Lipid Staining Protocols: Oil Red O, Nile Red, BODIPY & Filipin Methods Guide
Lipids are not only essential structural components of cells (such as biological membranes) and major forms of energy storage, but also play irreplaceable roles in key physiological and biochemical processes including cell signaling and molecular transport. Chemically, lipids are primarily esters of fatty acids or derivatives related to these esters. Neutral fats are mainly stored in adipose tissue and exist as oil droplet–like particles suspended within the cytoplasm. In recent years, with the deepening research into metabolic syndrome, obesity, atherosclerosis, non-alcoholic fatty liver disease, and tumor metabolism, the precise localization and quantitative detection of lipid deposition in tissues or cells have become high-frequency demands in pathological diagnosis and basic medical research. The following section provides a comprehensive review of the most representative lipid staining methods currently used in laboratories—covering both time-honored classical histological staining techniques (such as the Sudan dye method and Oil Red O staining) and modern fluorescent probe technologies suitable for high-resolution imaging (such as Nile Red, BODIPY 493/503, and the Filipin method).
Sudan Dye Staining
Staining Principle: Sudan dyes (such as Sudan III and Sudan IV) are a class of strongly lipophilic azo dyes. Their staining mechanism is not based on chemical reactions but relies on the classical principle of "like dissolves like," following physical dissolution and partition laws (i.e., extraction principle). These dyes have extremely low solubility in water but high solubility in organic solvents and tissue lipids (especially neutral fats such as triglycerides). During staining, when the dye-containing working solution covers the section, the solubility of the dye in lipid droplets is much higher than in the original solvent. As a result, dye molecules spontaneously migrate from the solvent and accumulate within intracellular lipid droplets. With continuous accumulation, the originally transparent lipid droplets display the intrinsic color of the dye (e.g., orange-red for Sudan III), thereby enabling direct localization and visualization of local lipids.
Applicable Samples:
- Tissue types: Mainly used for frozen sections (such as liver, kidney, blood vessels, and other tissues with fatty degeneration).
- Cell samples: Some cultured adherent cells.
Note: Conventional paraffin sections cannot be used because organic solvents (such as xylene and alcohol) used during dehydration and clearing will dissolve free lipids.
Staining Procedure:
(1)Fix tissues in 10% formaldehyde.
(2)After washing with water, prepare frozen or paraffin sections.
(3)After distilled water rinse, lightly counterstain with Harris hematoxylin or alum hematoxylin for 1–2 minutes.
(4)Rinse with running tap water.
(5)Wash with water and transfer to 70% alcohol for 5 seconds.
(6)Immerse in Sudan III staining solution for approximately 30 minutes or longer, and incubate at 56°C.
(7)Wash in 70% alcohol for 5–10 seconds (to remove excess surface stain).
(8)Rinse in distilled water, then transfer sections onto glass slides.
(9)Carefully blot excess water around the sections.
(10)Mount with glycerol gelatin.
Results: Lipids appear orange-red, bright red, or black; lipofuscin appears pale red; fatty acids are unstained; nuclei appear blue.
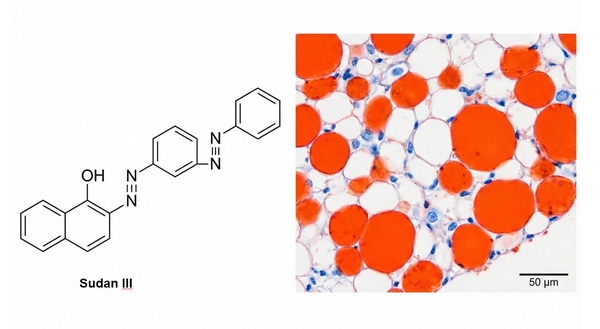 Fig. 1. Chemical structure of Sudan III and stained lipid droplets (BOC Sciences Authorized).
Fig. 1. Chemical structure of Sudan III and stained lipid droplets (BOC Sciences Authorized).
Precautions:
- After staining, quickly differentiate with 70% alcohol to remove excess surface stain. Excessive time will cause fading of lipid droplets.
- Aqueous mounting media (such as glycerol gelatin) must be used. Neutral resin mounting media must not be used, as they will dissolve lipids.
Applications:
- Pathological diagnosis: Diagnosis of early fat embolism and pathological observation of tissue fatty degeneration.
- Teaching and research: Classical demonstration of histological lipid staining.
Oil Red O Staining
Staining Principle: Oil Red O is a fat-soluble azo dye with structural similarity to lipids. It binds to lipids (especially neutral lipids) through physical adsorption. After staining, lipid droplets appear red or orange-red, while other cellular structures (such as nuclei and cytoplasm) remain unstained or show a light background.
Applicable Samples:
- Tissue types: Such as adipose tissue, liver (to display lipid deposition), and atherosclerotic plaques.
- Frozen sections: Oil Red O staining typically requires frozen sections, as paraffin embedding dissolves lipids.
Staining Procedure:
(1)Sample preparation: Fresh tissues are rapidly frozen and prepared as frozen sections (8–10 μm thickness). Alternatively, fix samples and dehydrate sequentially in 15% and 30% sucrose before preparing frozen sections. Avoid paraffin embedding to prevent lipid dissolution.
(2)Staining process: After sections return to room temperature, immerse in freshly prepared Oil Red O working solution for 6–10 minutes (protect from light). Differentiate sequentially in two jars of 60% isopropanol for 3–5 seconds (rapid rinse to remove excess dye). Rinse gently with running water. Counterstain nuclei with hematoxylin (optional, 6 minutes), differentiate, and blue with bluing solution. After washing, mount with glycerol gelatin.
(3)Microscopic observation: Lipid droplets appear red or orange-red; nuclei (if counterstained) appear blue. Observe under a standard light microscope. Avoid lipid-dissolving media (such as xylene).
Results: Lipid droplets within cells appear orange-red.
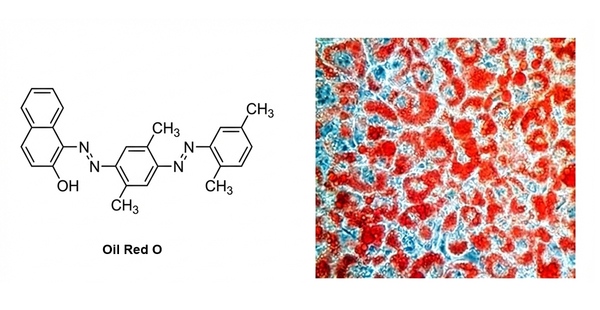 Fig. 2. Oil Red O chemical structure alongside stained lipid droplets (BOC Sciences Authorized).
Fig. 2. Oil Red O chemical structure alongside stained lipid droplets (BOC Sciences Authorized).
Precautions:
- Sample handling: Fresh or rapidly frozen samples must be used to prevent lipid loss. Paraffin sections are not suitable (lipids will be dissolved by organic solvents).
- Stability of staining solution: Oil Red O working solution should be freshly prepared before use, as prolonged storage leads to precipitation. Protect from light during staining to prevent dye degradation.
- Differentiation control: Over-differentiation causes fading, whereas insufficient differentiation leads to excessive background staining. Immediately rinse with water after differentiation to stop the reaction.
- Mounting and storage: Use aqueous mounting media (such as glycerol gelatin) to avoid lipid dissolution by organic solvents. Store stained sections protected from light, as fading occurs relatively quickly (photographic documentation is recommended).
Applications:
- Pathology: Detection of lipid cores in atherosclerotic plaques.
- Metabolic research: Lipid accumulation during hepatic steatosis and adipocyte differentiation.
- Drug screening: Evaluation of lipid-lowering drugs on cellular lipid metabolism.
Nile Red Staining
Staining Principle: Nile Red (also known as Nile Blue oxazone) is used for the localization and quantification of lipids, particularly intracellular neutral lipid droplets. Nile Red exhibits almost no fluorescence in water or other polar solvents, but its fluorescence is greatly enhanced in nonpolar environments, with significant blue shifts in absorption and emission (excitation/emission maxima in methanol: 552/636 nm). It is an excellent live-cell dye with high selectivity for lipid bodies in the cytoplasm. In cells, it appears as golden-yellow fluorescence (450–500 nm excitation; >528 nm emission).
Upgraded lipid staining tools: Nile Green and Droplite Red have properties similar to Nile Red and specifically stain intracellular lipid droplets, exhibiting green fluorescence. Nile Green can be used in combination with other fluorescent dyes for multiplex staining, compensating for certain limitations of Nile Red staining.
Applicable Samples:
- Live cells: Highly suitable for in situ lipid droplet staining of adherent or suspension cells.
- Microalgae and microorganisms: Commonly used for rapid screening of lipid content in oil-producing microalgae.
- Fixed samples: Lightly fixed cells (such as with PFA) or frozen tissue sections.
Staining Procedure:
(1)Cell culture: Culture cells on confocal dishes or glass slides to an appropriate density.
(2)Washing: Remove the culture medium and gently rinse cells 2–3 times with PBS.
(3)Staining: Add an appropriate concentration of working solution (typically 0.1–1 μg/mL, diluted from stock solution with PBS), and incubate at 37°C or room temperature for 10–15 minutes protected from light.
(4)Washing: Remove the staining solution and rinse with PBS three times, 5 minutes each, to eliminate nonspecific background.
(5)Observation: Observe directly under a fluorescence microscope or confocal microscope, or analyze using flow cytometry.
Results: Lipid droplets exhibit strong golden-yellow/yellow-green fluorescence (representing neutral lipids) or deep red fluorescence (representing polar lipids/phospholipids) under a fluorescence microscope.
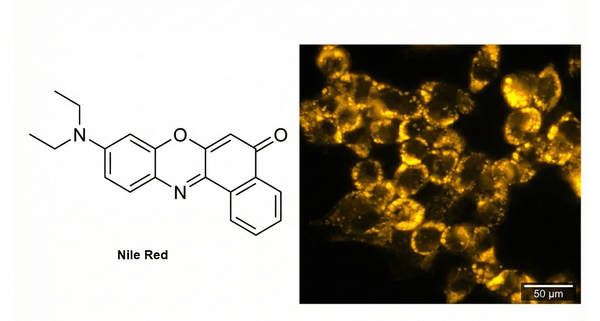 Fig. 3. Nile Red structure and fluorescent staining of cellular lipids (BOC Sciences Authorized).
Fig. 3. Nile Red structure and fluorescent staining of cellular lipids (BOC Sciences Authorized).
Precautions:
- Nile Red has poor water solubility and readily forms crystals when preparing working solutions. It is recommended to prepare fresh solutions before use, or add a small amount of DMSO during dilution to enhance solubility.
- The dye is prone to photobleaching; strict light protection is required throughout staining and observation.
- Due to its broad emission spectrum, careful selection of other dyes is necessary in multicolor fluorescence staining to avoid spectral bleed-through.
Applications:
- High-throughput analysis: Combined with flow cytometry for relative quantification of total intracellular lipid content and high-throughput drug screening.
- Bioengineering: Rapid evaluation of lipid content in industrial microalgae, yeast, and other oil-producing microorganisms.
BODIPY 493/503 Staining
Staining Principle: BODIPY 493/503 is a lipophilic fluorescent probe that localizes to polar lipids and is used to label neutral lipid content in cells, particularly lipid droplets. BODIPY 493/503 is compatible with epifluorescence microscopy, confocal microscopy, two-photon microscopy, and flow cytometry. It exhibits excitation/emission maxima at 493/503 nm and can be used in both live and fixed cell applications.
Applicable Samples:
- Live cells: BODIPY dyes are commonly used for live-cell imaging (such as dynamic tracking of lipid droplets).
- Fixed cells: Some BODIPY dyes (such as BODIPY 493/503) tolerate mild fixation (e.g., 4% PFA). If fixation is required, use 4% paraformaldehyde (PFA) at room temperature for 15 minutes; however, aldehyde fixation may quench fluorescence.
- Tissue sections: Suitable for frozen sections (paraffin sections may result in signal loss due to lipid extraction by organic solvents).
Staining Procedure:
(1)Carefully discard the culture medium and rinse once with PBS for 5 minutes.
(2)Fix with 4% formaldehyde for 10 minutes.
(3)Rinse twice with PBS, 5 minutes each.
(4)Stain with BODIPY 493/503 at 2.5 μM for 30 minutes.
(5)Rinse three times with PBS, 5 minutes each.
(6)Stain with DAPI (nuclear dye).
(7)Rinse with PBS 10 times.
(8)Observe under a microscope.
Results: Cellular lipid droplets appear green, and nuclei appear blue; live-cell staining is also possible.
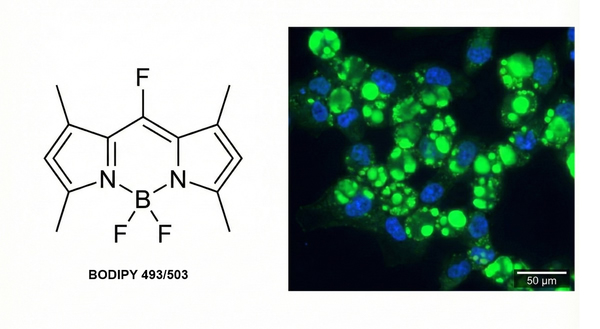 Fig. 4. BODIPY 493/503 structure and green fluorescent cellular lipid droplets (BOC Sciences Authorized).
Fig. 4. BODIPY 493/503 structure and green fluorescent cellular lipid droplets (BOC Sciences Authorized).
Precautions:
- Concentration control: The specificity of BODIPY is highly concentration-dependent. Excessive concentration increases background fluorescence and nonspecific binding to other organelle membranes; typically, 1–2.5 μM is the optimal range.
- Antibody co-staining sequence: If immunofluorescence co-staining is required, antibody incubation must be completed first, and BODIPY staining should be performed last, because washing buffers used during antibody incubation (often containing detergents such as Triton X-100) can disrupt lipid droplet structure.
- Mounting selection: Avoid mounting media containing glycerol, toluene, or other substances that may dissolve lipids or quench fluorescence. Use specialized aqueous anti-fade mounting media.
Applications:
- Organelle interaction studies: Use high-resolution confocal microscopy to investigate physical contacts and lipid transfer between lipid droplets and mitochondria or the endoplasmic reticulum.
- Metabolic pathology: Evaluate dynamic changes of lipid droplets under lipotoxicity conditions.
Filipin Staining
Staining Principle: Filipin fluorescent staining reagent is a classical and authoritative method for detecting total cholesterol by specifically binding filipin to cholesterol, forming a complex that produces blue fluorescence. This technique has been carefully optimized and experimentally validated. It is mainly used to detect the distribution of total cholesterol in various cells (animal, human, etc.) or cultured cells and is widely applied in studies of cellular pathophysiology. The product is strictly sterile, ready to use, easy to operate, stable in performance, and produces clear fluorescence signals.
Applicable Samples:
- Cultured cells: Such as macrophages (foam cell models), fibroblasts, etc.
- Frozen tissue sections: Cholesterol-rich tissues such as liver and arterial vessels.
Staining Procedure:
(1)Carefully discard the culture medium and rinse once with PBS for 5 minutes.
(2)Fix with 4% formaldehyde for 10 minutes.
(3)Rinse twice with PBS, 5 minutes each.
(4)Stain with Filipin at 0.5 mg/mL (prepare a 5 mg/mL stock solution in absolute ethanol and dilute with PBS) for 30 minutes.
(5)Rinse three times with PBS, 5 minutes each.
(6)Observe under a microscope.
Results: Neutral lipids in cells appear blue (free cholesterol-enriched regions show strong blue fluorescence, excitation wavelength approximately 340–380 nm, emission wavelength approximately 480 nm).
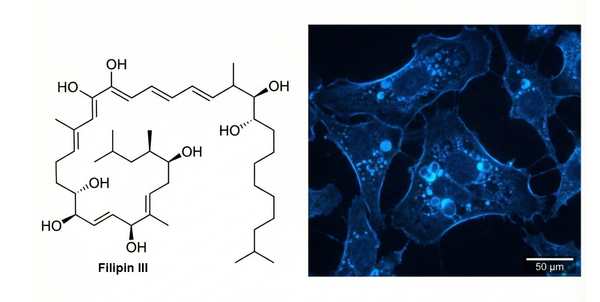 Fig. 5. Filipin chemical structure and blue fluorescent cellular cholesterol staining (BOC Sciences Authorized).
Fig. 5. Filipin chemical structure and blue fluorescent cellular cholesterol staining (BOC Sciences Authorized).
Precautions:
- Highly prone to photobleaching: Filipin is extremely light-sensitive, and fluorescence decays rapidly. Strict light protection is required throughout the procedure. Observe and capture images immediately after staining.
- Detergent sensitivity: Filipin binds to cholesterol, and detergents (such as Triton X-100 or Tween-20) can extract cholesterol from cell membranes. If antibody co-staining is required, carefully control permeabilization conditions or perform Filipin staining as the final step.
- Avoid spectral overlap: Filipin emits blue fluorescence and therefore cannot be used simultaneously with DAPI or Hoechst (both blue fluorescent nuclear dyes). For nuclear counterstaining, replace with PI (red) or other far-red dyes.
Applications:
- Diagnosis of lipid deposition diseases: Such as Niemann-Pick disease type C and other rare inherited cholesterol metabolism disorders using in vitro cytological diagnostic models.
- Atherosclerosis research: Evaluate free cholesterol accumulation during the process in which macrophages engulf oxidized low-density lipoprotein (ox-LDL) and transform into foam cells.
Fluorescent Dyes Recommended for Your Research Project
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0144 | Nile Red | 7385-67-3 | Bulk Inquiry |
| A16-0089 | CellHunt CM-DiI | 180854-97-1 | Bulk Inquiry |
| A16-0082 | DiOC16(3) | 161433-32-1 | Bulk Inquiry |
| A16-0156 | Speed DiI | 278173-35-6 | Bulk Inquiry |
| A16-0084 | Speed DiO | 164472-75-7 | Bulk Inquiry |
| R12-0001 | BODIPY 493/503 | 121207-31-6 | Bulk Inquiry |
| A16-0153 | NBD cholesterol | 78949-95-8 | Bulk Inquiry |
| A16-0073 | NBD Dihexadecylamine | 117056-66-3 | Bulk Inquiry |
| A16-0020 | Laurdan | 74515-25-6 | Bulk Inquiry |
| A16-0212 | DiO, lipophilic tracer | 28462-56-8 | Bulk Inquiry |
| F01-0274 | (E,E)-3,5-Bis(phenylethenyl)-BDP lipid stain | 148185-53-9 | Bulk Inquiry |
| A16-0206 | C-Laurdan | 959839-06-6 | Bulk Inquiry |
Corporate Profile
BOC Sciences is a premier global partner dedicated to the continuous innovation, comprehensive manufacturing, and reliable supply of high-performance fluorescent products and customized services. Catering to the rigorous demands of life sciences, diagnostic development, and materials science, we offer an extensive and versatile product portfolio. As a specialized hub for fluorescence technologies, our catalog encompasses a wide array of cutting-edge tools. This includes highly selective lipophilic tracers, organelle-targeted dyes, environment-responsive probes, conventional fluorophores, and advanced near-infrared imaging agents. These state-of-the-art molecules are indispensable for high-resolution in vitro and in vivo bioimaging, cellular metabolism studies, and precise molecular tracking.
Backed by a world-class R&D team of expert chemists and biologists, our advanced chemical synthesis platform excels beyond standard catalog offerings. We specialize in bespoke scientific services, providing custom structural modifications, precise spectral tuning, complex bioconjugation, and custom probe synthesis. Our state-of-the-art facility is equipped with advanced purification and analytical instrumentation.
A rigorous quality control system ensures that every product delivers exceptional purity, high photostability, and lot-to-lot consistency. Trusted by leading academic institutions, biopharmaceutical companies, and research centers worldwide, BOC Sciences goes beyond simply supplying reagents; we provide robust application support and tailored scientific solutions. We are fully committed to accelerating your research breakthroughs and serving as your most reliable, long-term partner in the dynamic field of fluorescent labeling and imaging sciences.
Custom Fluorescent Solutions Designed for Your Experiments
- DNA StainingPrecise fluorescent dyes for clear DNA visualization and analysis.
- Lipid StainingFluorescent solutions for effective lipid structure imaging.
- Cell StainingAdvanced staining for detailed cell morphology and analysis.
- Protein StainingHigh-quality staining for accurate protein detection and imaging.
- Bacteria ImagingFluorescent solutions to visualize bacterial structures and activity.
- Cell ImagingVisualize and analyze live or fixed cells using advanced fluorescence.
- Molecular ImagingCutting-edge fluorescent solutions for deep molecular analysis.
- Fluorescence ImagingHigh-resolution imaging solutions for detailed fluorescence studies.
- BioconjugationCustom bioconjugation services for protein, peptide, and dye linking.
- Drug DeliveryTailored fluorescent solutions for efficient drug delivery research.
- Molecular DiagnosticsFluorescent probes and markers for precise molecular diagnostics.
- Flow CytometryFluorescent dyes and reagents for enhanced flow cytometry analysis.
High-Performance Fluorescent Tools for Your Research
- Nitric Oxide (NO) & Reactive Oxygen Species (ROS) Probes for oxidative stress and signaling detection.
- Cell Proliferation Tracer Fluorescent Probes Long-term tracking of cell division processes.
- Mitochondrial Fluorescent Probes Targeted dyes for mitochondrial structure and function.
- Cell membrane Fluorescent Probes Surface-labeling dyes for membrane dynamics studies.
- Other Cell Fluorescent Probes Functional probes for diverse cellular imaging studies.
- Calcium, Chloride and Other indicators Fluorescent indicators for intracellular ion flux monitoring.
- Lysosomal Fluorescent Probes Acidic organelle markers for lysosome tracking.
- Endoplasmic Reticulum Fluorescent Probes ER-targeted dyes for organelle structure analysis.
Explore More Topics
Online Inquiry


