Lipid Staining – Lipid Droplet Green Fluorescence Staining Protocol
Green fluorescent Bodipy (Boron-dipyrromethene) staining is a highly sensitive fluorescence method commonly used to detect neutral lipids and lipid droplet distribution in cells or tissues. It is suitable for studying lipid dynamics, including lipid metabolism, lipid droplet formation, and lipid distribution analysis. Bodipy 493/503, as a lipophilic fluorescent probe, targets polar lipids and labels neutral lipid content in cells, especially the lipid content of lipid droplets. It is compatible with epifluorescence, confocal, and two-photon microscopy, as well as flow cytometry. With excitation/emission maxima at 493/503 nm, Bodipy can be applied in both live and fixed cells. Its use extends to aqueous systems in applications such as chemical sensors, cellular imaging, anticancer studies, biolabeling, biomedical research, metal ion detection, and photodynamic therapy. Bodipy is also widely applied in biosensing and drug screening to monitor active substances in biological systems, such as lipid peroxides.
Principle of BODIPY Staining
The high-efficiency and specific staining of lipid droplets by Bodipy dyes relies on their unique chemical structure and photophysical properties. The Bodipy core consists of pyrrole rings chelated with boron and fluorine atoms, forming a rigid planar scaffold that is electrically neutral and highly hydrophobic, giving it strong lipophilicity. Due to its small molecular weight and neutrality, Bodipy easily penetrates cell membranes via passive diffusion, quickly entering live or fixed cells. Once inside, based on the principle of "like dissolves like," Bodipy naturally excludes the aqueous cytoplasm and accumulates in the highly hydrophobic neutral lipid core of lipid droplets (e.g., triglycerides), producing a chemical-extraction-like targeted localization effect. Additionally, its fluorescence is highly environment-dependent: in aqueous solutions, molecules tend to aggregate and self-quench, resulting in very low background; in the nonpolar environment of lipid droplets, the molecules exist in a monomeric state and emit strong, specific fluorescence under excitation. Structural modifications, such as symmetry changes or side-chain modifications, can also adjust the emission wavelength to meet imaging requirements for various lipid dynamic studies.
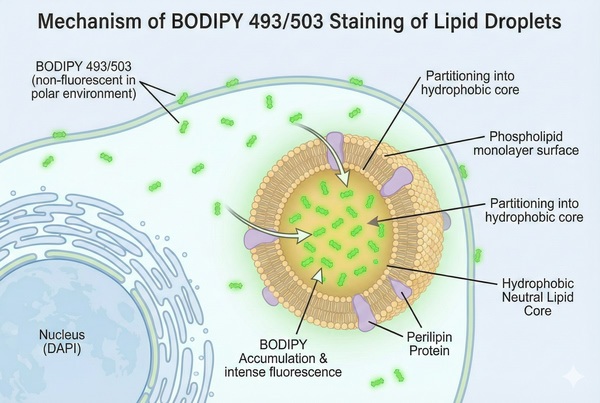 Fig. 1. Light schematic detailing BODIPY accumulation in cellular lipid droplets (BOC Sciences Authorized).
Fig. 1. Light schematic detailing BODIPY accumulation in cellular lipid droplets (BOC Sciences Authorized).
Features of BODIPY Staining
- High fluorescence quantum yield: Produces strong fluorescent signals under excitation, allowing clear imaging.
- Good photostability: Maintains fluorescence intensity under prolonged excitation, suitable for long-term observation experiments.
- Low cytotoxicity: Can label both live and fixed cells with minimal impact on cellular function.
- Adjustable wavelength: Fluorescence emission can be tuned through chemical modifications to meet different experimental needs.
Procedure of Lipid Droplet Staining
- Cell preparation: Culture cells to the appropriate density and seed on glass slides or culture dishes.
- Cell fixation: Fix cells on slides using 4% acetaldehyde or formaldehyde solution.
- Permeabilization: Treat the cell membrane with 0.1% Triton X-100 or other nonionic surfactants to enhance dye permeability.
- Staining solution preparation: Prepare an appropriate amount of Bodipy lipid droplet staining solution, either freshly made or commercially purchased.
- Staining: Add Bodipy solution to the fixed cells and incubate for 15–30 min to ensure thorough cellular uptake.
- Washing: Rinse cells with PBS or another buffer to remove unbound dye and impurities.
- Mounting: Seal slides or culture dishes with a suitable mounting medium to protect and stabilize the samples.
- Imaging: Observe and capture images using an appropriate fluorescence microscope or imaging system.
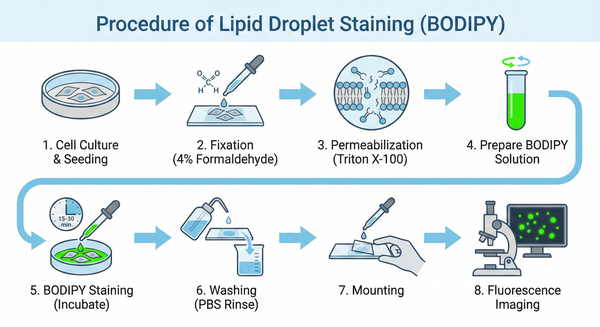 Fig. 2. Schematic protocol for BODIPY staining of cellular lipid droplets (BOC Sciences Authorized).
Fig. 2. Schematic protocol for BODIPY staining of cellular lipid droplets (BOC Sciences Authorized).
Example: BODIPY 493/503 Staining of Suspension Cells
1. Centrifuge at 1000 g for 3–5 min at 4°C, discard the supernatant, wash twice with PBS (5 min each), and adjust cell density to 1×10⁶/mL.
2. Add 1 mL of working solution and incubate at room temperature for 5–30 min.
3. Centrifuge at 400 g for 3–4 min at 4°C, discard supernatant.
4. Wash twice with PBS (5 min each).
5. Resuspend cells in serum-free culture medium or PBS for fluorescence microscopy or flow cytometry observation.
Tissue Staining:
1. Frozen section fixation: Remove frozen sections from the freezer, thaw, air-dry, fix with pure methanol for 10 min, allow slight drying, place in oven for 5 min, then wash 3 times in PBS (pH 7.4) on a shaking platform, 5 min each.
2. Staining: After slight drying, circle tissue edges with a hydrophobic pen to prevent antibody leakage, apply Bodipy solution at 1:1000 (adjustable per pre-experiment), incubate in a dark 37°C incubator for 30 min.
3. DAPI counterstaining: Wash slides 3 times with PBS on a shaking platform (5 min each). After slight drying, add DAPI solution within the circle, incubate at room temperature in the dark for 10 min.
4. Mounting: Wash 3 times with PBS, let dry slightly, then mount with anti-fade mounting medium.
5. Imaging: Observe and capture images under an inverted fluorescence microscope (UV excitation 330–380 nm, emission 420 nm; FITC green excitation 465–495 nm, emission 515–555 nm; Cy3 red excitation 510–560 nm, emission 590 nm).
Common Issues and Recommendations
1. Poor Staining
(1) Dye concentration issues: Excessively high concentrations can cause strong background fluorescence and uneven staining, while too low concentrations result in weak fluorescence signals. Concentration should be optimized according to the specific experiment. For intracellular lipid droplet staining, Bodipy 493/503 is typically used at 2–5 μM.
(2) Improper incubation time or temperature: Short incubation or low temperature may lead to insufficient binding of the dye to intracellular components; excessive time or high temperature can affect cell viability and staining quality. Generally, incubate at room temperature or 37°C for 5–30 min, with adjustments based on cell type and dye used.
2. Abnormal Cell Morphology
Cell handling and fixation, as well as improper storage or culture conditions, can affect cell function and morphology, influencing post-staining appearance. Ensure proper cell handling, use appropriate fixatives and incubation times, and optimize culture conditions to maintain healthy cells.
3. Weak or Unstable Fluorescence Signals
(1) Photobleaching: Prolonged or high-intensity excitation light can reduce fluorescence. Reduce light intensity and exposure time or use anti-photobleaching reagents.
(2) Dye degradation: Bodipy dyes are sensitive to light, temperature, and chemical environment. Improper storage or use can cause degradation. Store and handle as instructed, prepare fresh solutions, and avoid exposure to strong light or high temperatures.
4. Non-specific Staining
(1) Interaction with intracellular components: The dye may interact with other cell components, causing non-specific staining. Adjust dye concentration, incubation time, or conditions, or use alternative specific staining methods for verification.
(2) Cell permeability issues: Poor permeability may prevent sufficient dye entry. Increasing membrane permeability, for example with low-concentration surfactants or brief sonication, can improve staining, while monitoring effects on cell viability.
5. Dye Hydrolysis
Some Bodipy dyes in succinimidyl ester form are prone to hydrolysis in water, reducing staining efficiency. Ensure dyes are fully dissolved and not hydrolyzed before use. Prepare fresh solutions and store unused dye according to instructions.
6. Reagent Storage and Handling
Bodipy dyes should be stored at −20°C or lower, frozen, dry, and protected from light. Prepare fresh solutions when needed, avoid repeated freeze-thaw cycles, and store aliquots for single-use rather than in frost-free freezers.
7. Solvent Choice and Safety
Bodipy dyes are commonly dissolved in organic solvents such as DMSO. DMSO is toxic; wear lab coats and disposable gloves during handling.
8. Cell Preparation and Handling
Before staining, wash cells thoroughly with PBS or other buffers to remove culture medium or serum that may interfere with staining. Optimal conditions, such as dye concentration and incubation time, vary by cell type and should be optimized accordingly.
9. Staining Process Control
Monitor temperature and time during incubation. Typically, stain at room temperature or 37°C for 5–30 min, avoiding overly long or short incubation that can affect staining quality and cell viability.
10. Observation and Detection
When using fluorescence microscopes or other imaging instruments, select appropriate excitation and emission wavelengths for Bodipy dyes to achieve optimal signals, and take care to avoid photobleaching during observation.
Application Examples of Bodipy Staining
1. Bodipy Staining to Assess Renal Lipid Metabolism in Diabetic Mice
Using Bodipy staining to study lipid dynamics, kidney tissues from diabetic mice were examined for lipid accumulation. Compared to controls, diabetic kidney disease (DKD) mice showed markedly increased green fluorescence (bright green fluorescence occurs when bound to neutral lipids such as triglycerides), indicating significant fat accumulation. Treatment with Adropin-loaded nanocapsules (Ad@Gel) reduced green fluorescence in DKD kidneys, demonstrating Ad@Gel's therapeutic effect and highlighting Adropin as a potential DKD target. 1
2. Bodipy Staining to Analyze Cartilage Homeostasis in Mice
Neutral lipid fluorescent probe Bodipy was used to locate intracellular neutral lipids in chondrocytes. Cartilage from normal BMI (<25), 50–60-year-old men without hypertension or diabetes, and osteoarthritis (OA) patients undergoing total knee replacement (TKR) was studied. Cartilage was classified as relatively healthy (non-OA) or severely damaged (OA). Bodipy staining revealed almost no green fluorescence in non-OA samples, while OA regions showed strong green fluorescence, indicating lipid droplet accumulation in chondrocytes. This suggests that lipid droplet formation may contribute to OA pathogenesis. 2
3. Bodipy Staining Shows IC2 Promotes Lipid Droplet Formation in Cancer Cells
Bodipy dyes (Bodipy 493/503 for lipid droplets and Bodipy 558/568-C12 for fatty acids) were used to observe intracellular lipid distribution via confocal microscopy. Bodipy 493/503 primarily labeled lipid droplets to assess droplet formation after IC2 (an Icaritin derivative from Epimedium) treatment. Bodipy 558/568-C12 labeled fatty acids to monitor their intracellular distribution, particularly between lipid droplets and mitochondria. Results indicated that after IC2 treatment, fatty acids preferentially accumulated in lipid droplets rather than mitochondria. IC2-induced droplet formation was caused by mitochondrial dysfunction reducing fatty acid utilization. IC2 inhibits mitochondrial oxidative phosphorylation, lowering β-oxidation efficiency, leading to intracellular lipid accumulation and droplet formation. Lipid droplet formation may be a protective mechanism in cancer cells to mitigate lipotoxicity. Inhibiting droplet formation (e.g., using DGAT1 inhibitor T863) enhances IC2's anticancer effect, suggesting potential for combined therapies targeting lipid metabolism. 3
4. Bodipy Staining Indicates DCA Counteracts SCAD Knockdown Effects on H₂O₂-Induced Lipid Droplet Accumulation
Bodipy 493/503 labeled intracellular lipid droplets, allowing observation of droplet distribution via green fluorescence. LysoTracker Red was used to study co-localization with lysosomes, revealing lipophagy activity. In H₂O₂-induced senescent HepG2 cells, SCAD (short-chain acyl-CoA dehydrogenase) knockdown via Acads siRNA significantly increased droplet-lysosome co-localization, enhancing lipid droplet degradation. Treatment with dichloroacetate (DCA) raised acetyl-CoA levels, inhibiting SCAD knockdown-promoted droplet degradation and increasing droplet accumulation. SCAD regulates lipid autophagy via acetyl-CoA, influencing age-related hepatic steatosis. Knockdown of SCAD reduces acetyl-CoA production, relieving inhibition of lipophagy and promoting droplet degradation, thereby mitigating hepatic lipid accumulation. 4
5. Bodipy Staining Demonstrates KUR Treatment Reduces Lipid Accumulation in PO-Stimulated L02 and AML12 Cells
Bodipy staining evaluated lipid droplet formation under palmitic and oleic acid (PO) stimulation. Kurarinol (KUR) treatment reduced droplet number and distribution, indicating its ability to inhibit lipid accumulation. Combined with triglyceride levels and lipid synthesis-related gene expression, KUR's effects on lipid metabolism were comprehensively assessed. In PO-stimulated human and mouse hepatocyte lines (L02 and AML12), lipid droplets increased significantly, while KUR treatment markedly reduced droplet accumulation. By lowering droplet accumulation and inflammation, KUR may help prevent progression from NAFLD to hepatocellular carcinoma (HCC). 5
Summary
Bodipy staining is a highly sensitive fluorescent method commonly used to detect neutral lipids and lipid droplet distribution in cells or tissues. By specifically targeting intracellular neutral lipids, it is suitable for labeling both live and fixed cells. Applications include chemical sensors, cellular imaging, anticancer studies, biolabeling, biomedical research, metal ion detection, and photodynamic therapy. Bodipy is also widely used in biosensing and drug screening applications.
Fluorescent Dyes Recommended for Your Research Project
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| F01-0166 | BODIPY 493/503 NHS Ester | 216961-98-7 | Bulk Inquiry |
| F01-0151 | BODIPY 406/444 | 1309918-21-5 | Bulk Inquiry |
| F01-0045 | BODIPY 505/515 | 21658-70-8 | Bulk Inquiry |
| F01-0161 | BODIPY 558/568 C12 | 158757-84-7 | Bulk Inquiry |
| F01-0155 | BODIPY Fl C5-Ceramide | 133867-53-5 | Bulk Inquiry |
| F01-0044 | BODIPY-Cholesterol | 878557-19-8 | Bulk Inquiry |
| R12-0001 | BODIPY 493/503 | 121207-31-6 | Bulk Inquiry |
| F01-0046 | Bodipy C12-Ceramide | 1246355-58-7 | Bulk Inquiry |
| F01-0251 | BODIPY 576/589 | 150173-78-7 | Bulk Inquiry |
| F01-0254 | BODIPY 493/503 carboxylic acid | 216961-95-4 | Bulk Inquiry |
Corporate Profile
BOC Sciences serves global research and industry clients, specializing in the development, production, and supply of high-performance fluorescent probes, dyes, and related customized solutions. Leveraging an advanced chemical synthesis and functional materials platform, we provide a broad portfolio that includes small-molecule fluorescent dyes, cellular and molecular imaging probes, in vivo imaging agents, and targeted fluorescent molecules, widely applied across life sciences research, bioimaging, diagnostics, and materials science.
As a dedicated fluorescence-focused subsite, we offer innovative labeling technologies and a diverse range of probes, including conventional dyes, near-infrared probes, environment-sensitive probes, and conjugation-ready fluorescent molecules. Our services extend to structural modification, spectral optimization, conjugation labeling, and fully custom synthesis, ensuring that each product meets the specific requirements of complex experimental workflows.
BOC Sciences' R&D team comprises experienced chemists and biologists, supported by state-of-the-art synthesis, purification, and analytical instruments. A rigorous quality control system guarantees high standards of purity, stability, and optical performance for all products. Our fluorescent probes and services are trusted by universities, research institutions, and biopharmaceutical companies worldwide for applications such as live-cell imaging, lipid and protein labeling, mechanistic studies, and method development. We are committed to delivering reliable solutions and expert support, serving as a long-term partner in fluorescence-based research and innovation.
References
- Yu M, Wang D, Zhong D, et al. Adropin carried by reactive oxygen species-responsive nanocapsules ameliorates renal lipid toxicity in diabetic mice. ACS Appl Mater Interfaces. 2022; 14(33):37330–37344. DOI: 10.1021/acsami.2c06957. PMID: 35951354.
- Park S, Baek IJ, Ryu JH, et al. PPARα−ACOT12 axis is responsible for maintaining cartilage homeostasis through modulating de novo lipogenesis. Nat Commun. 2022; 13(1):3. DOI: 10.1038/s41467-021-27738-y. PMID: 34987154.
- Wu G, Ying L, Zhang Q, et al. Lipid droplet formation induced by icaritin derivative IC2 promotes a combination strategy for cancer therapy. Chin Med. 2024; 19(1):178. DOI: 10.1186/s13020-024-01050-5.
- Deng D, Yang S, Yu X, et al. Aging-induced short-chain acyl-CoA dehydrogenase promotes age-related hepatic steatosis by suppressing lipophagy. Aging Cell. 2024; 23(10):e14256. DOI: 10.1111/acel.14256. PMID: 38898632.
- Zheng L, Wang G, Yang H, et al. Kurarinol restrains non-alcoholic fatty liver disease-associated hepatocellular carcinoma (HCC) progression by suppressing EGFR signaling. J Funct Foods. 2024; 121:106374. DOI: 10.1016/j.jff.2024.106374.
Custom Fluorescent Solutions Designed for Your Experiments
- DNA StainingPrecise fluorescent dyes for clear DNA visualization and analysis.
- Lipid StainingFluorescent solutions for effective lipid structure imaging.
- Cell StainingAdvanced staining for detailed cell morphology and analysis.
- Protein StainingHigh-quality staining for accurate protein detection and imaging.
- Bacteria ImagingFluorescent solutions to visualize bacterial structures and activity.
- Cell ImagingVisualize and analyze live or fixed cells using advanced fluorescence.
- Molecular ImagingCutting-edge fluorescent solutions for deep molecular analysis.
- Fluorescence ImagingHigh-resolution imaging solutions for detailed fluorescence studies.
- BioconjugationCustom bioconjugation services for protein, peptide, and dye linking.
- Drug DeliveryTailored fluorescent solutions for efficient drug delivery research.
- Molecular DiagnosticsFluorescent probes and markers for precise molecular diagnostics.
- Flow CytometryFluorescent dyes and reagents for enhanced flow cytometry analysis.
High-Performance Fluorescent Tools for Your Research
- Nitric Oxide (NO) & Reactive Oxygen Species (ROS) Probes for oxidative stress and signaling detection.
- Lipid Fluorescent Probes Dyes for lipid droplets and membranes.
- Calcium, Chloride and Other indicators Fluorescent indicators for intracellular ion flux monitoring.
- Nuclear Fluorescent Probes DNA-binding dyes for nucleus visualization.
- Other Cell Fluorescent Probes Functional probes for diverse cellular imaging studies.
- Cell membrane Fluorescent Probes Surface-labeling dyes for membrane dynamics studies.
- Endoplasmic Reticulum Fluorescent Probes ER-targeted dyes for organelle structure analysis.
- Cell Proliferation Tracer Fluorescent Probes Long-term tracking of cell division processes.
Explore More Topics
Online Inquiry


