Cell Membrane Staining Guide: Fluorescent Dyes, Applications, and Key Considerations
Cell membrane staining is widely used in fluorescence-based cell analysis because the plasma membrane provides the most direct visual definition of where one cell ends and another begins. In many imaging workflows, researchers are not only trying to confirm that cells are present. They need to determine whether cell boundaries remain continuous, whether membrane contours appear smooth or irregular, whether cells spread broadly or stay compact, and whether neighboring cells can be separated clearly enough for morphology comparison or image segmentation. This is why membrane staining often becomes a central structural channel rather than a decorative add-on. A membrane stain that produces bright fluorescence but weak boundary logic can mislead downstream interpretation, especially in dense cultures, multicolor imaging panels, and experiments where morphology or cell-cell contact matters. A useful membrane staining page therefore needs to do more than list dye names. It should explain what membrane staining means in fluorescence imaging, how different membrane fluorescent dye classes behave, and how workflow choices affect whether a membrane signal truly supports boundary-level interpretation.
What Is Cell Membrane Staining in Fluorescence Imaging?
Cell membrane staining, also described in many workflows as plasma membrane staining, refers to the use of fluorescent dyes or fluorescent probes to label the outer membrane region of cells so that cell boundaries, membrane-associated contours, and spatial relationships at the cell periphery can be visualized under fluorescence imaging. In practical cell analysis, membrane staining is not simply about making the edge of a cell visible. It is used to support single-cell boundary definition, morphology assessment, contact-region observation, multicolor spatial interpretation, and segmentation-oriented image analysis. Depending on probe design, the observed fluorescence may remain predominantly associated with the plasma membrane during the intended imaging window, or it may gradually redistribute, become punctate through internalization-related processes, or respond differently after fixation and wash steps. That is why membrane staining should be treated as an interpretive workflow rather than as a routine color overlay.
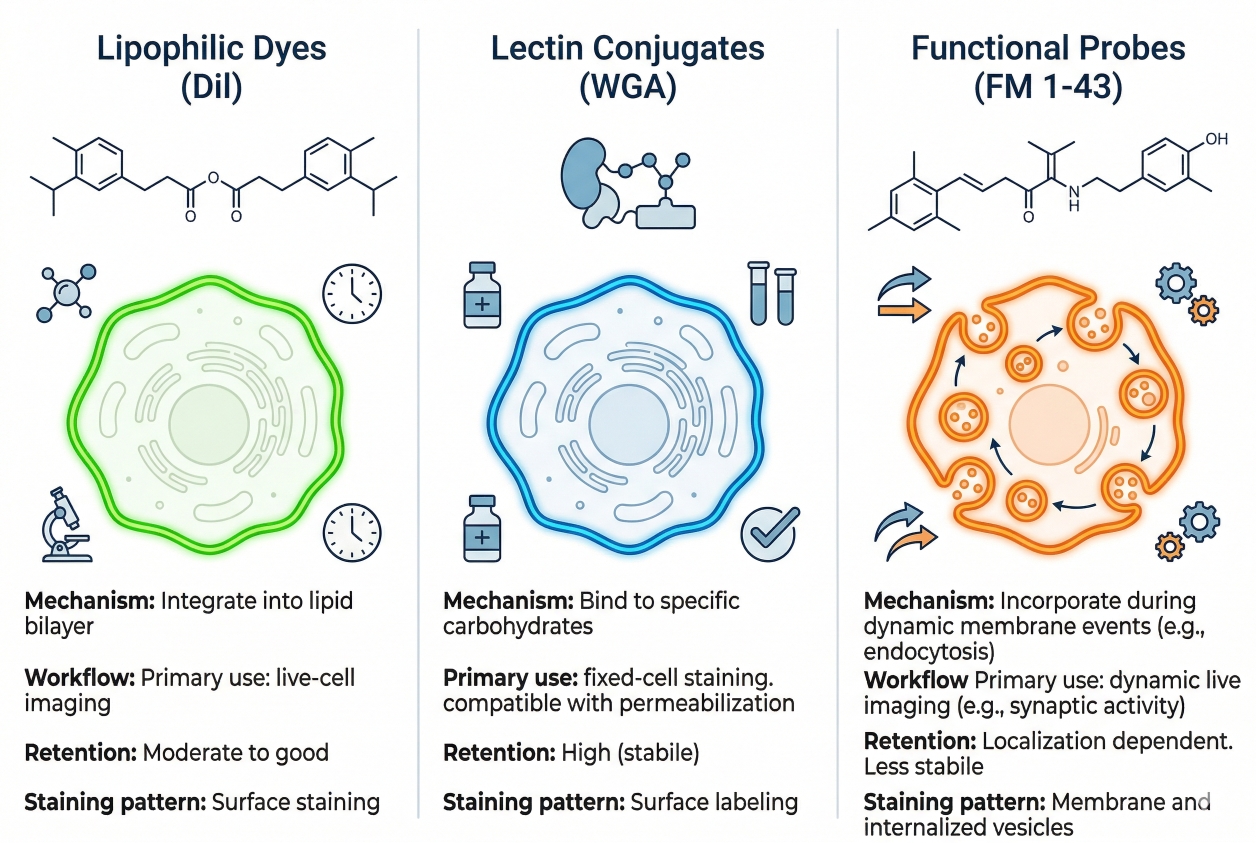 Fig. 1. Cell membrane stains vary in localization pattern, retention, and workflow compatibility (BOC Sciences Authorized).
Fig. 1. Cell membrane stains vary in localization pattern, retention, and workflow compatibility (BOC Sciences Authorized).
In fluorescence imaging, membrane staining means using a membrane fluorescent dye or membrane fluorescent probe to create a signal that defines the outer contour of a cell strongly enough to support visual or analytical interpretation. In simple applications, this can mean outlining individual cells so that shape and boundary continuity become easier to observe than with nuclear staining alone. In more advanced workflows, membrane staining helps locate internal organelle signals relative to the cell edge, supports measurements such as projected cell area or edge complexity, and improves the ability to distinguish adjacent cells in crowded fields. A useful membrane stain therefore does not only need fluorescence brightness. It also needs workflow behavior that matches the readout objective, whether that objective is clean endpoint morphology, short-term live observation, or multicolor panel integration.
Why Membrane Staining Is More Than a Visual Outline?
Membrane staining is more than a visual outline because the plasma membrane serves as a practical spatial frame for many kinds of cell-based interpretation. A nucleus stain can show where the cell center roughly lies, but it often does not reveal how much the cell has spread, whether the edge is continuous, or how neighboring cell boundaries relate to one another. By contrast, a membrane stain can help determine whether cells appear elongated, rounded, polarized, tightly packed, loosely distributed, or engaged in local contact. In image-analysis workflows, this becomes especially important because segmentation quality depends heavily on whether the cell edge is represented as a continuous and biologically believable boundary. A membrane channel that is too diffuse, too bright, or too unstable over time may still look attractive in a merged image while remaining poorly suited for reliable interpretation.
Where Membrane Staining Becomes Especially Useful?
Membrane staining becomes especially useful when researchers need more than presence-or-absence information. It is valuable in morphology-focused imaging where the main goal is to compare cell outline, spreading behavior, protrusions, or edge irregularity across experimental groups. It is also useful in high-density cultures where nuclei alone are insufficient for separating neighboring cells, in short live-cell imaging workflows where boundary position must be monitored over time, and in multicolor assays where organelle or intracellular signals need to be interpreted relative to the whole-cell outline. In addition, membrane staining becomes central in cell-cell interaction studies because the geometry of contact regions, boundary compression, and cluster organization often depends on having a clear membrane-level channel. For all of these reasons, membrane staining is frequently one of the most informative structural layers in cell imaging rather than a secondary visual effect.
How Membrane Stains Differ in Workflow Behavior?
The most practical differences among membrane stains include how rapidly they generate membrane-associated fluorescence, how cleanly they define the outer boundary, how likely they are to undergo time-dependent redistribution, how well they tolerate washing, and whether their interpretive meaning changes after fixation or downstream processing. Lipophilic carbocyanine-type membrane probes often provide strong membrane association and can be useful when clear outline labeling is needed. In contrast, some membrane-related fluorophores may be more attractive for panel design or custom conjugation logic than for direct plasma membrane outlining in routine cell imaging. Spectral channel matters too, but it should be treated as one part of a larger decision rather than as the only criterion. A membrane stain that fits the channel plan but fails to preserve a clean and interpretable cell boundary is rarely the right answer.
Main Types of Cell Membrane Stains
Cell membrane stains are best classified by workflow behavior rather than by color name alone. Two dyes may both appear to label the membrane, yet differ substantially in how fast they insert, how strongly they remain associated with the plasma membrane during imaging, how much intracellular redistribution appears over time, and how they behave after washing or fixation. In practice, researchers select membrane stains because they need a certain kind of boundary readout, not merely because they need fluorescence at the cell periphery. For this reason, membrane stain classification should begin with localization behavior, retention logic, live-cell compatibility, fixation sensitivity, and spectral placement. Once those points are clear, specific dye examples become much easier to position in a real workflow.
General Membrane Dyes
General membrane dyes are typically chosen when the primary goal is straightforward visualization of cell outline and membrane-associated contour. These dyes often rely on lipophilic interaction with the lipid bilayer and are favored in workflows where researchers want a practical structural channel rather than a highly specialized membrane readout. Examples from this product set include CellHunt CM-DiI and Speed DiI, both of which fit the broader logic of lipophilic membrane-labeling reagents used for contour-oriented imaging. Cy3 dic18 and Cy5 dic18 are also relevant because their diacyl or dialkyl membrane-anchoring design can support membrane-associated localization while placing signal in useful spectral regions for multicolor workflows. In broader fluorescent chemistry terms, membrane-staining discussions also frequently overlap with lipophilic dye families connected to rhodamine-type or carbocyanine-type design logic. The advantage of general membrane dyes is that they often provide a direct route to boundary visualization; the limitation is that signal quality can still depend heavily on sample handling, incubation timing, and whether the assay requires only short-term membrane localization or longer interpretation windows.
Live-Cell Membrane Probes
Live-cell membrane probes are selected when membrane staining must occur in intact cells under conditions compatible with immediate or short-duration imaging. Their value lies in providing usable membrane fluorescence without forcing the experiment into a fully fixed endpoint workflow. For example, CellHunt CM-DiI, Speed DiI, DiOC16(3), and 3,3'-Dipropyloxacarbocyanine iodide can all be discussed within a live-cell membrane imaging context because they are relevant to membrane-associated fluorescence design and can be used to support boundary visualization or surface-associated labeling during the chosen imaging window. The critical point, however, is not simply that a dye can be used in live cells. Researchers also need to know whether it maintains boundary clarity long enough for the planned acquisition, whether it gradually produces intracellular puncta, and whether the membrane signal remains analytically trustworthy as imaging proceeds. Live-cell membrane probes are therefore most valuable when the staining window, imaging timing, and boundary readout are defined in advance rather than assumed after the fact.
Fixation-Sensitive Membrane Stains
Fixation-sensitive membrane stains are probes whose localization pattern or interpretive meaning may change after fixation, permeabilization, or multistep downstream processing. This does not automatically make them poor membrane stains. It means their value must be judged within the actual handling sequence of the experiment. A membrane dye that gives a strong and convincing live-cell outline can become less reliable as a boundary marker if subsequent steps redistribute signal, alter membrane integrity, or reduce the contrast between true plasma membrane and surrounding regions. Octadecyl Rhodamine B Chloride is a useful example of a lipophilic fluorescent reagent that may be relevant when red-channel membrane-associated labeling is desired, but the ultimate value of such a stain still depends on how well it preserves meaningful boundary localization through the chosen workflow. In experiments that may transition between living samples and processed endpoints, membrane stains should always be evaluated with a live vs fixed staining mindset rather than treated as universally interchangeable.
Need a membrane staining strategy that fits your imaging workflow?
We can help align membrane probe selection, boundary quality, and channel design with your live-cell, endpoint, or multicolor imaging goals.
Choosing Membrane Stains for Different Readouts
The right membrane stain depends on what the membrane channel is supposed to tell you. This is the most important principle in membrane stain selection. Researchers often ask which membrane fluorescent dye is best, but the more useful question is which dye behavior best supports the intended readout. A stain chosen for clean cell boundary definition may not be the same as one chosen for multicolor panel design, short live imaging, or segmentation-oriented analysis. The decision should therefore begin with the imaging objective, followed by sample state, expected acquisition window, and only then by spectral channel preference. This logic reduces the common mistake of choosing a membrane stain based on visual brightness alone.
Choosing Stains for Cell Boundary Definition
If the main goal is to define cell boundaries clearly, the preferred membrane stain is one that produces continuous edge labeling with limited diffuse background and minimal ambiguity between adjacent cells. In this setting, dyes such as CellHunt CM-DiI or Speed DiI may be attractive because they align with the broader use of lipophilic membrane markers for cell contour visualization. The real selection criteria, however, are boundary continuity, membrane-to-background contrast, and the ability to separate neighboring cells rather than raw signal intensity alone. A boundary-focused stain should help reveal where the plasma membrane lies, not just generate strong fluorescence somewhere near the edge.
Choosing Stains for Morphology-Focused Imaging
In morphology-focused imaging, the membrane channel is often used to compare spreading, rounding, protrusion formation, perimeter irregularity, or edge collapse between conditions. This places greater importance on whether the stain preserves a believable contour of the cell rather than whether it produces the brightest image. Dyes that overaccumulate, broaden the apparent edge, or quickly generate intracellular signal can make cells look thicker, more diffuse, or more punctate than they truly are. When morphology is the priority, membrane stains should therefore be judged by shape fidelity and interpretive stability. A moderate but clean edge is often more useful than an intense signal that visually distorts the boundary.
Choosing Stains for Short Live Imaging vs Endpoint Analysis
Short live imaging and endpoint analysis place different demands on membrane stains. In short live imaging, the stain should provide membrane-associated fluorescence quickly and remain interpretable during the actual acquisition window. DiOC16(3) and 3,3'-Dipropyloxacarbocyanine iodide, for example, are relevant to discussions of membrane-associated live-cell fluorescence, but their usefulness still depends on timing, wash strategy, and how strictly the experiment depends on plasma membrane-specific interpretation. In endpoint analysis, especially where processing may be more extensive, retention behavior and post-handling interpretability become more important. A dye that works well during immediate live observation may not preserve the same meaning later. This is why membrane stain choice should be tied closely to whether the assay is fundamentally live, fixed, or mixed in design.
Choosing Stains for Segmentation-Oriented Workflows
Segmentation-oriented workflows require more than a visually pleasing membrane image. The membrane stain must support consistent boundary extraction across cells, fields, and experimental groups. This means researchers should look for membrane labeling that is reasonably uniform, sufficiently separated from background, and not excessively sensitive to minor workflow fluctuations. A stain that produces uneven retention, rapid punctate redistribution, or strong edge blooming may be difficult to use for downstream analysis even when it appears bright in representative images. In segmentation-driven assays, the best membrane fluorescent dye is usually the one that preserves boundary logic under standardized imaging settings, not the one that creates the most dramatic color contrast.
How Membrane Staining Fits into Multicolor Imaging?
Membrane staining often functions as a spatial reference layer within multicolor imaging rather than as the main biological endpoint. That role is extremely valuable because it helps anchor the interpretation of all the other channels in the experiment. A nucleus channel may define cellular occupancy, but the membrane channel defines the outer structural frame. Organelle or intracellular markers may show where a signal exists, but membrane staining helps indicate where that signal lies relative to the cell edge, neighboring cells, or contact regions. For this reason, membrane staining should be planned as part of the full spectral and interpretive architecture of the assay instead of being added at the end as an afterthought.
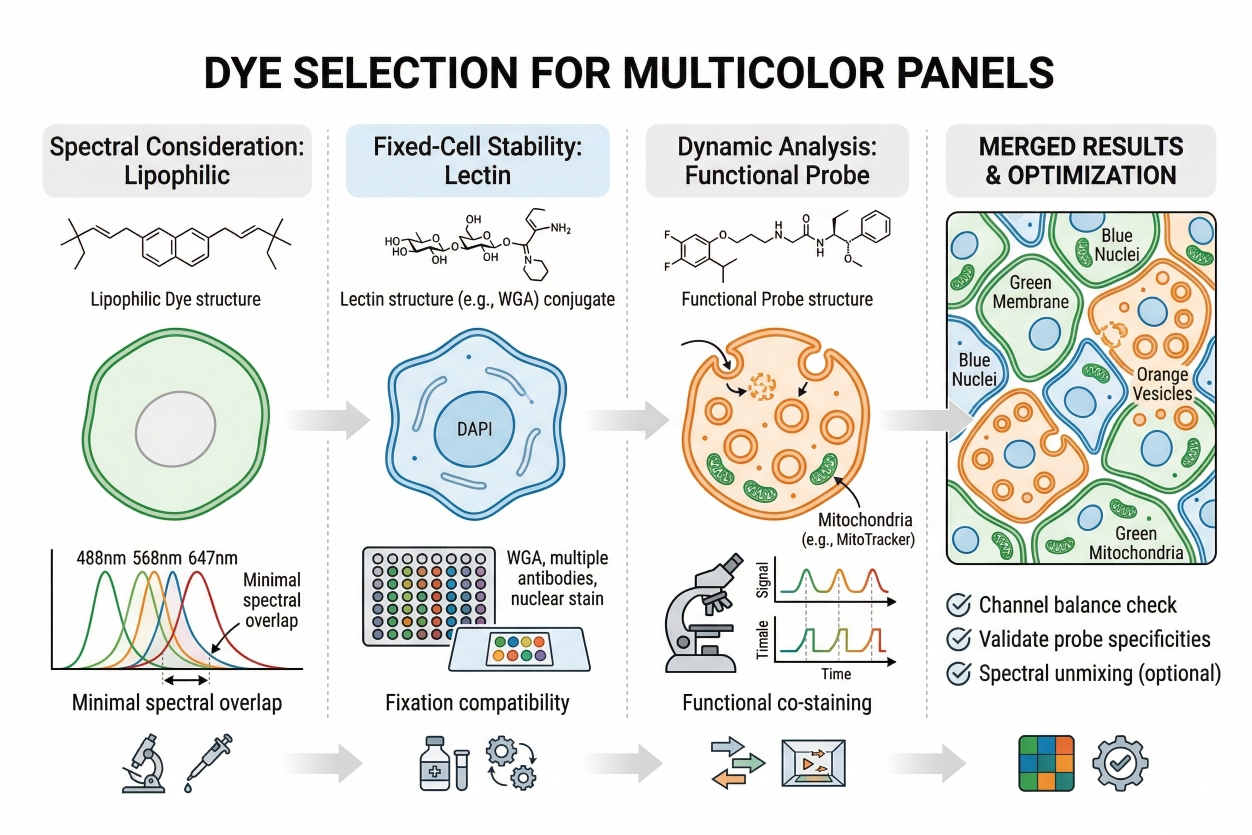 Fig. 2. Membrane stains should be selected with the full spectral panel in mind (BOC Sciences Authorized).
Fig. 2. Membrane stains should be selected with the full spectral panel in mind (BOC Sciences Authorized).
- Why Membrane Stains Often Serve as a Spatial Reference Channel: In multicolor imaging, membrane stains often serve as a spatial reference channel because they define the external contour against which internal fluorescent patterns can be interpreted. This becomes especially useful when signals from nuclei, vesicles, mitochondria, or reporter proteins need to be positioned within a whole-cell context. Without a membrane-associated boundary channel, it can be difficult to determine whether a signal is peripheral, central, clustered near contact zones, or extending into cell protrusions. A membrane stain therefore adds interpretive geometry to the image. This is also why membrane labeling can complement cell tracking workflows when short-term spatial behavior or boundary movement needs to be appreciated together with cell position.
- Pairing Membrane Stains with Nuclear and Organelle Dyes: Membrane stains pair naturally with nuclear dyes because the two channels together provide a much stronger basis for identifying single cells than either channel alone. The nucleus marks a central positional element, while the membrane defines the cell perimeter. This pairing becomes even more informative when organelle dyes are added, because intracellular structures can then be interpreted relative to the whole-cell outline rather than as isolated fluorescent objects. In practical panel design, a membrane dye such as Cy3 dic18 or Cy5 dic18 may be useful when the membrane channel needs to sit in a specific spectral region while leaving other colors available for nuclei or organelles. The best pairing strategy depends on which channels are already occupied, which readouts are most important, and whether membrane fluorescence is expected to serve mainly as a visual frame or as a source of analytical boundaries.
- Avoiding Spectral Conflict and Signal Imbalance: A major challenge in multicolor membrane staining is avoiding spectral conflict and signal imbalance. A membrane dye can be so bright that it compresses the dynamic range of weaker channels, creates spillover into nearby channels, or dominates the merged image to the point that other features become harder to evaluate. This is why the membrane channel should not automatically occupy the most convenient or brightest spectral position. For example, Cy5 dic18 may be attractive when longer-wavelength placement helps relieve crowding in visible channels, whereas Cy3 dic18 may be more useful when a mid-range channel remains available and the rest of the panel is balanced accordingly. Sulfo-Cy5 Azide belongs to a different practical category because it is more relevant to fluorescent conjugation or labeling strategy discussions than to routine direct membrane outlining, but its presence in the product set still reinforces the broader point that channel planning should begin with assay architecture rather than with dye popularity.
- Designing Membrane Staining into a Full Imaging Panel: Designing membrane staining into a full imaging panel means deciding early how much weight the membrane channel should carry in the interpretation. If membrane fluorescence is critical for segmentation or morphology, it deserves a cleaner channel and stricter optimization. If it functions mainly as a contextual outline, a somewhat less dominant placement may be acceptable as long as the cell edge remains interpretable. Researchers should also consider whether the membrane stain will be imaged briefly or repeatedly, whether the sample will remain alive throughout acquisition, and whether the full workflow may later require the kind of structured troubleshooting discussed in a broader cell staining troubleshooting guide. In good panel design, membrane staining is not chosen last. It is integrated from the beginning as part of the full imaging strategy.
Workflow Considerations for Clean Cell Boundaries
Clean membrane boundaries do not arise from dye choice alone. They depend on the relationship between cell condition, plating geometry, dye loading behavior, washing steps, imaging timing, and the interpretive demands of the assay. Many membrane staining problems that appear to be reagent failures are actually workflow mismatches. A stain can be chemically suitable for plasma membrane labeling and still perform poorly if cells are overconfluent, washes are poorly timed, or imaging is delayed long enough for redistribution to become significant. The most reliable membrane staining workflows therefore treat boundary quality as a systems outcome rather than as a single-parameter problem.
- Cell Condition and Plating Density Before Staining: The condition of the cells before staining strongly influences whether membrane boundaries can be interpreted cleanly. Cells that are unhealthy, poorly attached, highly stressed, or excessively confluent often yield membrane images that are harder to interpret, regardless of dye identity. In very dense cultures, adjacent plasma membranes may lie too close together to provide comfortable separation unless the stain gives exceptionally clean contrast. In overly sparse cultures, individual cells may show edge behaviors that are not representative of the intended biological state. Membrane staining works best when plating density supports the actual imaging objective, whether that is morphology comparison, contact analysis, or segmentation.
- Incubation and Washing Variables That Affect Membrane Clarity: Incubation time, dye concentration, loading medium, and washing intensity all influence the balance between membrane signal and unwanted background. Excessive loading can increase diffuse fluorescence, broaden the apparent edge, or leave residual extracellular background that compromises boundary sharpness. Insufficient loading can make the membrane appear broken or too weak for reliable use. The wash step is equally important. Inadequate washing can leave a bright haze around cells, while overly aggressive washing may reduce the practical retention of some membrane-associated dyes. For reagents such as CellHunt CM-DiI, Speed DiI, DiOC16(3), or related lipophilic probes, small workflow differences can shift the image from a crisp edge to a compromised outline. Optimization should therefore focus on membrane clarity, not simply on maximizing signal.
- Timing, Internalization, and Imaging Window: Timing is one of the most underestimated variables in membrane staining. A dye that looks convincingly membrane-localized shortly after loading may become less useful if imaging is delayed and intracellular puncta begin to appear. This is especially relevant for live-cell membrane probes and lipophilic dyes that can undergo time-dependent redistribution. Researchers should determine not only whether a stain labels the membrane, but for how long it provides the specific kind of boundary information the experiment requires. If the main readout depends on clean cell perimeters, the best imaging window may be narrower than expected. Early pilot imaging focused on time-course behavior is often more informative than a simple one-time intensity check.
- When Fixation or Downstream Staining Changes Interpretation: Fixation, permeabilization, and downstream staining can change the meaning of a membrane signal even when fluorescence remains visible. This is one of the main reasons membrane stains should not be treated as universally transferable across workflows. A probe that defines the plasma membrane well in live cells may become partially redistributed or less boundary-specific after additional processing. In workflows where membrane staining will be followed by other labels or handling steps, it is essential to evaluate not only whether fluorescence survives, but whether the signal still corresponds to the biological boundary being interpreted. This is particularly important in mixed workflows that combine membrane staining with later processing steps, where a live vs fixed staining perspective helps prevent overconfidence in visually preserved but functionally altered signal patterns.
Looking for a membrane stain that works with your full fluorescence panel?
We can help compare membrane labeling options based on boundary clarity, workflow compatibility, and spectral fit for your imaging setup.
Common Problems in Membrane Staining
Troubleshooting membrane staining requires paying attention to both image appearance and signal meaning. Some failures are obvious, such as weak fluorescence or heavy background. Others are more subtle, such as membrane-associated signal that gradually becomes punctate, or boundaries that look present but are too broad or inconsistent for analysis. The goal of troubleshooting is therefore not only to improve image quality but also to protect the interpretive value of the membrane channel. In practice, most membrane staining problems can be understood by asking whether the issue stems from insufficient membrane association, excessive non-boundary fluorescence, time-dependent redistribution, processing-induced change, or spectral interference within the larger imaging panel.
- Weak or Discontinuous Membrane Signal: Weak or discontinuous membrane signal often appears as broken cell outlines, partial labeling, or edge fluorescence that is too dim to support confident interpretation. Common causes include insufficient dye loading, poor cell condition, overly brief incubation, inappropriate imaging settings, or an assay design that places too much interpretive burden on a marginal membrane channel. In some cases, the chosen dye may simply not provide enough practical membrane contrast for the specific cell type or plating density. The first response should be to examine whether the problem is related to biology, loading conditions, or acquisition settings before concluding that the dye itself is unsuitable.
- High Background Outside the Cell Boundary: High background outside the cell boundary can make the entire field appear hazy and reduce the contrast needed for clean membrane interpretation. This problem may arise from excessive dye concentration, incomplete washing, medium-associated interference, serum effects, or a probe whose membrane association is less selective under the chosen conditions. Background is particularly damaging in boundary-focused workflows because it makes the edge appear thicker and less precise than it truly is. Optimization should therefore aim to reduce diffuse fluorescence while preserving enough membrane signal to maintain a believable outline. A lower but cleaner edge is usually preferable to a stronger signal embedded in haze.
- Punctate Intracellular Signal After Membrane Staining: Punctate intracellular signal after membrane staining often indicates that the fluorescent label is no longer functioning purely as a plasma membrane outline. Depending on the workflow, this may reflect internalization, time-dependent redistribution, vesicle-associated signal, or the natural behavior of the membrane-associated probe under prolonged live conditions. This does not always mean the stain has failed, but it does mean the membrane channel may now be reporting something different from a clean cell boundary. If boundary interpretation remains the main goal, the solution is usually to optimize the imaging window, shorten post-stain delay, or select a dye whose practical behavior better matches the assay timeframe.
- Boundary Changes After Fixation or Multistep Processing: Boundary changes after fixation or multistep processing are among the most common interpretive pitfalls in membrane staining. Researchers may still see fluorescence after fixation and assume the stain remains a reliable membrane marker, when in fact the spatial meaning has shifted. Signal can broaden, weaken, redistribute, or become less specific to the outer boundary after additional handling. These problems are especially important when the membrane channel is expected to support morphology analysis or segmentation after the full workflow is complete. The safest approach is to validate the exact sequence of staining, fixation, washing, and imaging rather than assuming that visible fluorescence guarantees preserved membrane logic.
- Crosstalk and Overexposure in Multicolor Imaging: Crosstalk and overexposure can make membrane signals appear less boundary-specific than they really are. When a membrane dye is too bright or placed too close spectrally to another fluorophore, the resulting image may show bleed-through, edge blooming, or distorted overlap that complicates interpretation. This is particularly relevant in panels using red and far-red membrane-associated dyes such as Cy3 dic18, Cy5 dic18, or Octadecyl Rhodamine B Chloride together with other structural markers. Solving the problem may require lower exposure, adjusted gain, different panel balance, or a more disciplined optimization strategy like those discussed in a broader cell staining troubleshooting guide. The key point is that some apparent membrane problems are actually panel-design problems.
Application Scenarios from Morphology to Cell Interaction Studies
The value of membrane staining becomes much clearer when it is discussed from the perspective of what researchers are actually trying to achieve in cell imaging workflows. In practice, investigators rarely add a membrane channel simply to generate a more visually complete image. They use it because many research questions cannot be answered confidently without a reliable representation of the cell boundary. A nucleus channel may indicate where a cell is located, and intracellular markers may show where a target accumulates, but those signals alone often do not resolve how far a cell has spread, whether its edge has become irregular, whether neighboring cells can be separated accurately, or whether an intracellular readout sits near the periphery, at a contact zone, or within a protrusive region. Membrane staining therefore becomes important when the outer geometry of the cell is part of the interpretation rather than background context.
Membrane Staining for Morphology Assessment
Researchers frequently use membrane staining when the central question involves whole-cell morphology rather than only intracellular signal distribution. This includes experiments comparing cell spreading, rounding, elongation, protrusion formation, perimeter irregularity, edge collapse, or treatment-associated changes in outer contour. In these workflows, the membrane channel provides something that nuclear staining cannot: a direct representation of the actual external geometry of the cell. That becomes particularly important when morphology itself is the readout, because a nucleus may remain relatively unchanged while the cell perimeter shifts substantially. The practical research need here is not just to see an outline, but to preserve enough boundary fidelity that differences between groups can be interpreted as real morphological changes rather than staining artifacts. For morphology-focused imaging, membrane staining is most useful when it captures edge structure cleanly enough to support side-by-side comparison under matched acquisition settings.
Membrane Staining in Dense Cultures and Boundary Segmentation
In dense cultures, confluent monolayers, or assays with strong cell-cell proximity, membrane staining becomes highly relevant because nuclei alone often do not provide enough information to assign accurate cell boundaries. Researchers in these settings commonly need to separate neighboring cells for segmentation, quantify cell area or perimeter, or generate object-level measurements across large image sets. The difficulty is that close cell packing can make boundaries visually ambiguous even when cells are clearly present. A membrane channel helps solve this by adding perimeter-level information that supports more reliable cell separation. The key research demand is consistency: the stain must define borders well enough that segmentation remains usable across replicates, treatment groups, and image fields rather than working only in selected examples. For segmentation-oriented workflows, membrane staining is valuable not because it is visually striking, but because it can turn crowded images into analytically tractable data.
Membrane Staining in Multicolor Spatial Analysis
Many researchers include membrane staining in multicolor assays because other fluorescent readouts become much more informative when interpreted within the full outline of the cell. A nuclear marker can identify occupancy, and organelle markers can show intracellular localization, but those signals alone may not reveal whether a target sits close to the periphery, is polarized to one side of the cell, accumulates near a contact region, or remains broadly distributed throughout the interior. Membrane staining addresses this need by supplying an external structural frame that organizes the meaning of the rest of the panel. This is especially useful when the research goal is not simply to detect multiple targets, but to understand how those targets are arranged spatially within each cell. In that context, the membrane channel functions as a contextual reference that improves the interpretability of multicolor imaging and helps researchers move from "what signals are present" to "how those signals are positioned within the cellular architecture."
Membrane Staining for Cell-Cell Interaction Studies
In studies involving cell-cell proximity, contact interfaces, clustering, or local community organization, membrane staining is often selected because the biologically relevant features are expressed at or near the cell boundary. Researchers may need to determine whether neighboring cells remain clearly separated, whether contact zones appear broadened or compressed, whether treatment changes local packing behavior, or whether interface geometry differs across conditions. These questions cannot be answered well from nuclei alone, because the research interest lies in how cell surfaces relate to one another rather than simply where cell centers are positioned. A membrane channel therefore becomes part of the primary evidence rather than a supplementary label. For this application, the practical demand on the stain is high: it must preserve interpretable edge definition at the interface between adjacent cells without excessive haze, signal spread, or redistribution that would obscure boundary-level relationships. When cell-cell interaction is the focus, membrane staining helps researchers evaluate not just occupancy, but organization at the level where cells actually meet.
Membrane Staining in Short-Term Live Observation
Short-term live imaging is another major application area because researchers often need to observe dynamic boundary behavior without immediately converting the assay into a fixed endpoint workflow. This may include monitoring contour stability, peripheral reshaping, local edge movement, short-term migration-related changes, or treatment-associated alterations in cell outline during a defined acquisition window. The research need in these experiments is time-resolved boundary information that remains interpretable long enough to answer the specific live-cell question. That requirement changes how membrane stains should be judged. The best probe is not necessarily the brightest, but the one that remains sufficiently boundary-focused during the actual period of observation. In live workflows, membrane staining is useful when it allows researchers to capture dynamic structural information while limiting confusion from time-dependent internalization, diffuse background, or rapid loss of edge specificity. In other words, its value lies in supporting real-time interpretation of cell boundary behavior, not just in labeling the membrane at a single time point.
How BOC Sciences Supports Membrane Staining?
BOC Sciences supports membrane staining projects through integrated capabilities in fluorescent product supply, custom probe development, staining workflow optimization, and technical service support. Rather than treating membrane staining as a simple dye-selection step, we help researchers align probe type, membrane-labeling behavior, spectral placement, and imaging conditions with the actual goals of the experiment. This is especially important in membrane staining workflows, where boundary sharpness, retention behavior, live-cell compatibility, and multicolor panel design all influence whether the membrane channel remains truly useful. Our support is therefore designed to cover both routine membrane visualization needs and more specialized assay development applications.
Membrane Fluorescent Dye and Probe Supply
- Access to a broad range of membrane-associated fluorescent dyes and probes suitable for plasma membrane outlining, cell boundary visualization, morphology-oriented imaging, and short live-cell observation workflows.
- Support for membrane probe selection across multiple spectral regions, including green, red, and far-red fluorescence, to better fit multicolor staining and imaging experiments.
- Availability of membrane-labeling products with different retention and staining behaviors, helping researchers choose more suitable reagents for live-cell imaging, endpoint analysis, or structurally focused membrane visualization.
- Product support spanning diverse membrane-related fluorescent formats, including lipophilic membrane dyes, fluorophore-conjugated membrane labels, and complementary imaging reagents adapted to different assay needs.
Customized Membrane Probe Development
- Custom design of membrane-targeted fluorescent probes according to specific imaging goals, cell models, and desired boundary-labeling performance.
- Structural modification support for fluorescent dyes, including adjustment of hydrophobicity, membrane affinity, linker design, and charge properties to improve membrane localization and signal behavior.
- Development support for functional membrane-related probes when membrane visualization needs to be integrated with additional responsive or assay-specific fluorescence logic.
- One-stop development services covering molecular design, synthesis, modification, purification, and characterization for customized membrane-labeling fluorescent probes.
Membrane Staining Workflow Optimization and Application Support
- Practical guidance for optimizing membrane staining conditions, including dye concentration, incubation duration, washing strategy, imaging timing, and handling conditions that affect boundary clarity.
- Application support for different experimental systems, including adherent cells, suspension cells, dense cultures, and short live-cell observation setups, with attention to how sample state influences membrane signal quality.
- Suggestions for multicolor staining strategies involving membrane labeling together with nuclei, mitochondria, lysosomes, or other intracellular reference channels.
- Troubleshooting assistance for common membrane staining challenges, including weak boundary definition, excessive background, dye redistribution, over-labeling, or unclear separation between adjacent cells.
Quality Control and Technical Service Support
- Supply of high-purity membrane fluorescent products supported by strict quality control processes, with analytical methods such as HPLC, NMR, and MS used where applicable.
- Attention to batch-to-batch consistency and product stability to support reproducibility across repeated membrane staining and imaging experiments.
- Availability of complete technical documentation, including COA, MSDS, and product application information, to support evaluation, workflow planning, and implementation.
- Access to professional technical support for both pre-sale consultation and post-sale troubleshooting, helping researchers address issues from probe selection to membrane imaging optimization.
Do You Need A Consultation?
BOC Sciences integrates cutting-edge fluorescence technologies to accelerate your research, driving next-generation solutions for drug discovery and diagnostics.
Transform Your Studies with Cutting-Edge Fluorescent Products
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0036 | Calcein Blue | 54375-47-2 | Bulk Inquiry |
| A16-0089 | CellHunt CM-DiI | 180854-97-1 | Bulk Inquiry |
| A16-0082 | DiOC16(3) | 161433-32-1 | Bulk Inquiry |
| A16-0156 | Speed DiI | 278173-35-6 | Bulk Inquiry |
| A16-0035 | Calcein Blue AM | 168482-84-6 | Bulk Inquiry |
| A16-0163 | 3,3'-Dipropyloxacarbocyanine iodide | 53213-79-9 | Bulk Inquiry |
| A16-0171 | Octadecyl Rhodamine B Chloride | 65603-19-2 | Bulk Inquiry |
| F02-0136 | Sulfo-Cy5 Azide | 1481447-40-8 | Bulk Inquiry |
| F02-0108 | Cy3 dic18 | 22366-93-4 | Bulk Inquiry |
| F02-0115 | Cy5 dic18 | 75539-51-4 | Bulk Inquiry |
High-Performance Fluorescent Tools for Your Research
- Ion Fluorescent Probes Indicators for real-time ion concentration imaging.
- Calcium, Chloride and Other indicators Fluorescent indicators for intracellular ion flux monitoring.
- Apoptosis Fluorescent Probes Probes detecting programmed cell death events.
- Endoplasmic Reticulum Fluorescent Probes ER-targeted dyes for organelle structure analysis.
- Cell membrane Fluorescent Probes Surface-labeling dyes for membrane dynamics studies.
- pH Indicators Fluorescent sensors for intracellular pH monitoring.
- Golgi Fluorescent Probes Targeted probes for Golgi apparatus visualization.
- Cell Proliferation Tracer Fluorescent Probes Long-term tracking of cell division processes.
Explore More Topics
Online Inquiry


