![]()
MitoTracker Fluorescent Staining Protocols: Mitochondrial Fluorescent Dye
03-26-2026
MitoTracker is a mitochondrial fluorescent probe with cell permeability that can accumulate in active mitochondria and is widely used for specific fluorescent staining of mitochondria in live cells. Traditional fluorescent dyes such as tetramethylrosamine and rhodamine 123 can also accumulate in active mitochondria; however, they are easily washed out when the mitochondrial membrane potential is lost. Some MitoTracker dyes contain a mildly thiol-reactive chloromethyl moiety, allowing them to be retained within mitochondria even after the loss of membrane potential. This property makes them compatible with aldehyde fixation and suitable for downstream applications such as immunocytochemistry or in situ hybridization.
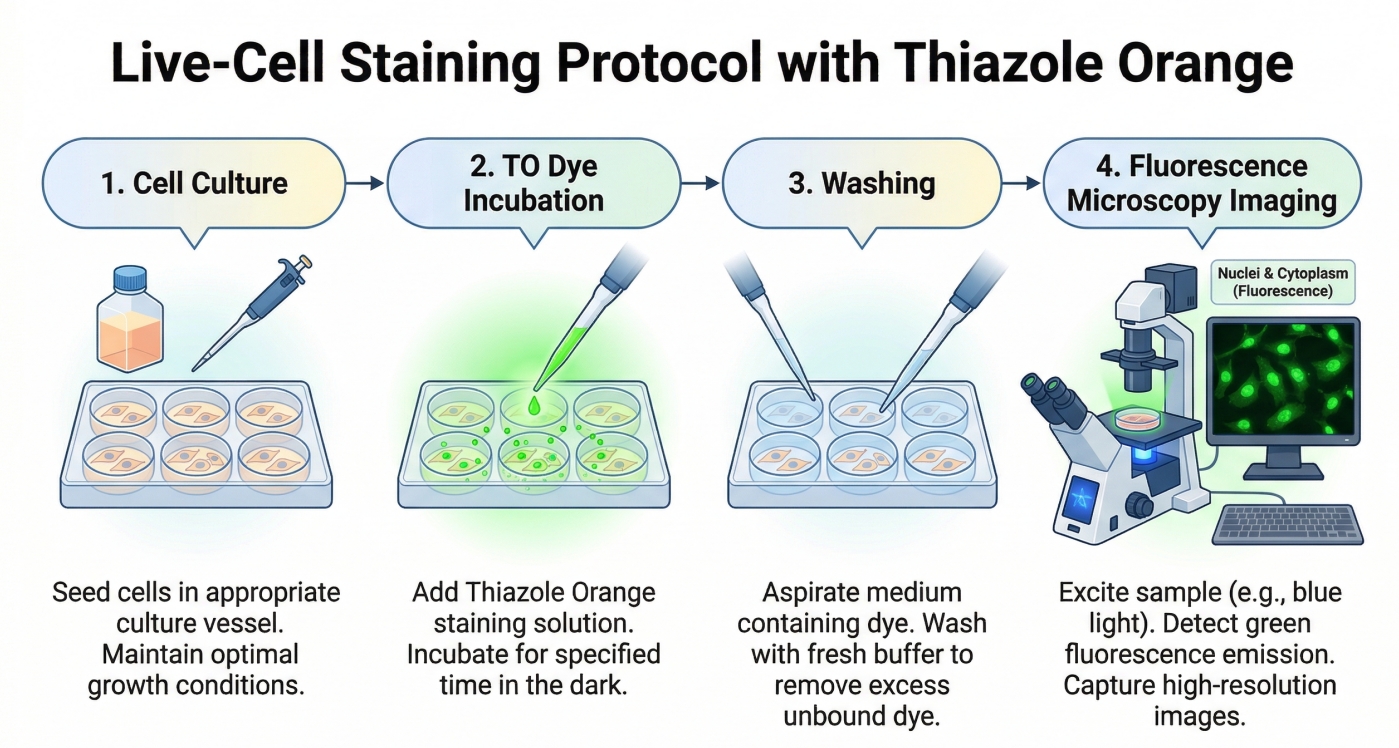
Thiazole Orange (TO) Fluorescent Staining Protocols: Nucleic Acid Fluorescent Dye
03-26-2026
Thiazole Orange (TO) is a highly sensitive fluorescent dye that binds strongly to nucleic acids, producing a pronounced increase in fluorescence upon interaction with DNA or RNA. Its ability to label both single- and double-stranded nucleic acids makes TO an ideal tool for detecting cellular nucleic acid content, performing flow cytometry analyses, and constructing nucleic acid probe systems. With its high brightness and low background, TO enables real-time imaging and accurate nucleic acid detection across a variety of experimental applications.
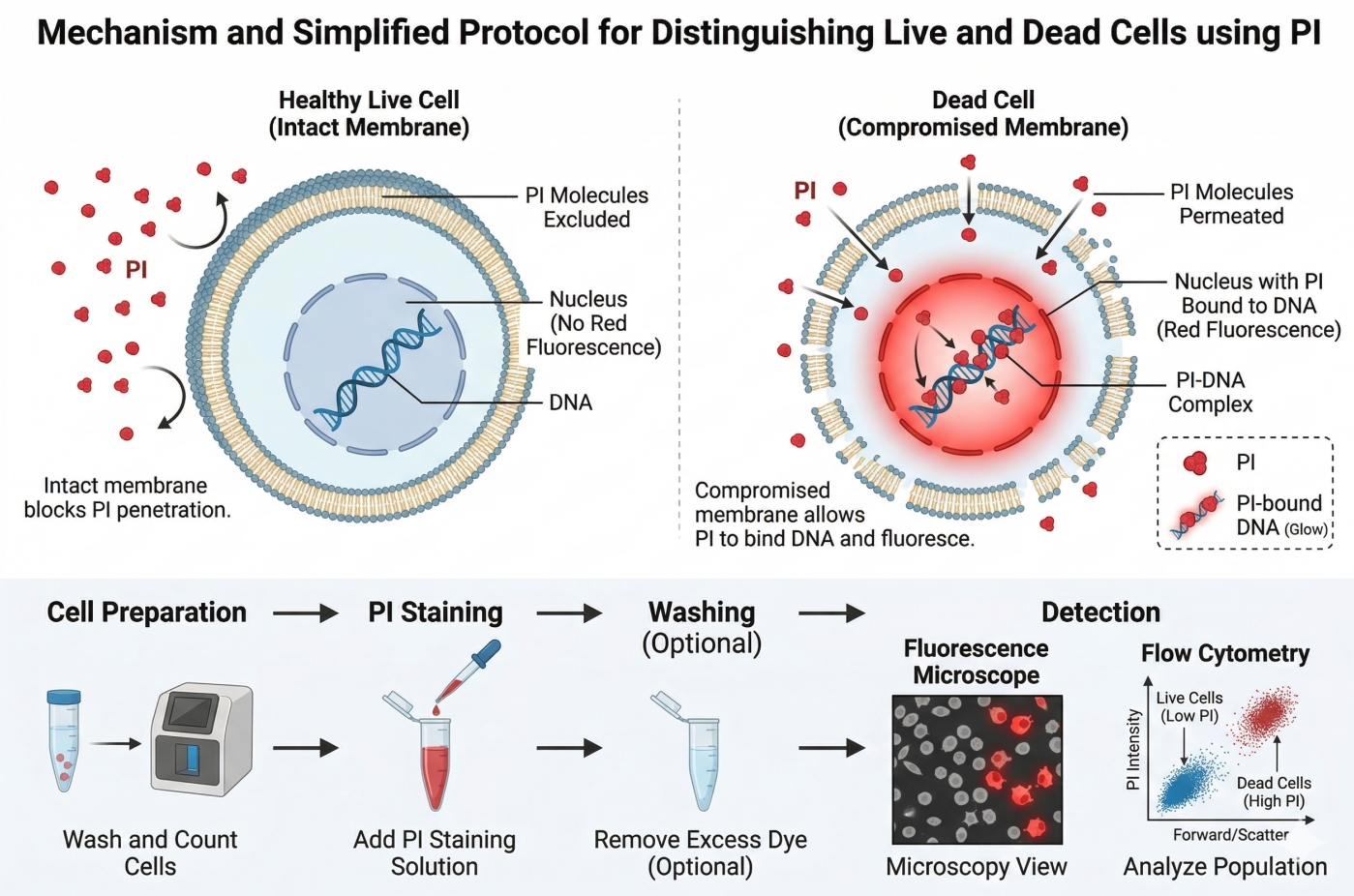
Propidium Iodide (PI) Fluorescent Staining Protocols: Apoptosis Detection Dye
03-26-2026
Propidium Iodide (PI) is a widely used red-fluorescent nucleic acid dye that plays a crucial role in cell biology research, particularly for apoptosis detection and viability assessment. Its unique property of selectively entering cells with compromised membranes makes it an ideal tool for distinguishing live from dead cells in flow cytometry, fluorescence microscopy, and microbial viability studies. Beyond viability assays, PI is frequently combined with other fluorescent probes such as Annexin V, DAPI, and Hoechst to enable multi-parameter analysis of cell death, cell cycle distribution, and DNA content, providing researchers with reliable and quantitative insights into cellular health and function.
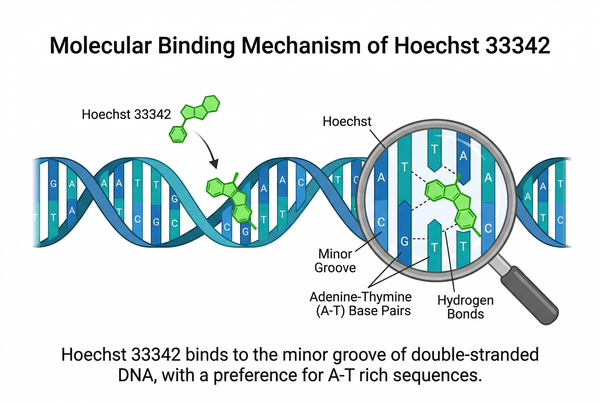
Hoechst 33342 Fluorescent Staining Protocols: Live Cell Imaging Dye
03-26-2026
Hoechst 33342 is a widely used fluorescent dye for staining cell nuclei in both live and fixed cells. Its ability to selectively bind to DNA allows researchers to visualize nuclear morphology, assess cell cycle status, and track cellular dynamics with high specificity. This versatile dye is compatible with various imaging techniques, including confocal and conventional fluorescence microscopy, as well as flow cytometry. In this article, we provide detailed protocols and practical tips for optimizing Hoechst 33342 staining, helping researchers achieve clear, bright nuclear fluorescence while minimizing background signals. Whether studying mammalian cells, microbial samples, or tissue sections, these protocols ensure reliable and reproducible results for accurate cellular analysis.
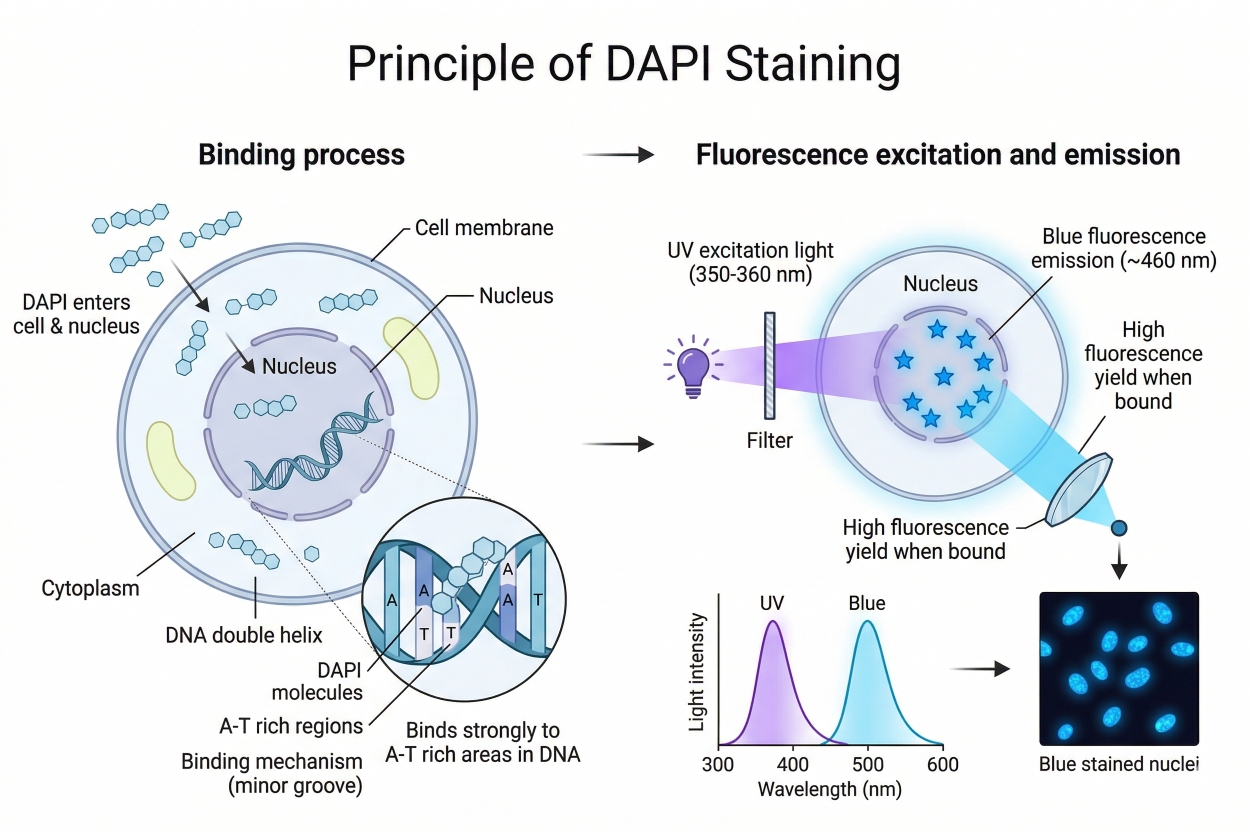
DAPI Fluorescent Staining Protocols
03-26-2026
DAPI is a blue fluorescent dye that binds strongly to DNA, commonly used for nuclear staining. Its blue fluorescence contrasts sharply with other green, yellow, or red fluorescent proteins, making it widely used in multicolor fluorescence applications. DAPI can penetrate cell membranes, allowing staining of both live and fixed cells; however, live-cell staining requires precise control of DAPI concentration, so it is mainly used for fixed-cell staining.
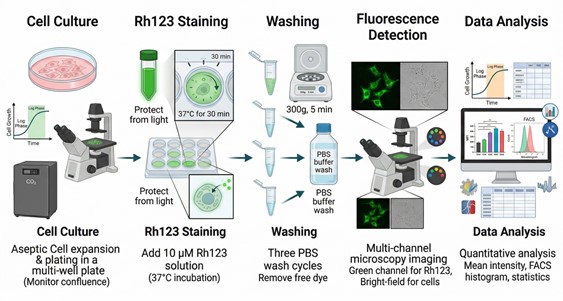
Rhodamine 123 Fluorescent Staining Protocols
03-26-2026
Rhodamine 123 (Rh123) is a widely used fluorescent dye in cell biology research that selectively accumulates in the mitochondria of living cells. This property makes it an essential tool for studying mitochondrial function and assessing cell viability. Rh123 has unique chemical characteristics as a lipophilic cationic dye, allowing it to easily penetrate cell membranes and accumulate within mitochondria driven by the mitochondrial membrane potential. Optically, Rh123 is excited at a wavelength of 488 nm and emits a strong green fluorescence at 530 nm. Due to its high quantum yield, Rh123 produces high-contrast fluorescent signals, making it especially valuable for cellular biology experiments.
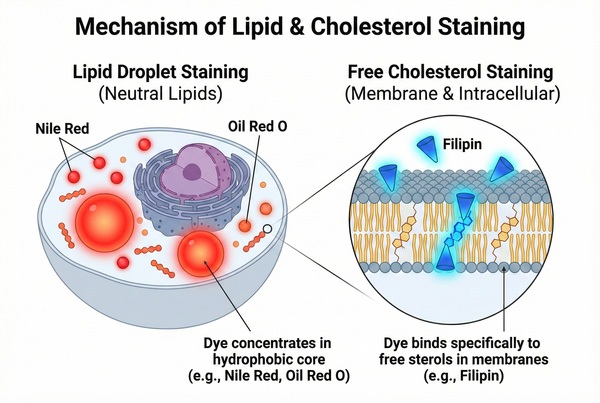
Lipid Staining – Guide to Lipid Droplet & Cholesterol Staining Protocols
02-26-2026
Lipids are a class of organic molecules widely present in living organisms, playing essential roles in cellular structure and function. Lipid droplets and cholesterol represent different forms of lipids with distinct structural and functional characteristics. Lipid droplets serve as the primary storage sites for neutral lipids, including triglycerides and cholesteryl esters. Under pathological conditions, such as organ steatosis, increased intracellular lipid droplets may appear, which can be clearly visualized by lipid staining. Cholesterol is an essential component of eukaryotic cell membranes, maintaining membrane integrity, regulating membrane fluidity, and participating in signal transduction. It plays critical roles in the development and progression of diseases such as atherosclerosis, Niemann-Pick disease type C (NPC), and Alzheimer’s disease.
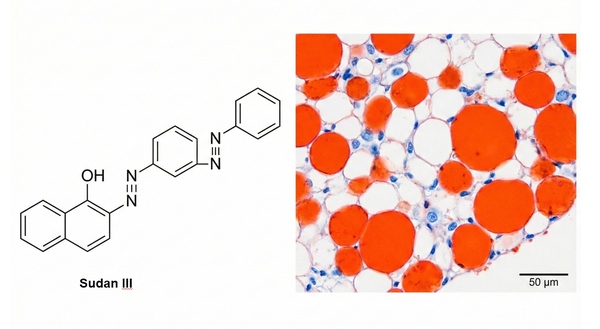
Lipid Staining Protocols: Oil Red O, Nile Red, BODIPY & Filipin Methods Guide
02-26-2026
Lipids are not only essential structural components of cells (such as biological membranes) and major forms of energy storage, but also play irreplaceable roles in key physiological and biochemical processes including cell signaling and molecular transport. Chemically, lipids are primarily esters of fatty acids or derivatives related to these esters. Neutral fats are mainly stored in adipose tissue and exist as oil droplet–like particles suspended within the cytoplasm. In recent years, with the deepening research into metabolic syndrome, obesity, atherosclerosis, non-alcoholic fatty liver disease, and tumor metabolism, the precise localization and quantitative detection of lipid deposition in tissues or cells have become high-frequency demands in pathological diagnosis and basic medical research. The following section provides a comprehensive review of the most representative lipid staining methods currently used in laboratories—covering both time-honored classical histological staining techniques (such as the Sudan dye method and Oil Red O staining) and modern fluorescent probe technologies suitable for high-resolution imaging (such as Nile Red, BODIPY 493/503, and the Filipin method).
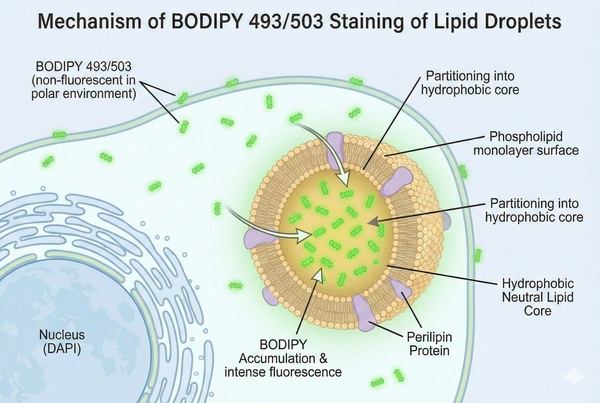
Lipid Staining – Lipid Droplet Green Fluorescence Staining Protocol
02-26-2026
Green fluorescent Bodipy (Boron-dipyrromethene) staining is a highly sensitive fluorescence method commonly used to detect neutral lipids and lipid droplet distribution in cells or tissues. It is suitable for studying lipid dynamics, including lipid metabolism, lipid droplet formation, and lipid distribution analysis. Bodipy 493/503, as a lipophilic fluorescent probe, targets polar lipids and labels neutral lipid content in cells, especially the lipid content of lipid droplets. It is compatible with epifluorescence, confocal, and two-photon microscopy, as well as flow cytometry. With excitation/emission maxima at 493/503 nm, Bodipy can be applied in both live and fixed cells. Its use extends to aqueous systems in applications such as chemical sensors, cellular imaging, anticancer studies, biolabeling, biomedical research, metal ion detection, and photodynamic therapy. Bodipy is also widely applied in biosensing and drug screening to monitor active substances in biological systems, such as lipid peroxides.
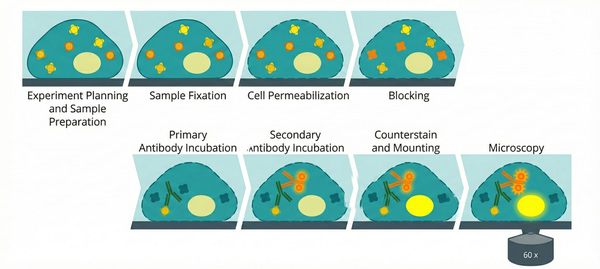
Immunofluorescence - Staining Protocol Guide and Combined Applications
02-26-2026
Immunofluorescence (IF) is an optical imaging sample preparation technique widely used in biomedical and cell biology research. It enables the detection and localization of specific proteins in cells or tissue sections through fluorophore-labeled antibodies. This technique combines the specific recognition of antigen–antibody interactions with fluorescent molecular labeling, allowing researchers to observe the distribution and localization of target proteins in cells or tissues under a fluorescence microscope. When combined with advanced imaging techniques such as super-resolution microscopy or fluorescence lifetime imaging, it can yield unexpectedly intuitive and high-resolution results.
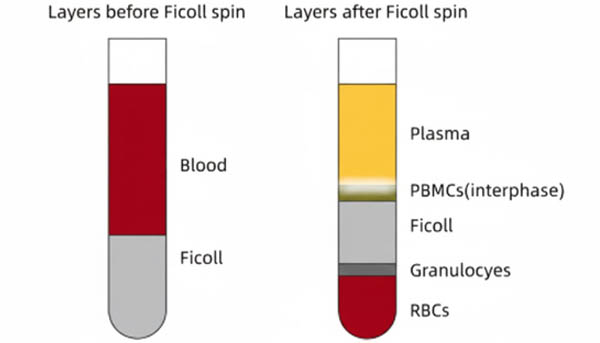
Flow Cytometry – 8 Common Sample Preparation and Intracellular Staining Protocols
01-27-2026
Flow cytometry (FCM), as a precise technology for multi-parameter quantitative analysis of heterogeneous cell populations at the single-cell level, relies heavily on the quality of sample pre-processing for accurate and reliable data. Preparing highly viable, non-aggregated single-cell suspensions with stable antigen expression is the cornerstone for subsequent fluorescence compensation, population gating, and signal analysis. Maintaining cell viability, removing dead cell debris, and excluding doublets directly affect the signal-to-noise ratio (S/N ratio) and the effectiveness of gating strategies. Physical dissociation tailored to tissue mechanics, such as spleen, bone marrow, or thymus, or density gradient centrifugation and red blood cell lysis for peripheral blood, must strictly follow standardized operating procedures (SOPs) to prevent excessive cell damage or enzymatic/epitope loss. For intracellular targets like cytokines and transcription factors, precise fixation and permeabilization are crucial for crossing biological membranes, ensuring antibody access, and maintaining specific binding. This article systematically outlines eight typical single-cell preparation and intracellular staining protocols, providing standardized references for establishing high-quality flow cytometry workflows.
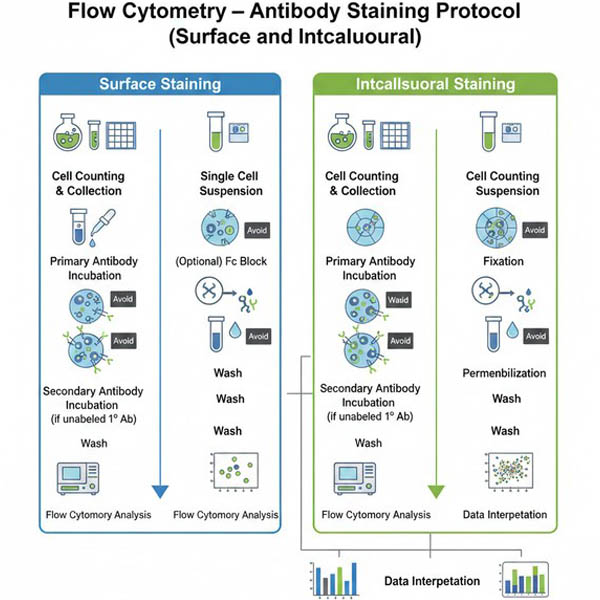
Flow Cytometry – Antibody Staining Protocol (Surface and Intracellular)
01-27-2026
Flow cytometry antibodies are designed for use in flow cytometry experiments. Compared to antibodies used in ELISA, Western blotting (WB), or immunofluorescence (IF), they generally require less complex sample preparation and experimental steps. Flow cytometry antibodies can detect either live or fixed cells and provide quantitative, single-cell, multi-parameter data. They are available as either unlabeled or fluorescently labeled antibodies. Unlabeled antibodies require a corresponding secondary antibody, while fluorescently labeled antibodies carry the fluorophore and do not require a secondary antibody.
Flow Cytometry – Intracellular and Nuclear Factor Staining Protocol
01-27-2026
Staining and detecting intracellular molecules helps identify cell subpopulations and states. Unlike traditional surface antibody staining, flow cytometry analysis of intracellular proteins requires initial fixation to stabilize the cell membrane, followed by permeabilization to allow antibodies to access intracellular antigens. Mass cytometry extends this capability by simultaneously analyzing both surface markers and intracellular or nuclear components across multiple channels, without the need for voltage adjustment or compensation, greatly enhancing detection power.
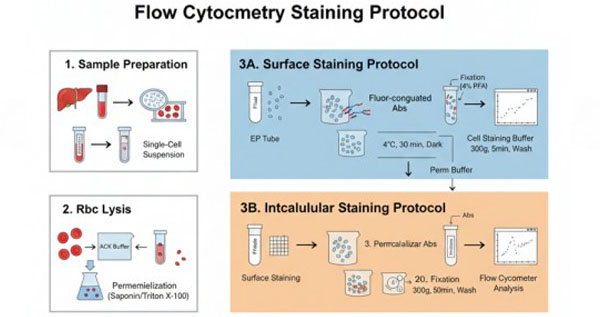
Flow Cytometry - Surface and Intracellular Cell Staining Protocol
01-27-2026
Flow cytometry (FCM) is a single-cell–based quantitative analysis and sorting technology performed using a flow cytometer. Flow cytometry is a high-technology platform developed through the advanced integration of monoclonal antibody and immunocytochemistry techniques, laser technology, and computer science. It enables effective discrimination of heterogeneous cell populations at the single-cell level. Detection targets include, but are not limited to, suspended cells, adherent cells, single-cell suspensions dissociated from solid tissues, and other biological particles.


