Multicolor Cell Staining: Principles, Dye Selection, and Panel Design
Multicolor cell staining has become a core strategy in modern fluorescence-based cell analysis because many biological questions cannot be answered by a single stain alone. Researchers often need to examine several cellular features at the same time, such as nuclear position, membrane integrity, organelle distribution, viability status, and the expression or localization of multiple targets within the same sample. By combining fluorophores with distinguishable spectral properties, multicolor staining makes it possible to analyze these signals in parallel rather than through separate experiments that are harder to compare directly. The value of this approach is clear, but so is the difficulty. A multicolor panel can fail even when every individual stain appears reasonable on its own. Spectral overlap, signal spillover, unequal fluorophore brightness, poor control design, autofluorescence, and incompatibility between live-cell and fixed-cell workflows can all undermine interpretation. That is why multicolor cell staining is not simply an extension of single-color staining. It requires a more deliberate framework that connects the biological question, the instrument configuration, the marker hierarchy, and the fluorophore selection strategy.
What Is Multicolor Cell Staining?
Multicolor cell staining refers to the simultaneous use of two or more fluorescent stains, probes, or labeled reagents within the same sample so that multiple cellular structures, markers, or functional states can be analyzed in parallel. Its central purpose is to preserve biological context while expanding analytical depth. Instead of asking one question per sample, researchers can ask several related questions at the same time, such as where different structures are located, whether markers co-localize, how distinct cell populations differ, or whether a treatment changes more than one cellular parameter within a single experiment.
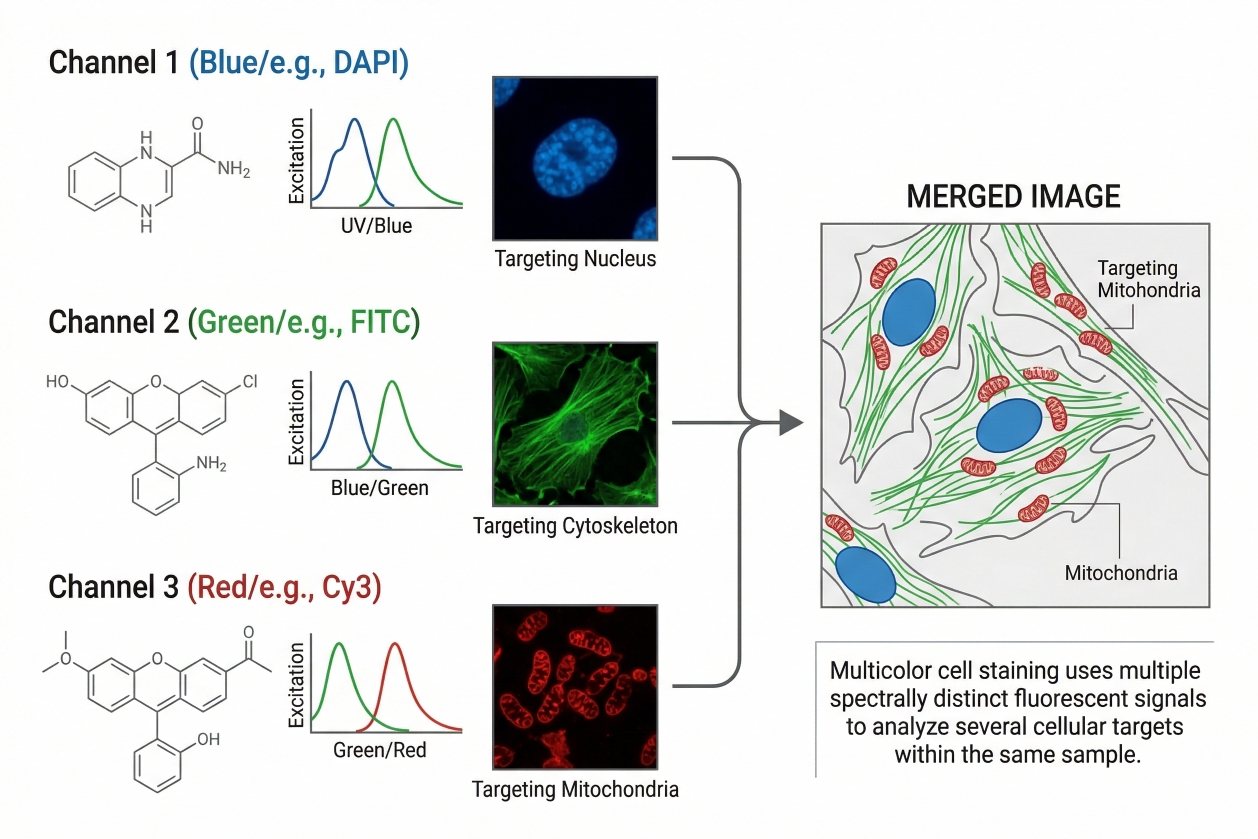 Fig. 1. Multicolor cell staining uses multiple spectrally distinct fluorescent signals to analyze several cellular targets within the same sample (BOC Sciences Authorized).
Fig. 1. Multicolor cell staining uses multiple spectrally distinct fluorescent signals to analyze several cellular targets within the same sample (BOC Sciences Authorized).
In practical laboratory terms, multicolor cell staining is a fluorescence workflow in which multiple dyes with distinguishable excitation and emission profiles are assigned to different targets or readout layers in one specimen. These targets may include nuclei, membranes, organelles, viability state, intracellular markers, or cell-type-associated signals. The goal is not simply to generate several colors in one image. The real goal is to generate separable, interpretable fluorescence channels that can be linked to different biological features without excessive cross-interference. This means multicolor cell staining is inherently more demanding than single-stain workflows. The suitability of one fluorophore can no longer be judged in isolation, because its usefulness depends on how it behaves relative to the other dyes in the same panel. A dye that works well alone may become a poor choice if it overlaps strongly with another channel, dominates a dim marker, or becomes difficult to compensate or unmix in the chosen instrument.
How It Differs from Single-Color Staining?
Single-color staining usually focuses on one structure or one readout at a time. That makes it easier to optimize concentration, background, and signal quality because the fluorophore does not need to coexist with multiple competing channels. Multicolor staining is fundamentally different because the panel must behave as an integrated system. Each dye has to be selected not only for target relevance, but also for brightness balance, spectral spacing, platform compatibility, and how its signal will affect interpretation of the other dyes. This difference becomes especially important when signals vary widely in abundance or when co-expressed markers must be separated clearly. In single-color workflows, a bright fluorophore is usually helpful. In multicolor workflows, the same fluorophore may be too bright for a highly abundant marker and may create unnecessary spread into nearby channels. As a result, multicolor design requires more planning before the first staining step is performed.
Common Readout Platforms: Flow Cytometry, Immunofluorescence, Live-Cell Imaging
Multicolor staining is widely used across flow cytometry, immunofluorescence staining, and live-cell imaging. In flow cytometry, the emphasis is usually on distinguishing cell populations, marker-positive subsets, and viability or state-related differences across large numbers of events. In immunofluorescence and related microscopy workflows, the emphasis is more often on co-localization, spatial organization, and morphological context. In live-cell imaging, multicolor staining can be used to follow several structures or states in real time, but this requires especially careful management of phototoxicity, photobleaching, and dye compatibility with living cells. Although the same general multicolor principles apply across these platforms, the operational priorities are different. Flow cytometry emphasizes clean signal separation and compensation behavior. Microscopy emphasizes localization fidelity, visual contrast, and image interpretability. Live-cell imaging adds the extra requirement that the dyes should not compromise the biology being observed.
Why Multicolor Staining Matters for Complex Cell Analysis?
Complex cell systems often cannot be understood through a single label because multiple cellular features interact at the same time. A treatment may alter viability, organelle distribution, and marker localization simultaneously. A cell population may need to be distinguished not only by one marker, but by a combination of structural and functional readouts. Multicolor staining is valuable precisely because it preserves these relationships within one sample. It reduces the need to infer cross-sample relationships from separate experiments and instead allows multiple dimensions of cell state to be observed together. For this reason, multicolor staining has become central in applications such as immunophenotyping, co-localization analysis, organelle tracking, high-content imaging, and multiplex cell-based assay development. It is most powerful when each fluorophore is chosen to support a specific analytical role within a coherent panel rather than when several stains are simply combined for convenience.
Core Principles Behind Multicolor Cell Staining
Once the concept is clear, the next step is understanding why multicolor staining succeeds or fails. Good panels are not created only by choosing visually different dyes. They work because the fluorophores are compatible with the available optical system, separable enough for the intended readout, balanced in brightness, and appropriate for the biological sample. The core principles below explain the main technical factors that shape panel performance.
- Excitation and Emission Basics: Every fluorescent dye is defined in part by the wavelengths at which it is excited and the wavelengths at which it emits fluorescence. In a multicolor experiment, these two properties determine whether a dye can be used effectively with the lasers, light sources, filters, or detector windows available on the instrument. The important point is not simply whether the dye can be excited, but whether its emitted signal can be collected in a way that remains distinguishable from the rest of the panel. Many multicolor problems begin when dye selection is based only on nominal emission peaks. In real instruments, emission is not a single sharp line; it occurs across a spectrum, and these spectra can overlap substantially. Therefore, channel assignment should always account for spectral width and not just for the central value in a dye datasheet.
- Signal Separation, Spectral Overlap, and Spillover: Spectral overlap occurs when the emission of one dye extends into a detector channel intended for another dye. In panel design, this creates spillover, and in many systems it also contributes to spread that makes dim signals harder to distinguish. In microscopy, the consequence may appear as misleading co-localization or hazy color separation. In flow cytometry, it may appear as compensation burden, distorted distributions, or reduced resolution between populations. Effective multicolor panels therefore aim for usable signal separation rather than merely different names or colors. Two fluorophores that both look "greenish" or "red-shifted" are not necessarily incompatible, but they must be assessed according to the actual optical configuration and signal intensity relationships within the panel. This is one reason why panel design should start before reagents are purchased in quantity.
- Brightness, Photostability, and Background: Brightness matters because low-abundance targets need enough fluorescence to remain distinguishable above background and spillover. However, brightness must be considered in relation to marker abundance and panel structure. A very bright dye assigned to an already abundant target can worsen spread and make adjacent dim channels more difficult to interpret. Conversely, a dimmer fluorophore may be acceptable or even preferable for highly expressed targets. Photostability matters most in microscopy, especially in repeated acquisition or confocal scanning. A fluorophore that bleaches quickly may compromise time-lapse observation or make later-acquired channels less reliable than earlier ones. Background is equally important. Sample autofluorescence, non-specific staining, and diffuse retained dye can all reduce useful contrast. In complex samples, the best fluorophore is often the one that provides the clearest biological separation after background is considered, not just the one with the highest theoretical brightness.
- The Role of Instrument Configuration in Panel Feasibility: Instrument configuration is one of the most decisive but sometimes overlooked factors in multicolor feasibility. The number and wavelength of lasers, the available filters or spectral detectors, detector sensitivity, and software compensation or unmixing capacity all shape what is realistically possible. A panel that performs well on one instrument may be difficult or impossible to reproduce on another system with a different optical layout. This is why panel design should never rely only on generic fluorophore charts. The instrument itself defines the operational meaning of spectral compatibility. In practice, successful multicolor staining begins by matching the biological question not only to dye properties, but also to the exact platform that will be used to read the panel.
Planning a multicolor cell staining panel with minimal spectral overlap?
We can help you select compatible dyes based on excitation and emission profiles, target abundance, imaging platform, and overall panel balance.
Dye Selection for Multicolor Cell Staining
After understanding the core principles, the next step is fluorophore selection. This is the point where general rules become real decisions. Researchers need to move from "which dyes exist" to "which dyes make sense for this panel, this sample, and this instrument." The most effective way to do that is through a structured selection framework rather than through a simple list of fluorophore names.
- Key Selection Criteria: Brightness, Spectral Separation, and Stability: Three of the most important criteria in multicolor dye selection are brightness, spectral separation, and stability. Brightness affects detectability, spectral separation affects distinguishability, and stability affects whether the signal remains reliable across the acquisition window. A fluorophore that is bright but overlaps heavily with another channel may create more problems than value. A fluorophore that is spectrally attractive but bleaches rapidly may be poor for repeated imaging. A fluorophore that is stable but too dim for a low-abundance target may fail to provide sufficient analytical resolution. In practice, these three criteria must be evaluated together. Researchers should think in terms of usable signal in the real panel rather than theoretical fluorophore performance in isolation.
- Matching Dyes to Antigen Abundance or Target Expression Level: One of the most important panel design rules is to pair dim targets with bright fluorophores and highly abundant targets with less demanding channels. This principle improves panel balance because low-expression markers need stronger signal support to remain distinguishable, while highly abundant targets can usually tolerate less bright dyes without becoming unreadable. Ignoring this rule often leads to panels in which the brightest fluorophores are wasted on strong markers, while the biologically critical dim markers are left with weaker channels. This logic applies in both flow and imaging workflows, although the exact expression hierarchy may need to be estimated differently depending on the platform. The goal is not maximum brightness for every target, but optimal resolution across the panel as a whole.
- Choosing Dyes for Fixed Cells vs. Live Cells: Live-cell and fixed-cell workflows place different demands on fluorophore selection. In live-cell experiments, dyes must be compatible with cell health, repeated observation, and the biological process being monitored. In fixed-cell experiments, the emphasis often shifts toward structural stability, wash tolerance, and compatibility with permeabilization or multistep staining. A fluorophore chosen for one workflow should not automatically be assumed suitable for the other. This distinction becomes especially important in multicolor panels because live-only dyes may not retain meaningful localization after fixation, while some fixed-cell reagents may be too disruptive or impractical in living systems. Researchers planning mixed workflows should account for this difference at the dye-selection stage rather than after the panel has already been assembled.
- Common Dye Classes Used in Multicolor Staining: Multicolor cell staining can draw from several broad dye and probe families, including general fluorescent dyes, fluorescent probes, and more specific fluorophore families such as Coumarin, Fluorescein FAM, Rhodamine, Cyanine, BODIPY, and Alexa Fluor. Each class has advantages and limitations in spectral placement, brightness, stability, and biological compatibility. These dye classes should not be chosen merely because they are well known. Their relevance depends on how the fluorophore behaves in the actual multicolor panel and whether it is being used for structural labeling, functional readout, live-cell staining, or endpoint analysis.
- When to Avoid Tandem Dyes or Highly Overlapping Fluorophores: Tandem or closely overlapping fluorophores can be useful in some systems, but they are not always the best choice for routine multicolor panel design. Whenever overlap is high, the panel becomes more dependent on compensation quality, detector consistency, and stable fluorophore behavior. If the biological question can be answered with cleaner spectral separation, that option is usually more robust. Highly overlapping dyes are particularly risky when the panel already includes dim markers, high autofluorescence, or co-expressed targets that must be separated cleanly. In general, the more complex the sample and the more demanding the interpretation, the more valuable it becomes to reduce unnecessary overlap instead of trying to manage it after the fact.
How to Design a Reliable Multicolor Staining Panel?
This is the practical core of multicolor cell staining. A strong panel is not built by adding fluorophores one by one until all channels are occupied. It is built through a staged design process that begins with the biological question and ends with validation through proper controls. The most reliable panels are usually those that are intentionally designed to solve a defined analytical problem rather than those assembled around available reagents.
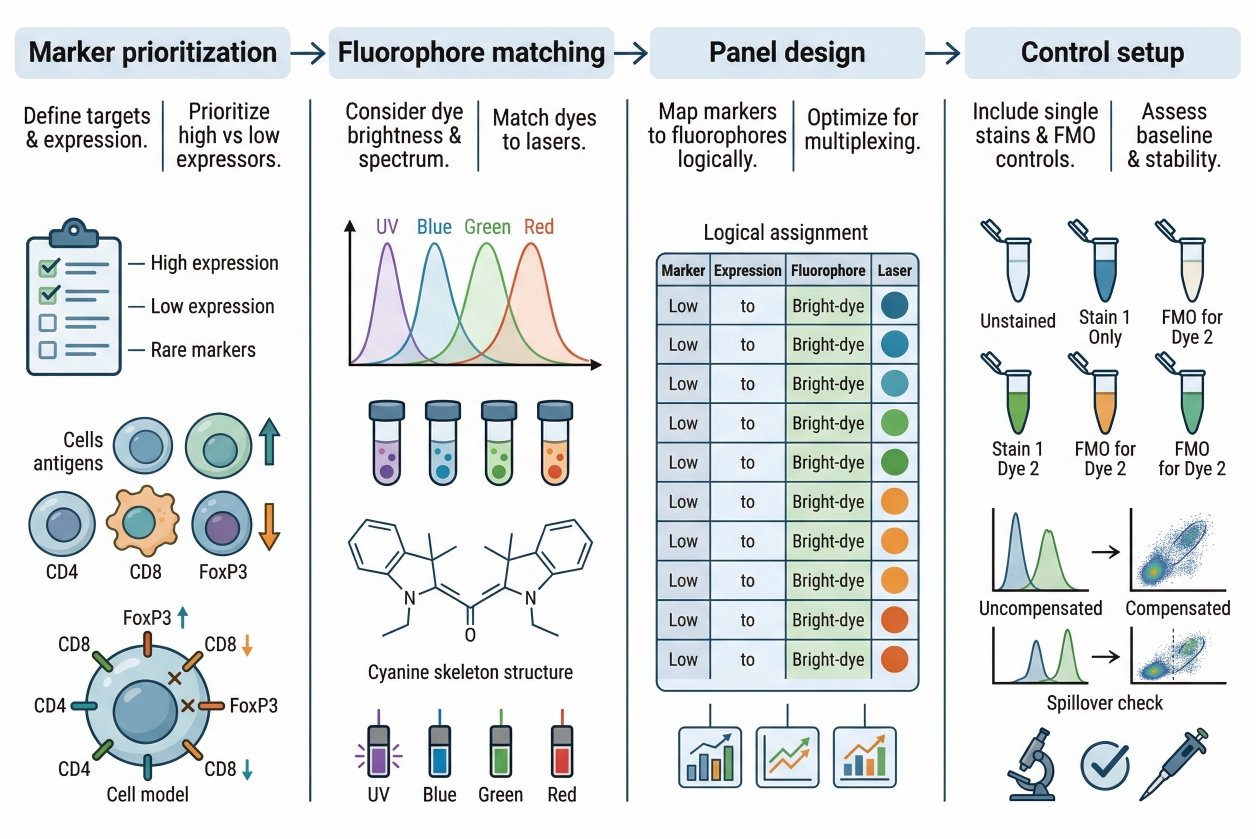 Fig. 2. A practical workflow for designing and validating a multicolor staining panel (BOC Sciences Authorized).
Fig. 2. A practical workflow for designing and validating a multicolor staining panel (BOC Sciences Authorized).
Step 1: Define the Biological Question and Marker Hierarchy
Every panel should begin with a clear biological objective. Which cell states, structures, or targets must be distinguished, and which of them are essential versus optional? Once that is established, the markers can be ranked according to importance and likely abundance. This hierarchy matters because the most critical and most difficult targets should receive the strongest fluorophore support. Without this hierarchy, panel design often becomes visually balanced but analytically weak. The panel may contain several nice-looking channels while still failing to resolve the most important biological distinction.
Step 2: Review Instrument Lasers, Filters, and Detector Channels
Before assigning fluorophores, researchers should review the exact optical layout of the intended instrument. Which lasers are available? Which filters or detection windows will be used? How much flexibility exists for compensation or spectral unmixing? These questions determine the actual panel space, and they often narrow the fluorophore options more effectively than a general catalog search. This step is particularly important when working across multiple instruments or planning a panel that may later be transferred. A panel should be designed for real hardware, not for an idealized instrument configuration.
Step 3: Pair Dim Markers with Bright Fluorophores
Low-abundance or weak targets should generally receive the brighter and cleaner fluorophores in the panel. This improves the likelihood that they will remain distinguishable above background and spillover. Strongly expressed markers can usually tolerate more moderate channels without becoming unreadable. This principle is one of the most widely accepted and practically useful rules in panel design because it improves resolution where it is most needed. Importantly, "bright" should still be evaluated in context. A bright fluorophore that creates strong spread into nearby channels may be less useful than a slightly less bright fluorophore that gives a cleaner panel overall.
Step 4: Separate Co-Expressed Markers to Reduce Spillover Risk
Markers that are commonly co-expressed within the same cell should be assigned fluorophores with stronger spectral separation whenever possible. If co-expressed markers are placed into channels with substantial overlap, the resulting spread or bleed-through may make biologically distinct populations look less resolved. In imaging, this can complicate co-localization analysis. In flow, it can reduce discrimination between related subsets. This principle matters less for mutually exclusive markers than for markers expected to appear together. Good panel design anticipates these relationships rather than treating all targets as optically independent.
Step 5: Plan Controls, Compensation, and Validation Strategy
A multicolor panel is not reliable until it has been validated with appropriate controls. Depending on the platform, this may include single-stain controls, unstained controls, fluorescence minus one controls, and compensation or spectral unmixing validation. These controls are not optional extra steps. They are how the researcher confirms that the panel behaves as intended rather than only appearing reasonable in a representative image or initial run. Validation should also include a pilot phase whenever possible. Small-scale testing helps identify unexpected overlap, weak target performance, or channel imbalance before the full experiment is committed. This step often saves more time than it consumes.
Need help optimizing dye combinations for imaging, flow cytometry, or assay development?
We can help match fluorophore brightness, channel spacing, and marker priorities to your workflow for clearer signal separation and more reliable data.
Common Challenges in Multicolor Cell Staining and How to Solve Them
Even a carefully planned multicolor panel can encounter problems once it is tested in real samples. The most important point is that multicolor failures are usually panel-level problems rather than isolated reagent problems. A dye that appears acceptable by itself may become problematic only when used beside other fluorophores, in highly autofluorescent samples, or under a detection setup that increases spillover burden. Understanding the most common failure modes makes troubleshooting more systematic and less dependent on guesswork.
- Spectral Overlap and Compensation Errors: Spectral overlap is one of the defining challenges of multicolor staining. When overlap is substantial, compensation or unmixing becomes more sensitive to control quality and detector stability. Small errors can then propagate through the panel and reduce confidence in marker separation. The most effective solution is usually to reduce preventable overlap at the design stage rather than relying on downstream correction alone.
- High Background and Non-Specific Staining: High background in multicolor experiments is especially problematic because it can affect several channels simultaneously and obscure dim markers. Non-specific staining, insufficient washing, sample debris, and excessive fluorophore concentration can all contribute. In multicolor workflows, background is more than a visual nuisance. It reduces dynamic range and makes compensation or channel interpretation more difficult.
- Uneven Signal Intensity Across Markers: Uneven signal intensity is often a sign that marker-fluorophore pairing was not well matched to target abundance. Panels in which some channels are saturated while others are barely visible are harder to interpret and more difficult to compare between runs. This problem can often be reduced by reassigning brighter fluorophores to dimmer targets and re-optimizing reagent levels for each stain rather than using uniform assumptions across the panel.
- Autofluorescence in Complex Cell Samples: Autofluorescence can compromise multicolor experiments by elevating baseline signal in specific spectral regions, especially in complex or stressed samples. Its impact depends on the sample type, platform, and fluorophore choices. Panels that place weak biologically important signals into autofluorescence-heavy regions often perform poorly even when the fluorophore itself is otherwise suitable. This is one reason sample background should be considered during early fluorophore assignment.
- Reproducibility Issues and Batch-to-Batch Variation: Reproducibility problems in multicolor staining may arise from differences in reagent handling, instrument settings, sample condition, or control design between runs. Because multicolor panels are more interconnected than single-color assays, small inconsistencies can have amplified effects. Standardized preparation, documented panel versions, stable control strategies, and careful tracking of staining conditions are therefore important for maintaining reproducibility over time.
Applications of Multicolor Cell Staining
Multicolor staining is valuable because it supports several important application types across imaging and cell analysis. Its usefulness is greatest where multiple biological dimensions need to be measured together rather than inferred from separate experiments. The applications below represent some of the most common and most informative uses of multicolor workflows.
Immunophenotyping and Cell Population Profiling
One of the most established applications of multicolor cell staining is immunophenotyping and cell population profiling, especially in fluorescence-based single-cell analysis workflows. In these experiments, one marker is rarely enough to define a biologically meaningful population, because related cell subsets often share partial marker expression while differing in only a few critical signals. Multicolor staining allows several markers to be measured in the same cell at the same time, making it possible to distinguish complex subpopulations, resolve heterogeneous samples, and define cell states more precisely than single-color staining ever could. This is particularly important when the goal is to identify rare subsets, compare activation or differentiation patterns, or separate biologically similar populations that would otherwise overlap if only one or two signals were available.
Co-Localization Analysis in Multicolor Immunofluorescence
In multicolor immunofluorescence, multicolor staining is widely used for co-localization analysis, where the purpose is to determine whether two or more targets appear in the same spatial region, partially overlap, or remain clearly separated within a cell. This application is especially valuable in studies of intracellular organization, protein redistribution, compartment interaction, and treatment-induced structural change. By assigning different fluorescent channels to different targets, researchers can evaluate whether two signals follow the same spatial pattern or whether apparent association is only partial, conditional, or absent. The power of this application lies in preserving spatial context, but it also demands careful interpretation because meaningful co-localization depends on clean signal separation, low background, and appropriate controls to rule out bleed-through or false overlap caused by optical rather than biological factors.
Tracking Organelles or Cellular States in Imaging Experiments
Multicolor staining is also highly useful when researchers need to track several intracellular compartments or cellular states within the same imaging experiment. For example, one channel may define the nucleus, another may label a specific organelle such as mitochondria or lysosomes, and a third may report viability, membrane behavior, or another state-related parameter. This type of design allows structural and functional information to be interpreted together rather than in isolation. The result is a more complete view of how cellular organization changes across time, treatment, or stress conditions. In practice, this application is especially valuable when a biological process affects more than one compartment or when a functional readout needs to be interpreted in relation to intracellular localization rather than as a standalone intensity measurement.
High-Content and Multiplex Cell-Based Assays
High-content and multiplex cell-based assays are another major application area for multicolor cell staining because these workflows depend on extracting multiple layers of information from the same sample in a scalable and reproducible way. Instead of running separate assays for morphology, viability, organelle distribution, or treatment response, multicolor staining allows these features to be integrated into a single acquisition and analysis framework. This improves efficiency, preserves sample context, and often yields more informative datasets because several cellular endpoints can be compared directly within the same cells. In assay development and screening environments, this approach is especially useful for distinguishing broad toxicity from specific phenotype change, identifying responder versus non-responder cells, and building richer decision criteria than any one stain could provide by itself.
Comparing Applications in Flow Cytometry vs. Microscopy
Although the same multicolor logic applies across platforms, the application of multicolor staining differs substantially between flow cytometry and microscopy. In flow cytometry, multicolor staining is mainly used to resolve populations, classify cell subsets, and measure marker-defined heterogeneity across large numbers of cells with high statistical power. In microscopy, the emphasis is usually different: multicolor staining is used to preserve spatial information, assess co-localization, compare morphology, and interpret how different signals are organized within the same cellular architecture. As a result, the value of a multicolor panel must always be judged in platform context. A panel designed for clean population separation in flow may not be ideal for image-based localization work, while an imaging-oriented panel with excellent spatial contrast may not offer the same efficiency or separation quality in flow-based analysis. Understanding this difference helps researchers choose fluorophores and controls that support the real application rather than assuming one multicolor design principle works equally well everywhere.
How BOC Sciences Supports Multicolor Cell Staining?
BOC Sciences supports multicolor cell staining projects through integrated capabilities in fluorescent dye and probe supply, multichannel staining strategy design, custom multifunctional probe development, and workflow-oriented imaging support. Rather than treating multicolor staining as a simple combination of different fluorophores, we help researchers align spectral distribution, organelle targeting, signal balance, imaging platform settings, and downstream analysis goals with the actual needs of the experiment. This is especially important in multicolor workflows, where channel compatibility, fluorophore brightness, crosstalk control, and assay complexity all influence whether the final panel remains interpretable. Our support is therefore structured to cover both routine multicolor imaging needs and more advanced multiplexed cell analysis applications.
Multicolor Fluorescent Dye and Probe Supply
- Access to a broad range of fluorescent dyes spanning multiple spectral regions, including blue, green, red, and near-infrared fluorescence, to support different multichannel imaging requirements.
- Support for selecting specialized fluorescent probes targeting different cellular structures, including nuclei, mitochondria, endoplasmic reticulum, cell membranes, and other intracellular compartments commonly used in multicolor staining workflows.
- Availability of highly photostable, high signal-to-noise fluorescent dyes better suited to complex multicolor experiments where signal separation and image clarity are critical.
- Product support for diverse dye combinations appropriate for both live-cell and fixed-cell staining strategies, depending on sample handling and imaging design.
Multicolor Staining Strategy Design and Channel Optimization
- Practical support for designing multichannel staining panels that improve spectral separation between fluorophores and reduce avoidable interference across channels.
- Guidance on matching dye selection to microscope excitation sources, filter sets, and detection channel configurations so that fluorophore choice fits the actual imaging platform.
- Assistance with balancing fluorescence intensity across multiple stains to reduce crosstalk, minimize signal competition, and improve overall panel readability.
- Suggestions for more complex staining architectures, including multi-organelle co-staining and combined structural plus functional fluorescence workflows.
Customized Multifunctional Fluorescent Probe Development
- Custom development of multifunctional fluorescent probes according to specific experimental needs, including probes designed for ROS, pH, ion-associated, or related intracellular fluorescence readouts.
- Support for multi-target labeling strategies or responsive probe designs adapted to more complex cell analysis workflows where one probe may need to contribute more than a simple localization signal.
- Structural modification and performance optimization of fluorescent dyes to improve stability, selectivity, cell permeability, and practical behavior in multicolor imaging systems.
- One-stop custom services covering molecular design, synthesis, purification, and characterization for specialized multicolor fluorescent probe development.
Integrated Multicolor Imaging Workflow and Application Support
- Support for integrating multicolor staining into complete experimental workflows that connect staining, imaging, data acquisition, and downstream analysis rather than treating these steps separately.
- Guidance for staining strategies compatible with high-content screening (HCS) and high-throughput screening (HTS), helping researchers build more scalable multicolor assay systems.
- Application support for complex data analysis needs, including multiparameter phenotypic profiling, cell segmentation, and quantitative image-based analysis in multicolor experiments.
- Workflow-oriented assistance aimed at improving experimental reproducibility, data consistency, and cross-platform usability in larger multicolor imaging and screening programs.
Do You Need A Consultation?
BOC Sciences integrates cutting-edge fluorescence technologies to accelerate your research, driving next-generation solutions for drug discovery and diagnostics.
Transform Your Studies with Cutting-Edge Fluorescent Products
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0018 | N-(2-hydroxyethyl)-Naphthalimide | 5450-40-8 | Bulk Inquiry |
| A16-0178 | 3,4,5,6-Tetrabromophenol sulfonephthalein | 77172-72-6 | Bulk Inquiry |
| A16-0027 | Coumarin hydrazine | 113707-87-2 | Bulk Inquiry |
| A16-0015 | WSP-5 | 1593024-78-2 | Bulk Inquiry |
| A16-0176 | True BLUE chloride | 71431-30-6 | Bulk Inquiry |
| A16-0030 | 6-NBDG | 108708-22-1 | Bulk Inquiry |
| A16-0039 | C6 NBD L-threo-dihydroceramide | 114301-95-0 | Bulk Inquiry |
| A16-0065 | Lucifer Yellow CH dilithium salt | 67769-47-5 | Bulk Inquiry |
| A16-0009 | L 012 sodium salt | 143556-24-5 | Bulk Inquiry |
| A16-0075 | P-Xylene-bis-pyridinium bromide | 14208-10-7 | Bulk Inquiry |
High-Performance Fluorescent Tools for Your Research
- Nerve Terminal Probes Fluorescent tracers for synaptic activity analysis.
- Apoptosis Fluorescent Probes Probes detecting programmed cell death events.
- Calcium, Chloride and Other indicators Fluorescent indicators for intracellular ion flux monitoring.
- Endoplasmic Reticulum Fluorescent Probes ER-targeted dyes for organelle structure analysis.
- Other Cell Fluorescent Probes Functional probes for diverse cellular imaging studies.
- Nitric Oxide (NO) & Reactive Oxygen Species (ROS) Probes for oxidative stress and signaling detection.
- Mitochondrial Fluorescent Probes Targeted dyes for mitochondrial structure and function.
- Ion Fluorescent Probes Indicators for real-time ion concentration imaging.
Explore More Topics
Online Inquiry


