared channels available for organelle probes, viability stains, or target-specific markers. However, alternative nuclear staining options become useful when panel architecture requires a different spectral layout. For example, TO-PRO 1, TOTO 1, and TOTO 3 represent nucleic-acid-binding probe options that may be considered when researchers need to reassign the nuclear channel to other spectral regions in a multicolor assay. In practical panel design, the nuclear stain should support segmentation and structural interpretation without occupying a channel that is better reserved for a weaker or more biologically critical signal. This makes spectral planning a central part of nuclear stain selection whenever the workflow involves multiple fluorophores.
Nuclear Probes Used as Structural References vs State-Sensitive Readouts
Most nuclear stains are used primarily as structural reference channels, but the way they are applied can differ depending on whether the nucleus is serving only as a segmentation anchor or also as part of the biological readout. In many assays, reagents such as DAPI dihydrochloride or Hoechst 33258 are selected because they provide a stable nuclear layer for counting cells, defining object position, and anchoring interpretation of other fluorescence channels. In other workflows, nuclear-associated probe families such as YOYO 1, YO-PRO 3, TOTO 1, and TOTO 3 may be evaluated when researchers need different signal characteristics, spectral positions, or workflow-specific nucleic-acid readout behavior. The important point is that the nuclear channel should be chosen according to its analytical role. If it is needed mainly for segmentation and normalization, contrast and reproducibility may matter most. If nuclear morphology or nuclear-associated phenotype interpretation is also important, then avoiding oversaturation, preserving structural detail, and maintaining compatibility with careful image analysis become much more critical.
How to Choose the Right Nuclear Stain for Your Workflow?
Selecting a nuclear stain is easiest when it is treated as a workflow decision instead of a reagent habit. The right choice depends on sample state, readout platform, channel architecture, and the practical role of the nuclear signal in the experiment. A stain that is ideal for one workflow may be a poor fit for another, even when both are described broadly as nuclear staining.
- Choosing Nuclear Stains for Live Cells vs Fixed Cells: The first selection question is whether the nuclear stain is needed in live cells, fixed cells, or both. If the assay depends on dynamic observation or repeated imaging before endpoint processing, live-cell compatibility becomes essential. If the workflow emphasizes endpoint morphology, segmentation, or multistep fixed-cell staining, fixed-cell compatibility may matter more than live performance. In many research settings, this is the most decisive selection factor because it immediately narrows the set of stains that can behave meaningfully under the required sample conditions.
- Matching Dye Color to Other Channels in Multicolor Imaging: In multicolor imaging, the nuclear channel should be assigned strategically rather than by default. A very convenient nuclear dye may not be appropriate if it occupies a channel needed for a weaker biological target or creates avoidable spillover into nearby channels. The right nuclear stain is therefore often the one that supports strong segmentation while leaving more analytically valuable channels available for the rest of the panel. This is especially important when nuclear staining is combined with organelle channels, viability dyes, or target-specific fluorescence layers.
- Balancing Brightness, Background, and Photostability: Brightness alone is not a sufficient selection criterion. A very bright nuclear stain may complicate image analysis if it oversaturates nuclei or creates channel imbalance, while a dimmer stain may be perfectly suitable if it provides strong nucleus-to-background contrast. Photostability also matters in repeated or prolonged imaging workflows, especially where the nuclear layer must remain consistent across fields or time points. The best stain is one that provides adequate contrast with manageable background and stable behavior under the intended imaging conditions.
- When to Prioritize Segmentation Quality, Morphology, or Panel Compatibility: Different workflows emphasize different aspects of nuclear performance. High-content imaging often prioritizes segmentation quality. Comparative phenotypic analysis may place more value on preserving nuclear morphology. Multicolor imaging may prioritize panel compatibility above raw nuclear intensity. Recognizing which of these goals is most important helps prevent poor choices based on generic nuclear staining recommendations. In practice, the right nuclear stain is the one that solves the most important analytical problem of the assay.
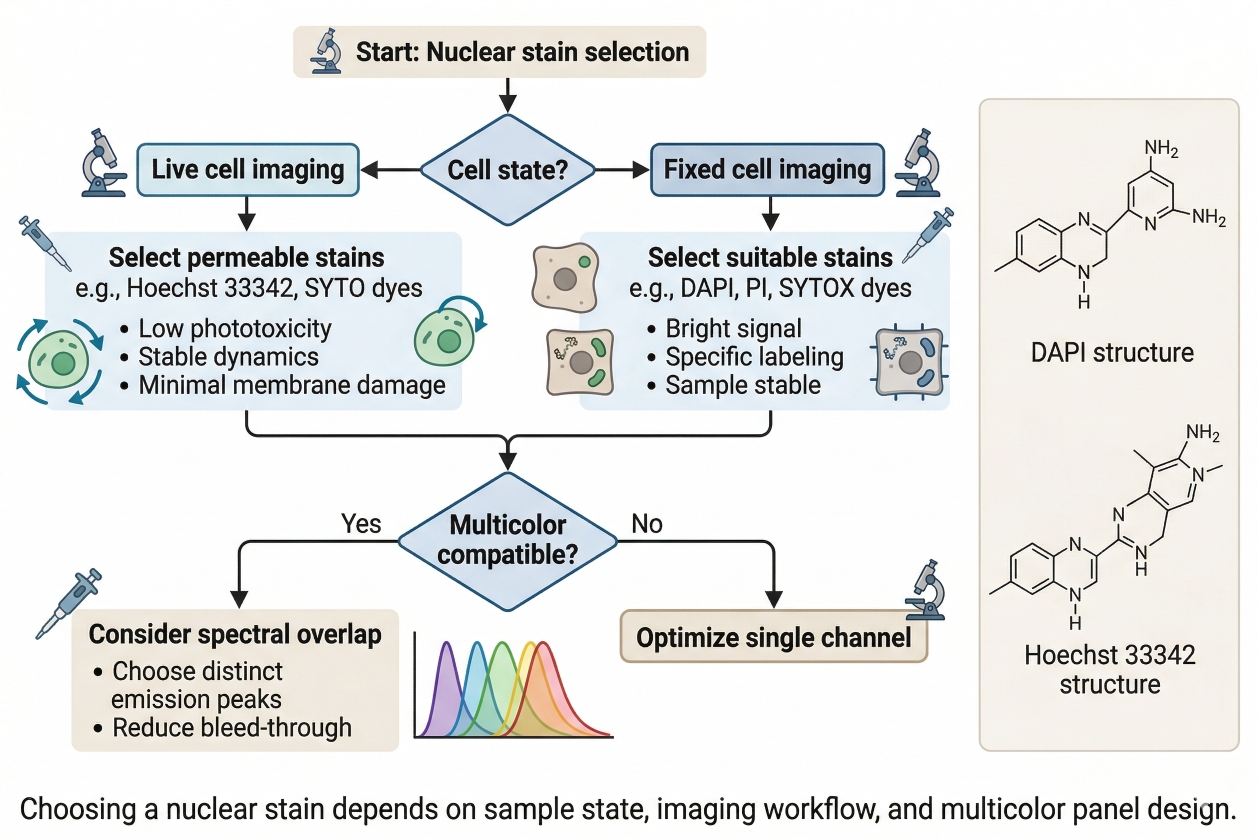 Fig. 2. Choosing a nuclear stain depends on sample state, imaging workflow, and multicolor panel design (BOC Sciences Authorized).
Fig. 2. Choosing a nuclear stain depends on sample state, imaging workflow, and multicolor panel design (BOC Sciences Authorized).
Nuclear Staining Applications in Research Workflows
Nuclear staining is one of the most widely used analytical layers in fluorescence-based cell experiments because it supports far more than simple nucleus visualization. In real research workflows, the nuclear channel often serves as a structural reference, a computational anchor, a morphology-sensitive readout, and a normalization layer that helps make comparisons between treatment groups more reliable. Its value is especially high in settings where large numbers of images, multiple fluorescent channels, or condition-to-condition comparisons must be interpreted consistently. For research teams in pharma, biotech, drug discovery, and CDMO environments, this makes nuclear staining a practical tool for improving both image quality and data usability across a broad range of assay formats.
Nuclear Staining in Fluorescence Microscopy and Cell Imaging
In fluorescence microscopy and general cell imaging workflows, nuclear staining is primarily valuable because it provides one of the clearest and most stable intracellular landmarks in the image. This helps researchers define where cells are located, how many intact cells are present, and how other intracellular or membrane-associated signals should be interpreted spatially. A strong nuclear layer makes it easier to distinguish isolated cells from clustered regions, improves visual orientation within complex fields, and supports more confident interpretation of whether a signal is nuclear, perinuclear, cytoplasmic, or broadly distributed across the cell. In practical microscopy work, this becomes especially important when image quality varies across fields, when cells are heterogeneous in shape or density, or when additional fluorescent channels need a clear spatial anchor to remain biologically interpretable. As a result, nuclear staining often contributes directly to the clarity, reproducibility, and analytical value of microscopy datasets rather than functioning only as a visual enhancement.
Nuclear Staining in High-Content Imaging and Phenotypic Screening
In high-content imaging and phenotypic screening, nuclear staining often moves from being helpful to being foundational because it supports automated segmentation, object recognition, cell counting, and per-cell feature extraction across large datasets. Many image analysis pipelines begin with the nucleus because it is usually easier to detect consistently than the whole-cell boundary, especially in dense or morphologically variable samples. Once nuclei are identified, software can assign surrounding signals to individual cells more reliably, enabling quantitative analysis of treatment response, morphology shifts, intracellular redistribution, and population-level heterogeneity. For discovery and screening groups, this is particularly valuable because the nuclear channel helps transform microscopy from descriptive imaging into scalable assay readout. It allows researchers to compare many compounds, concentrations, or time points with stronger normalization logic, and it reduces the risk that apparent phenotype changes are driven only by differences in cell number or field composition. In this context, the quality of nuclear staining can directly affect segmentation accuracy, downstream quantification, and the confidence of screening decisions.
Nuclear Staining in Multicolor Organelle and Localization Studies
In multicolor organelle and localization studies, nuclear staining is highly valuable because it provides the central positional reference against which other fluorescent signals are interpreted. When researchers combine nuclear, membrane, mitochondrial, lysosomal, ER, Golgi, or target-specific channels in one experiment, the nucleus often becomes the anchor that helps organize the rest of the image spatially. This is important not only for visual clarity, but also for preventing false conclusions about localization or redistribution. A perinuclear signal, for example, carries a different meaning from a truly nuclear signal, and that difference can be hard to judge confidently without a clean nuclear reference layer. In co-localization studies, the nuclear channel can also help define whether apparent overlap is biologically plausible or more likely to be the result of bleed-through, background, or generalized intracellular staining. For research programs studying intracellular trafficking, mechanism of action, or organelle interaction, nuclear staining therefore plays a critical role in preserving spatial logic across the full multicolor panel and improving the interpretability of localization-based conclusions.
Nuclear Staining in Viability-Aware and Morphology-Based Assays
In viability-aware and morphology-based assays, nuclear staining can provide important contextual information that helps researchers separate changes in cell number from changes in cell state or intracellular phenotype. A reduction in total fluorescence from another channel may reflect loss of cells, altered segmentation, nuclear damage, or a real target-specific biological response, and the nuclear layer often helps distinguish among these possibilities. In morphology-driven experiments, nuclear size, condensation, fragmentation, or positional changes may themselves become informative readouts when compared across treated and untreated groups. This makes nuclear staining especially useful in assays where treatment exposure can influence both cell abundance and cellular architecture, such as comparative imaging studies, stress-response assays, and compound-evaluation workflows. For teams working in drug discovery and translational research, the nuclear channel often provides the structural baseline needed to interpret whether an observed phenotype represents true intracellular biology, a broader morphology shift, or a more general effect on cell integrity across the sample.
Choosing between live-cell, fixed-cell, and multicolor nuclear staining strategies?
We can help match nuclear dye selection, channel placement, and workflow logic to your imaging purpose, sample state, and analysis needs.
Common Problems in Nuclear Staining and How to Avoid?
Although nuclear staining is widely used, it is not immune to failure or misinterpretation. Problems often arise not because the dye does not fluoresce, but because the signal is too strong, too weak, poorly contrasted, or interpreted too simplistically. In research workflows that depend on reproducibility and accurate comparative analysis, these problems can have consequences far beyond the nuclear channel itself.
- Overstaining, Oversaturation, and Loss of Nuclear Detail: One common problem is using a nuclear stain at conditions that produce extremely bright but analytically poor images. Oversaturated nuclei can lose internal detail, reduce segmentation quality, and overwhelm nearby channels in a multicolor panel. In such cases, stronger nuclear signal does not improve the assay. It actively reduces interpretability. Optimizing for usable contrast rather than maximum brightness is therefore essential.
- Weak Nuclear Signal and Poor Nucleus-to-Cytoplasm Contrast: A weak nuclear channel can be just as problematic as an overloaded one. If nuclei are not clearly separated from cytoplasmic or background signal, cell counting and segmentation become less reliable, and the nuclear layer loses value as a spatial reference. Weak contrast may reflect an unsuitable dye for the workflow, insufficient access to the nucleus, or imaging conditions that do not support the chosen stain effectively.
- Background Fluorescence and Non-Nuclear Signal Interference: Background fluorescence or off-target signal can compromise the usefulness of a nuclear stain even when nuclei are visible. This is especially important in dense samples and multicolor workflows where non-nuclear interference can make the channel less clean than it first appears. A nuclear stain should therefore be evaluated by how well it defines nuclei relative to the full sample background, not just by whether a fluorescent spot is visible where nuclei should be.
- Misreading Abnormal Nuclear Morphology After Treatment or Stress: Nuclear morphology can change substantially after treatment or sample stress, and these changes are sometimes mistaken for poor staining when they are actually biologically relevant, or vice versa. This is why nuclear channels should be interpreted together with sample condition and the rest of the assay. An irregular nuclear pattern may reflect a real response, but it should not automatically be assumed to do so unless technical causes such as overstaining, unhealthy samples, or imaging artifacts have been considered.
Best Practices for Building Reliable Nuclear Staining Workflows
Reliable nuclear staining rarely comes from choosing a famous dye and using it unchanged across every assay. It comes from aligning stain choice, sample state, imaging conditions, and analytical purpose. The most robust workflows treat the nuclear channel as part of assay design rather than as a last-minute reference layer.
- Start with the Experimental Purpose, Not the Dye Name: The best starting point is to define what the nuclear channel needs to do. Is it mainly for segmentation, morphology comparison, multicolor reference, live-cell observation, or endpoint counting? Once that purpose is clear, stain choice becomes much more rational. Starting from the dye name instead of the assay need often leads to habitual but suboptimal reagent use.
- Validate Concentration, Incubation, and Imaging Settings Systematically: Nuclear staining performance depends strongly on how the dye is used. Concentration, incubation duration, imaging settings, and channel assignment should be validated systematically rather than inherited from unrelated workflows. Small optimization steps can often transform a mediocre nuclear channel into a highly useful analytical layer without changing the stain itself.
- Use Nuclear Staining as a Designed Analytical Layer in Multicolor Experiments: In multicolor experiments, the nuclear stain should be planned deliberately as part of the panel. That includes deciding which spectral channel it should occupy, how bright it needs to be relative to the rest of the panel, and whether it is being used only as a reference or also as a morphology-sensitive readout. This mindset reduces channel conflict and improves the coherence of the overall assay.
- Document Stain Choice and Imaging Logic for Reproducibility: For workflows that need reproducibility across experiments or teams, documenting the reason for stain choice, the working concentration, the sample state, and the imaging logic is as important as documenting the dye name itself. This is particularly relevant in pharma, biotech, and CDMO settings where assays may need to scale, transfer, or support cross-experiment comparisons. A documented nuclear staining strategy is easier to reproduce and much easier to troubleshoot.
Nuclear Staining Solutions at BOC Sciences
BOC Sciences provides comprehensive nuclear staining support to meet diverse fluorescence imaging and cell analysis needs in pharma, biotech, drug discovery, and CDMO research workflows. Whether the goal is live-cell nuclear visualization, fixed-cell endpoint analysis, high-content imaging, multicolor fluorescence panel development, or morphology-based comparative studies, we offer relevant fluorescent dye resources and workflow-oriented support to help research teams build more reliable nuclear staining strategies. Our support is designed to improve nuclear signal clarity, channel compatibility, imaging reproducibility, and analytical consistency across different experimental systems.
Nuclear Fluorescent Dye and Probe Supply
- Provides nucleus-relevant product categories including nuclear fluorescent probes and DNA stains for nuclear visualization, segmentation, cell counting, and morphology analysis.
- Supports a range of fluorescent products suitable for live-cell imaging, fixed-cell staining, endpoint microscopy, and routine fluorescence-based assays.
- Offers dye options that help improve nucleus-to-cytoplasm contrast, signal clarity, and overall imaging consistency.
- Provides access to broader fluorescent dyes and fluorescent probes for customers requiring expanded comparison and selection across different nuclear staining workflows.
Customized Nuclear Staining Strategy Support
- Supports nuclear stain selection based on specific experimental goals, including structural visualization, cell counting, morphology profiling, and image-based comparison.
- Helps align stain choice with workflow requirements such as live-cell compatibility, fixed-cell use, low background, and stable fluorescence performance.
- Provides support for multicolor fluorescence panel design, helping customers choose nuclear stains compatible with membrane, organelle, viability, or target-specific channels.
- Assists customers in building more application-oriented nuclear staining strategies for both routine assays and complex imaging studies.
Application Support for Imaging, Screening, and Cell Analysis
- Supports nuclear staining workflows used in cell imaging, fluorescence imaging, and high-throughput screening, where nuclear signals are essential for segmentation and comparative analysis.
- Provides guidance for applications involving morphology analysis, nucleus-based object recognition, treatment-response comparison, and multichannel image interpretation.
- Supports integration of nuclear staining into broader cell staining workflows and fluorescence-based assay systems.
- Helps research teams use the nuclear channel as an analytical reference layer for counting, normalization, localization, and phenotype assessment.
Expanded Fluorescence Resources and Related Service Support
- Provides related product categories such as NHS esters, hydrazides, azides, alkynes, and click chemistry reagents for customized fluorescent labeling and advanced reagent development.
- Offers related service support including fluorescent protein services, fluorescent nanoparticle services, and fluorescent bead services for broader fluorescence-enabled research programs.
- Supports nuclear staining projects that extend into intracellular imaging, assay development, and multiparameter fluorescence analysis.
- Helps customers move from routine nuclear labeling toward more flexible and scalable fluorescence solutions for screening, mechanism studies, and advanced cell analysis.
Do You Need A Consultation?
BOC Sciences integrates cutting-edge fluorescence technologies to accelerate your research, driving next-generation solutions for drug discovery and diagnostics.
Transform Your Studies with Cutting-Edge Fluorescent Products
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0201 | DAPI dihydrochloride | 28718-90-3 | Bulk Inquiry |
| A16-0222 | YOYO 1 | 143413-85-8 | Bulk Inquiry |
| A16-0223 | YO-PRO 3 | 157199-62-7 | Bulk Inquiry |
| A16-0220 | TOTO 3 | 166196-17-4 | Bulk Inquiry |
| A16-0023 | 7-ethoxy-4-Methylcoumarin | 87-05-8 | Bulk Inquiry |
| A16-0221 | TO-PRO 1 | 157199-59-2 | Bulk Inquiry |
| A19-0041 | Hoechst 33258 | 23491-45-4 | Bulk Inquiry |
| A16-0219 | Toto-1 | 143413-84-7 | Bulk Inquiry |
High-Performance Fluorescent Tools for Your Research
- Ion Fluorescent Probes Indicators for real-time ion concentration imaging.
- Lysosomal Fluorescent Probes Acidic organelle markers for lysosome tracking.
- pH Indicators Fluorescent sensors for intracellular pH monitoring.
- Apoptosis Fluorescent Probes Probes detecting programmed cell death events.
- Nitric Oxide (NO) & Reactive Oxygen Species (ROS) Probes for oxidative stress and signaling detection.
- Mitochondrial Fluorescent Probes Targeted dyes for mitochondrial structure and function.
- Fluorescent Probes Versatile tools for biomolecular and cellular imaging.
- Nuclear Fluorescent Probes DNA-binding dyes for nucleus visualization.
Explore More Topics
Online Inquiry


