Immunofluorescence - Staining Protocol Guide and Combined Applications
Immunofluorescence (IF) is an optical imaging sample preparation technique widely used in biomedical and cell biology research. It enables the detection and localization of specific proteins in cells or tissue sections through fluorophore-labeled antibodies. This technique combines the specific recognition of antigen–antibody interactions with fluorescent molecular labeling, allowing researchers to observe the distribution and localization of target proteins in cells or tissues under a fluorescence microscope. When combined with advanced imaging techniques such as super-resolution microscopy or fluorescence lifetime imaging, it can yield unexpectedly intuitive and high-resolution results.
Basic Principles
1. Antibody Recognition of Target Proteins: A specific antibody (referred to as the primary antibody) recognizes the target protein (antigen).
2. Detection with Fluorescently Labeled Antibody: A fluorescently labeled secondary antibody binds to the primary antibody. The secondary antibody carries a fluorophore that emits light at a specific wavelength under a fluorescence microscope.
3. Fluorescence Microscopy Imaging: The fluorophore is excited by a fluorescence microscope to emit light signals, allowing researchers to observe the expression location and relative abundance of the target protein in the sample.
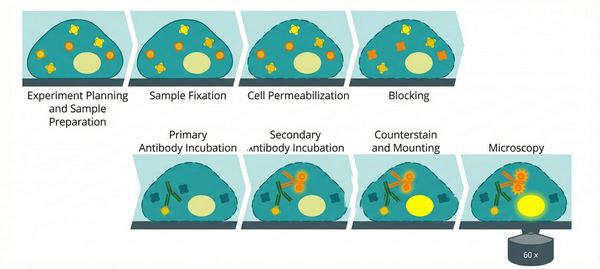 Fig. 1. Schematic illustration of the multi-step immunofluorescence staining protocol (BOC Sciences Authorized).
Fig. 1. Schematic illustration of the multi-step immunofluorescence staining protocol (BOC Sciences Authorized).
Experimental Reagents and Consumables
- Adherent cells or tissue sections
- 4% paraformaldehyde (PFA) fixative
- 0.1%–0.5% Triton X-100 (permeabilization reagent)
- PBS (phosphate-buffered saline)
- Blocking solution (5%–10% normal goat serum or 1%–5% BSA)
- Primary antibody (specific to the target protein)
- Fluorescently labeled secondary antibody (e.g., Alexa Fluor 488, Alexa Fluor 594)
- DAPI or Hoechst (nuclear stains)
- Anti-fade mounting medium (e.g., ProLong Gold Anti-fade Mountant)
- Glass slides and coverslips
- 6-well plates or glass-bottom culture dishes
- Absorbent paper and lens paper
- Various fluorescence microscopes
Experimental Procedure
01 Cell Preparation
(1) Adherent cells: Seed cells onto coverslips or glass-bottom culture dishes and culture until 50%–70% confluence. Ensure that the cells are in good condition and firmly attached to the coverslip surface.
(2) Tissue sections: Prepare tissue sections and place them on glass slides.
02 Fixation
(1) Remove the culture medium and gently wash cells or tissues with PBS 2–3 times.
(2) Add 4% PFA fixative and incubate at room temperature for 10–20 minutes. PFA crosslinks intracellular proteins, thereby fixing their positions and preventing degradation.
(3) After fixation, immediately wash with PBS 3 times, 5 minutes each time, to thoroughly remove residual fixative.
✔ Purpose: To fix cells or tissues, preserve protein localization and subcellular structures, and prevent degradation.
03 Permeabilization
(1) Permeabilize cells with 0.1%–0.5% Triton X-100 solution and incubate at room temperature for 10 minutes. Triton X-100 dissolves the cell membrane, allowing antibodies to penetrate into the cell interior.
(2) After permeabilization, wash with PBS 3 times, 5 minutes each time.
✔ Purpose: To allow antibodies to enter cells or tissues and detect intracellular proteins.
04 Blocking
(1) Add 5%–10% normal goat serum or 1%–5% BSA (bovine serum albumin) solution and incubate at room temperature for 30–60 minutes.
(2) No washing is required; proceed directly to the next step.
✔ Purpose: To block nonspecific binding sites, reduce background staining, and ensure specific recognition of the target antigen by the antibody.
05 Primary Antibody Incubation
(1) Dilute the primary antibody to an appropriate concentration (e.g., 1:100–1:500) according to the manufacturer's instructions, using blocking solution (such as PBS containing BSA) as the diluent.
(2) Add the diluted primary antibody solution to the cells or tissue samples and incubate overnight at 4°C (or 1–2 hours at room temperature). Protect from light during operation to avoid fluorescence quenching.
(3) After incubation, wash with PBS 3 times, 5 minutes each time, to remove unbound antibodies.
✔ Purpose: The primary antibody specifically binds to the target protein, labeling the specific protein within cells or tissues.
06 Secondary Antibody Incubation
(1) Dilute the fluorescently labeled secondary antibody according to the recommended concentration (e.g., 1:500–1:1000) and protect from light.
(2) Add the diluted secondary antibody solution and incubate at room temperature for 1 hour in the dark.
(3) After incubation, wash with PBS 3 times, 5 minutes each time, to remove unbound secondary antibodies.
✔ Purpose: The fluorescently labeled secondary antibody binds to the primary antibody, forming an antigen–primary antibody–secondary antibody complex. The fluorophore on the secondary antibody is used for subsequent fluorescence microscopy observation.
07 Counterstaining (Optional)
(1) During the final PBS wash, add DAPI or Hoechst nuclear stain at a concentration of 1 μg/mL and incubate at room temperature for 5–10 minutes, protected from light.
(2) Wash again with PBS 2–3 times, 5 minutes each time.
✔ Purpose: To stain the cell nucleus with DAPI or Hoechst, providing a structural reference for the cells.
08 Mounting
(1) Add an appropriate amount of anti-fade mounting medium (e.g., ProLong Gold Anti-fade Mountant) onto the coverslip.
(2) Carefully place the coverslip to avoid air bubbles.
(3) After mounting, samples can be stored at low temperature in the dark. It is recommended to perform microscopy observation as soon as possible.
✔ Purpose: To protect fluorescently stained samples, prevent fluorescence quenching, and provide optically suitable samples for microscopy observation.
09 Fluorescence Microscopy Imaging
(1) Use appropriate excitation light and filter sets under a fluorescence microscope to observe the labeled fluorescent signals.
(2) If multiple staining is performed, ensure the correct filter sets are selected according to different fluorophores (such as Alexa Fluor 488 and Alexa Fluor 594) and observe different fluorescence signals sequentially.
(3) Capture images and save data.
✔ Purpose: To observe the target proteins in the sample or evaluate staining quality using a fluorescence microscope.
If staining quality is confirmed to be satisfactory, advanced imaging systems such as laser scanning confocal microscopes or super-resolution microscopes may be selected for higher-level imaging.
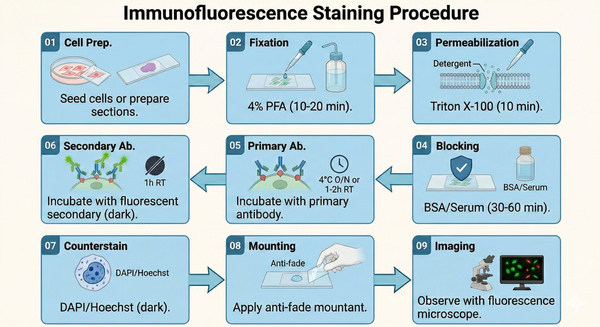 Fig. 2. Schematic flowchart detailing the complete immunofluorescence staining experimental procedure (BOC Sciences Authorized).
Fig. 2. Schematic flowchart detailing the complete immunofluorescence staining experimental procedure (BOC Sciences Authorized).
Precautions
- Protect from Light: Fluorescently labeled antibodies gradually lose activity upon light exposure. Therefore, protect samples from light during incubation and washing steps, especially after adding the secondary antibody.
- Optimize Antibody Concentrations: Before formal experiments, optimize the concentrations of primary and secondary antibodies through preliminary testing. Excessive antibody concentration may cause background staining, while insufficient concentration may result in weak signals.
- Thorough Washing: Ensure washing steps are thorough to prevent residual unbound antibodies from causing nonspecific background signals. If necessary, surfactants such as Triton or Tween-20 may be added to PBS for washing.
- Mounting Medium Selection: Using an anti-fade mounting medium can effectively prolong sample preservation time and fluorescence signal stability.
- Multiplex Staining: For multiplex staining (labeling multiple different target proteins), select fluorescently labeled antibodies from different host species and with different wavelengths, and ensure there is no cross-reactivity between antibodies.
Common Fluorophores for Immunofluorescence Staining
Different fluorophores have different excitation wavelengths, emission wavelengths, and colors. Selection should be based on the filter configuration of the fluorescence microscope. Common types are as follows:
| Fluorophore | Excitation (nm) | Emission (nm) | Fluorescence Color | Typical Applications |
|---|---|---|---|---|
| DAPI | 358 | 461 | Blue | Classic DNA nuclear stain for fixed cells and tissue sections |
| Hoechst 33258 | 352 | 461 | Blue | Nuclear staining for fixed cells with low background |
| Hoechst 33342 | 350 | 461 | Blue | Cell-permeable nuclear stain for live-cell imaging |
| Alexa Fluor 405 | 401 | 421 | Violet-Blue | Compatible with DAPI channel for multiplex staining |
| FITC | 494 | 520 | Green | Routine detection, cost-effective option |
| Alexa Fluor 488 | 495 | 519 | Green | High brightness and photostability, suitable for weak signal detection |
| Cy2 | 492 | 510 | Green | Improved photostability compared to FITC |
| GFP | 488 | 509 | Green | Commonly used for fusion protein localization |
| Cy3 | 550 | 570 | Orange-Red | Stable signal, widely used in standard immunofluorescence |
| Alexa Fluor 555 | 555 | 565 | Orange-Red | Brighter alternative to Cy3 |
| Alexa Fluor 568 | 578 | 603 | Red | Suitable for tissue section staining |
| TRITC | 550 | 570 | Red | Classic red-channel fluorophore |
| Alexa Fluor 594 | 590 | 617 | Red | Compatible with green channels for dual staining |
| Texas Red | 595 | 615 | Red | Commonly used in tissue imaging |
| Cy5 | 650 | 670 | Far-Red | Low background, ideal for multiplex experiments |
| Alexa Fluor 647 | 650 | 668 | Far-Red | High sensitivity detection for flow cytometry and confocal imaging |
| Alexa Fluor 680 | 679 | 702 | Near-Infrared | Deep tissue imaging applications |
| Alexa Fluor 750 | 749 | 775 | Near-Infrared | In vivo imaging applications |
Typical Application Scenarios
- Basic Research: Localization of cytoskeletal proteins (such as tubulin and actin), detection of cell surface receptor expression, and tracking the intracellular distribution of viral particles. For example, in studies of cell migration mechanisms, researchers use single staining with an anti-actin antibody labeled with Alexa Fluor 488 to clearly visualize the dynamic distribution of actin at the leading edge of migrating cells, providing intuitive evidence for elucidating the molecular basis of cell migration.
- Clinical Diagnosis: Detection of autoimmune diseases (such as anti-nuclear antibody testing, in which fluorescently labeled anti-nuclear antibodies are used to observe specific fluorescence in the nucleus), and rapid screening of tumor markers (such as small cell lung cancer markers in cytology smears). In breast cancer diagnosis, single staining with a FITC-labeled anti-HER2 antibody enables rapid assessment of HER2 protein expression on the surface of tumor cells, providing critical information for precise breast cancer subtyping and the development of targeted therapy strategies.
- Pathological Section Observation: Identification of specific cell types in tissue sections (such as recognizing epithelial cells using fluorescently labeled anti-keratin antibodies). In dermatopathological diagnosis, staining skin tissue sections with a Cy3-labeled anti-keratin antibody clearly distinguishes epithelial cell layers from other tissue components, assisting in the diagnosis of skin tumors, inflammation, and other diseases.
Common Combined Staining Strategies
In practical experiments, single staining is often combined with non-specific fluorescent labeling to simultaneously observe the target antigen together with cellular structures or specific substances. The following are eight high-frequency combined staining strategies, described in detail by staining type:
01 DAPI Staining
- Core Function: Specifically labels the cell nucleus (by binding to DNA) and emits blue fluorescence;
- Rationale for Combination with Single Staining: Single staining localizes the target antigen (e.g., using a green FITC-labeled antibody), while DAPI labels the nucleus, allowing rapid determination of whether the antigen is distributed in the nucleus, perinuclear region, or cytoplasm;
- Typical Application Scenarios: Observing whether transcription factors (such as NF-κB) translocate from the cytoplasm to the nucleus, or analyzing the spatial relationship between tumor cell markers and the nucleus to assist in evaluating cellular functional status.
02 Nile Red Staining
- Core Function: Specifically binds to lipids (including neutral lipids and phospholipids) and emits red or yellow fluorescence;
- Rationale for Combination with Single Staining: Single staining detects lipid-related proteins (such as adipogenesis-related proteins), while Nile Red labels intracellular lipid droplets, enabling direct observation of co-localization between the target protein and lipids;
- Typical Application Scenarios: In adipocyte differentiation studies, observing the relationship between lipid droplet formation and adipocyte protein expression; or in liver disease research, detecting lipid deposition in liver tissue and correlating protein expression with abnormal lipid metabolism.
03 FJB Staining (Fluoro-Jade B Staining)
- Core Function: Specifically labels degenerating or necrotic neurons (by binding to the cytoplasm and processes of damaged neurons) and emits green fluorescence;
- Rationale for Combination with Single Staining: Single staining detects neuron-specific proteins (such as NeuN, a marker of mature neurons), while FJB labels injured neurons, clearly distinguishing normal neurons from pathological neurons;
- Typical Application Scenarios: In cerebral ischemia models, observing neuronal injury regions and changes in specific protein expression; or in neurodegenerative diseases such as Alzheimer's disease, analyzing the association between neuronal necrosis and abnormal protein expression in diseased brain tissue.
04 WGA Staining (Wheat Germ Agglutinin Staining)
- Core Function: Specifically binds to glycoproteins on the cell membrane surface (such as N-acetylglucosamine and sialic acid), commonly labeled with fluorophores such as Alexa Fluor 594 to emit red fluorescence;
- Rationale for Combination with Single Staining: Single staining detects membrane proteins (such as receptors or channel proteins), while WGA outlines the cell membrane, clarifying whether the target protein is localized on the membrane;
- Typical Application Scenarios: Observing the distribution of epidermal growth factor receptor (EGFR) on the cell membrane, or verifying membrane localization of cell junction proteins (such as tight junction proteins), supporting studies of cell signaling or intercellular communication.
05 ROS Staining (Reactive Oxygen Species Staining, e.g., DCFH-DA Probe)
- Core Function: Specifically detects intracellular reactive oxygen species (ROS); the probe emits green fluorescence after oxidation by ROS;
- Rationale for Combination with Single Staining: Single staining detects oxidative stress-related proteins (such as Nrf2, a key protein in the antioxidant stress pathway), while ROS staining indicates cellular oxidative stress levels, enabling correlation between target protein expression and oxidative status;
- Typical Application Scenarios: In drug toxicity evaluation, observing the relationship between drug-induced ROS elevation and stress protein expression; or in inflammation models, analyzing the association between oxidative stress and protein expression to investigate inflammatory mechanisms.
06 Phalloidin Staining
- Core Function: Specifically binds to actin in the cytoskeleton (F-actin), commonly labeled with Alexa Fluor 594 (red) or FITC (green);
- Rationale for Combination with Single Staining: Single staining detects cytoskeleton-related proteins (such as myosin), while phalloidin labels actin filaments, allowing observation of co-localization and analysis of the target protein's influence on cytoskeletal structure;
- Typical Application Scenarios: In cell migration research, observing the distribution relationship between migration-related proteins and the actin cytoskeleton to elucidate molecular mechanisms of migration; or in cell morphology studies, analyzing the relationship between morphological changes and cytoskeletal protein expression.
07 Hoechst Staining
- Core Function: Specifically binds to nuclear DNA (especially A–T-rich regions) and emits blue fluorescence (excitation wavelength 350 nm, emission wavelength 460 nm);
- Rationale for Combination with Single Staining: Functionally similar to DAPI, it serves as an alternative nuclear marker. When combined with single staining (e.g., antigen labeled with red Cy3), it clearly distinguishes the nucleus from the target antigen;
- Typical Application Scenarios: In live-cell experiments, Hoechst has low cytotoxicity and is suitable for long-term observation of nuclear changes; or in combined flow cytometry and fluorescence imaging experiments, simultaneously labeling nuclei and antigens to assist in cell sorting and localization analysis.
08 PI Staining (Propidium Iodide Staining)
- Core Function: Penetrates only cells with compromised membranes (dead cells or late apoptotic cells); after binding to DNA, it emits red fluorescence and specifically labels dead cells;
- Rationale for Combination with Single Staining: Single staining detects apoptosis-related proteins (such as Caspase-3, a key apoptosis protein), while PI staining distinguishes live and dead cells, clarifying the association between target protein expression and cell viability;
- Typical Application Scenarios: In apoptosis experiments, observing the overlap ratio between apoptotic protein expression and PI-positive dead cells to determine whether the protein participates in apoptosis; or in drug screening, analyzing the relationship between drug-induced cytotoxicity and target protein expression to assist in evaluating drug efficacy.
Precautions for Combined Operations:
- Staining Order: For fixed samples, it is recommended to complete single staining first (primary antibody incubation + washing), followed by combined staining (such as DAPI, Hoechst, PI, etc.), incubating at room temperature for 5–10 minutes before washing and mounting. For live-cell staining (such as Hoechst or ROS probes), perform combined staining first, then fix the sample for single staining to avoid fixation affecting probe activity;
- Fluorophore Selection: Strictly avoid spectral overlap (e.g., if single staining uses green FITC, ROS staining or FJB staining should be switched to red or blue). Prefer the three-color combination blue (Hoechst/DAPI) + green (FITC) + red (Cy3/PI);
- Special Reagent Handling: ROS probes (such as DCFH-DA) should be diluted in serum-free medium and incubated entirely protected from light; phalloidin staining should be performed after permeabilization to ensure probe entry and binding to F-actin; PI staining requires careful discrimination of live/dead cells and should not be mixed with live-cell probes (such as Hoechst) to avoid signal interference;
- Concentration Control: Combined staining reagents should be diluted according to instructions (e.g., ROS probes commonly 1:1000, phalloidin commonly 1:200, Hoechst commonly 1:2000) to avoid excessive fluorescence intensity that may obscure target antigen signals.
Fluorescent Dyes Recommended for Your Research Project
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0201 | DAPI dihydrochloride | 28718-90-3 | Bulk Inquiry |
| A19-0101 | Propidium Iodide | 25535-16-4 | Bulk Inquiry |
| A19-0060 | Hoechst 34580 | 23555-00-2 | Bulk Inquiry |
| A19-0034 | DAPI dilactate | 28718-91-4 | Bulk Inquiry |
| 407628-15-3 | Alexa Fluor 680 NHS ester | 407628-15-3 | Bulk Inquiry |
| 1374019-99-4 | Alexa Fluor488 NHS | 1374019-99-4 | Bulk Inquiry |
| R01-0451 | AF 488 TFP ester | 2133404-55-2 | Bulk Inquiry |
| R01-0469 | AF 647 NHS ester | 1620475-28-6 | Bulk Inquiry |
| R01-0039 | AF430 NHS ester | 467233-94-9 | Bulk Inquiry |
| A03-0015 | 6-Carboxy-H2DCFDA | 247044-02-6 | Bulk Inquiry |
Corporate Profile
BOC Sciences serves global research and industry clients, specializing in the development, production, and supply of fluorescent products and related technical services. Leveraging a mature chemical synthesis and functional materials platform, the company offers a comprehensive portfolio including fluorescent dyes, labeled antibodies, molecular and cellular imaging probes, and in vivo fluorescent tracers, broadly applied in life sciences research, biomedical imaging, diagnostics, and translational studies.
As a dedicated hub for fluorescence solutions, BOC Sciences focuses on high-performance probes and innovative labeling technologies. Our offerings include conventional dyes (FITC, TRITC, Cy series, Alexa Fluor series), near-infrared probes, environment-responsive probes, and targeted fluorescent molecules. We also provide custom services such as structural modification, spectral tuning, conjugation labeling, and tailored synthesis to meet diverse experimental requirements.
Our R&D team, composed of experienced chemists and biologists, is supported by advanced synthesis, purification, and analytical instrumentation. A rigorous quality control system ensures each product meets high standards for purity, stability, and optical performance. BOC Sciences' fluorescent reagents and services are trusted by universities, research institutions, and biopharmaceutical companies worldwide for cell imaging, protein localization, molecular tracking, mechanistic studies, and method development, with validation in numerous peer-reviewed publications. We are committed to delivering reliable tools and expert support, empowering researchers to accelerate discovery and achieve reproducible, high-quality fluorescence imaging results.
Custom Fluorescent Solutions Designed for Your Experiments
- DNA StainingPrecise fluorescent dyes for clear DNA visualization and analysis.
- Lipid StainingFluorescent solutions for effective lipid structure imaging.
- Cell StainingAdvanced staining for detailed cell morphology and analysis.
- Protein StainingHigh-quality staining for accurate protein detection and imaging.
- Bacteria ImagingFluorescent solutions to visualize bacterial structures and activity.
- Cell ImagingVisualize and analyze live or fixed cells using advanced fluorescence.
- Molecular ImagingCutting-edge fluorescent solutions for deep molecular analysis.
- Fluorescence ImagingHigh-resolution imaging solutions for detailed fluorescence studies.
- BioconjugationCustom bioconjugation services for protein, peptide, and dye linking.
- Drug DeliveryTailored fluorescent solutions for efficient drug delivery research.
- Molecular DiagnosticsFluorescent probes and markers for precise molecular diagnostics.
- Flow CytometryFluorescent dyes and reagents for enhanced flow cytometry analysis.
High-Performance Fluorescent Tools for Your Research
- Nitric Oxide (NO) & Reactive Oxygen Species (ROS) Probes for oxidative stress and signaling detection.
- Calcium, Chloride and Other indicators Fluorescent indicators for intracellular ion flux monitoring.
- Apoptosis Fluorescent Probes Probes detecting programmed cell death events.
- Fluorescent Probes Versatile tools for biomolecular and cellular imaging.
- Cytoskeleton Fluorescent Probes Probes for actin and microtubule visualization.
- Nuclear Fluorescent Probes DNA-binding dyes for nucleus visualization.
- Lysosomal Fluorescent Probes Acidic organelle markers for lysosome tracking.
- Golgi Fluorescent Probes Targeted probes for Golgi apparatus visualization.
Explore More Topics
Online Inquiry


