Cytoskeleton Staining: Principles, Probe Selection, and Workflow Design
Cell staining of the cytoskeleton is one of the most informative approaches in fluorescence-based cell analysis because the cytoskeleton is not a single decorative structure. It is the architectural framework that helps define cell shape, intracellular organization, force distribution, surface protrusions, division-related remodeling, and how cells respond to their local environment. In many experiments, researchers are not simply trying to make filaments visible. They want to understand whether actin remains organized into stress fibers, whether cortical actin becomes redistributed, whether microtubules retain a continuous radial network, whether spindle-like structures appear during mitosis, and whether those structural changes are robust enough to support side-by-side interpretation across treatment groups. That is why cytoskeleton staining needs to be planned as a readout strategy rather than treated as a routine add-on channel.
What Is Cytoskeleton Staining?
Cytoskeleton staining refers to the use of cytoskeleton fluorescent probes, labeled binding reagents, antibodies, or genetically encoded markers to visualize structural elements of the cellular framework under fluorescence imaging. In most research workflows, this means focusing on actin filaments, microtubules, or the broader spatial architecture formed by cytoskeleton-associated structures. Cytoskeleton staining is not typically performed for appearance alone. It is used because the cytoskeleton provides a high-value readout for cell morphology, adhesion, motility-related organization, intracellular tension patterns, mitotic remodeling, and treatment-dependent phenotypic change. For this reason, cytoskeleton staining is most useful when researchers first define whether the primary objective is to visualize F-actin, microtubule organization, or a broader structural context that supports comparison across samples.
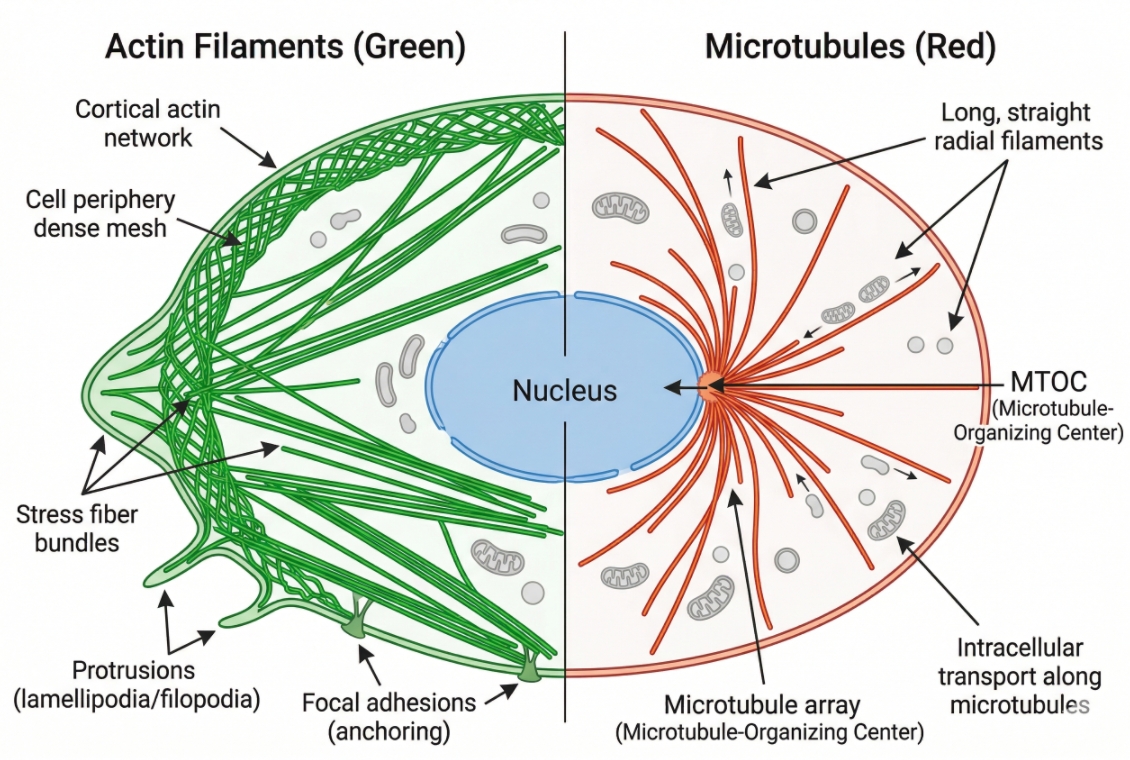 Fig. 1. Actin and microtubules provide distinct structural readouts (BOC Sciences Authorized).
Fig. 1. Actin and microtubules provide distinct structural readouts (BOC Sciences Authorized).
In practical imaging workflows, cytoskeleton staining often becomes most valuable when it is integrated into broader cell imaging and multicolor analysis panels. A cytoskeleton channel can provide the spatial framework that makes other intracellular channels easier to interpret, especially when researchers need to compare cellular organization rather than fluorescence intensity alone. This is also why cytoskeleton staining fits naturally within broader organelle cell staining workflows. When combined with nuclear staining, cell membrane staining, mitochondrial staining, Golgi fluorescent staining, endoplasmic reticulum staining, or lysosome staining, the cytoskeleton channel can help explain whether intracellular organization remains coordinated or becomes visibly altered.
What cytoskeleton staining is designed to visualize?
Cytoskeleton staining is designed to reveal more than the presence of intracellular fibers. In actin-focused experiments, it can show stress fibers, cortical organization, lamellipodia-associated structure, cell edge remodeling, and changes in spreading behavior. In microtubule-oriented imaging, it can show radial networks, local disorganization, spindle-associated patterns, and the relationship between microtubule structure and intracellular positioning. In multiparameter assays, a cytoskeleton channel can provide the organizing framework that helps users understand where organelles, nuclei, and membrane signals sit relative to the overall cellular architecture. The value of this readout depends on using a staining method that matches the structural question being asked rather than assuming that all cytoskeleton probes behave similarly.
Why the cytoskeleton is not a single staining target?
The term cytoskeleton sounds unified, but the actual staining targets are not interchangeable. F-actin probes, phalloidin conjugates, tubulin trackers, antibody markers, and fluorescent protein constructs operate through different mechanisms and support different kinds of interpretation. A workflow optimized for actin-rich structures is not automatically suitable for microtubule imaging, and a live-cell microtubule stain is not equivalent to a fixed-cell phalloidin labeling strategy. This is why users should not start with the broad question of which cytoskeleton probe is best. The more useful question is whether the assay needs to emphasize actin architecture, microtubule organization, dynamic live-cell observation, fixed-cell endpoint comparison, or multicolor structural context.
What this readout can and cannot tell you?
Cytoskeleton staining can provide strong structural evidence when the probe, sample handling method, and imaging settings are well aligned. It can reveal whether actin fibers appear organized or diffuse, whether microtubules form a coherent network or seem disrupted, and whether morphology changes between control and treated groups are accompanied by visible structural differences. At the same time, staining alone usually cannot establish the underlying mechanism responsible for those changes. Altered signal can reflect genuine biological remodeling, but it can also reflect fixation sensitivity, probe loading conditions, background imbalance, or timing-related artifacts. Reliable cytoskeleton interpretation therefore requires researchers to treat the staining method as part of the analytical logic rather than as a neutral visualization step.
Need help selecting the right cytoskeleton fluorescent probes?
We can help compare actin probes, tubulin trackers, fixed-cell markers, and multicolor channel options for your cytoskeleton staining workflow.
Why Cytoskeleton Staining Matters in Cell Analysis?
Cytoskeleton staining matters because the cytoskeleton is one of the clearest structural readouts available in fluorescence microscopy. It helps explain how cells maintain shape, spread on a surface, reorganize under stress, polarize during migration, or change architecture during division and treatment response. In practical assay development, the cytoskeleton channel is often not the final biological answer, but it is frequently the channel that makes the rest of the image interpretable. That is why cytoskeleton staining plays a central role in morphology studies, phenotypic imaging, and multicolor structural analysis.
- Cytoskeleton staining as a structural readout: Actin filaments and microtubules provide direct visual information about cell shape, internal support, spatial tension patterns, edge activity, and network continuity. A well-resolved cytoskeleton channel often reveals whether a cell is spread, contracted, polarized, rounded, dividing, or structurally stressed even before downstream markers are analyzed in detail.
- Why cytoskeleton imaging matters in morphology and phenotypic comparison: In comparative experiments, morphology shifts are often among the earliest visible indicators of treatment effects or environmental response. Cytoskeleton staining gives researchers a way to assess whether those changes correspond to reorganization of actin structure, loss of filament continuity, altered cortical arrangement, or microtubule redistribution under matched imaging settings.
- The value of cytoskeleton staining in multicolor cell imaging: Cytoskeleton staining becomes especially informative when combined with nuclear, membrane, mitochondrial, Golgi, ER, or lysosomal channels because it provides the structural framework within which those signals are distributed. In such panels, the cytoskeleton is not just another fluorescent color. It is the spatial layer that helps users determine whether the broader intracellular organization remains coordinated or appears altered.
- Why cytoskeleton signals are often interpretation-sensitive: Cytoskeleton structure can be strongly affected by fixation, permeabilization, mechanical handling, cell confluence, drug exposure, and imaging timing. This means a visually attractive signal is not always a biologically trustworthy one. The more the experiment relies on structural comparison, the more important it becomes to standardize the workflow that generated the image.
- How cytoskeleton staining supports higher-content imaging logic: In screening and multi-feature image analysis, the cytoskeleton channel often improves segmentation, morphology classification, and structure-aware interpretation. Even when the primary assay endpoint is not "cytoskeleton biology," a well-designed cytoskeleton stain can substantially improve the interpretability of the overall image set.
Core Principles Behind Cytoskeleton Fluorescent Probes
The most important principle behind cytoskeleton fluorescent probes is that they do not all report structure in the same way. Some bind polymerized actin directly, some are designed for microtubule-associated imaging, some are best suited for fixed and permeabilized cells, and some are selected because they support live-cell visualization over a limited acquisition window. In practice, this means probe choice should be driven by structural target, sample state, and interpretation needs rather than by color alone.
- Phalloidin-based probes for F-actin staining: Phalloidin conjugates are among the most established tools for F-actin visualization because they bind filamentous actin with high affinity and can produce strong structural contrast in fixed-cell workflows. Products such as Phalloidin-FITC, Sulforhodamine 101-X Phalloidin, BF 350 Phalloidin, BF 488 Phalloidin, BF 568 Phalloidin, BF 594 Phalloidin, Biotin-XX phalloidin, Phalloidin-TFAX 488, and Phalloidin-TRITC illustrate how the same core targeting logic can be adapted to different spectral needs and downstream workflow designs.
- Tubulin probes for microtubule visualization: Microtubule-oriented probes are chosen when the goal is to observe tubulin-based networks, spindle-associated structures, or broader microtubule organization. In a practical product context, Tubulin Tracker Green represents a direct route to microtubule-focused staining, while broader structural tools may support microfilament or filament-associated readouts depending on the experimental design.
- Antibody-based staining vs direct fluorescent probes: Direct fluorescent probes can simplify the workflow and reduce the number of staining steps, especially when structural visualization is the main objective. Antibody-based methods, by contrast, are often preferred when users need protein-level specificity, expanded multiplexing logic, or fixed-cell endpoint analysis where structure must be interpreted together with other immunofluorescent markers.
- Live-cell cytoskeleton probes vs fixed-cell staining strategies: Live-cell probes are typically selected for dynamic imaging, repeated observation, or rapid structure-aware screening, but they must be evaluated for cell compatibility, staining stability, and potential perturbation of the system being observed. Fixed-cell strategies are often better when the goal is strong endpoint contrast, stable multicolor integration, and more controlled side-by-side comparison.
- Why spectral design matters as much as target specificity: A cytoskeleton stain is only useful if it integrates cleanly into the rest of the imaging panel. Selecting the correct spectral region is often just as important as selecting the correct structural target, particularly when the assay already includes strong nuclear, organelle, or membrane channels.
Common Types of Cytoskeleton Fluorescent Probes
Cytoskeleton probes are best organized by structural target and workflow role rather than by chemistry alone. This is the most useful approach for real experimental design because users are usually trying to choose between actin-oriented imaging, microtubule-focused observation, endpoint morphology comparison, or live-cell structural tracking.
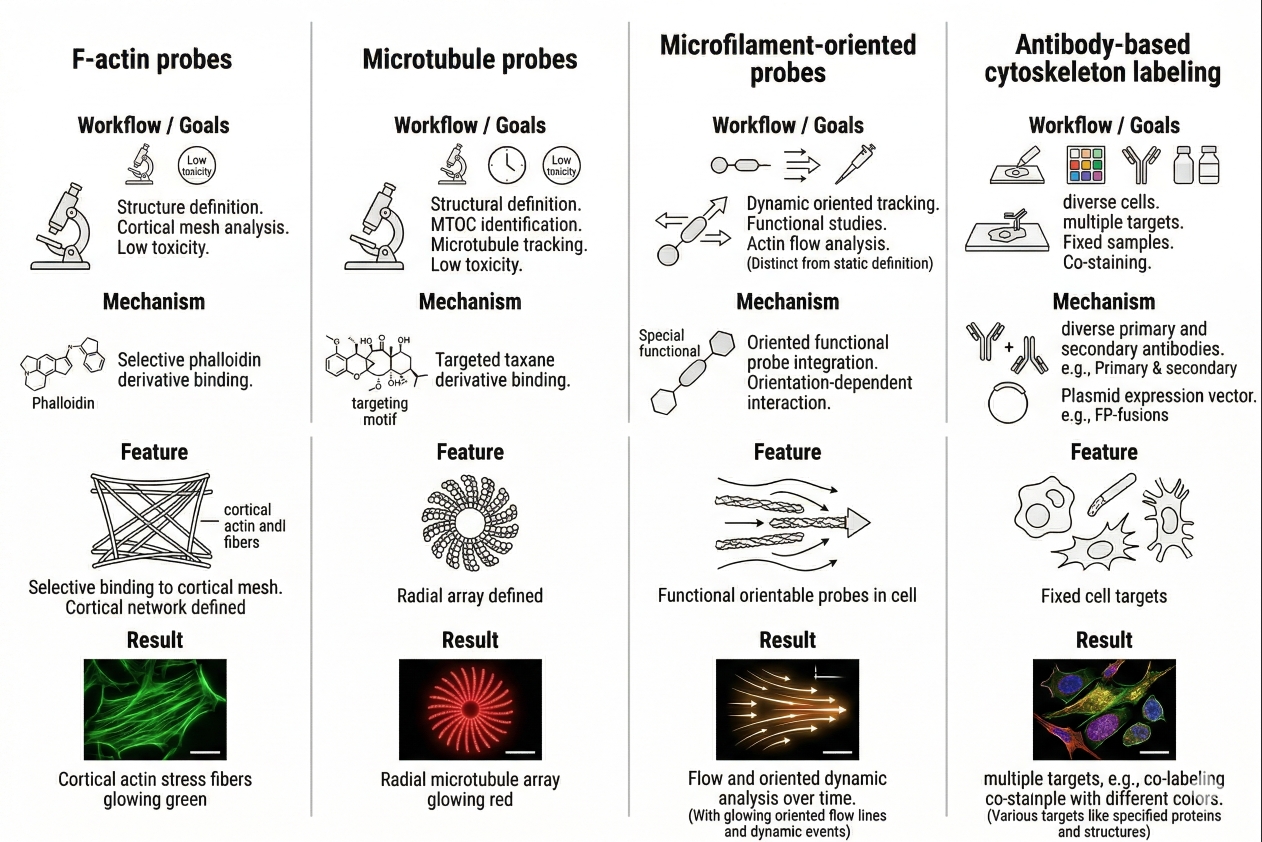 Fig. 2. Different cytoskeleton probe classes support different imaging goals (BOC Sciences Authorized).
Fig. 2. Different cytoskeleton probe classes support different imaging goals (BOC Sciences Authorized).
F-actin probes for fixed-cell structure imaging
F-actin probes are the natural choice when researchers need strong visualization of filamentous actin structure in fixed and permeabilized cells. In many morphology-oriented workflows, phalloidin conjugates remain the most practical route because they produce clear labeling of stress fibers, cortical actin, and other filament-rich structures under controlled endpoint conditions. The availability of multiple spectral variants gives users flexibility in panel planning. For example, BF 488 Phalloidin or Phalloidin-FITC may suit green-channel designs, while BF 568 Phalloidin, BF 594 Phalloidin, Sulforhodamine 101-X Phalloidin, and Phalloidin-TRITC may be more useful when the green channel is already occupied. These probes are particularly valuable when structural clarity and multicolor integration matter more than live-cell observation.
Microtubule probes for live-cell and endpoint imaging
Microtubule probes are selected when the experimental objective centers on tubulin-based architecture, microtubule continuity, intracellular radial structure, or spindle-related organization. Tubulin Tracker Green is a natural example in workflows that need a direct microtubule-oriented signal. Depending on the sample and readout design, microtubule imaging may be carried out in live cells or under endpoint conditions, but the staining logic should always reflect whether the goal is dynamic observation, structural mapping, or comparative morphology across groups. The best choice is not simply the brightest signal. It is the one that preserves meaningful microtubule interpretation under the real handling conditions of the experiment.
Microfilament-oriented probes for broader actin-associated structure
Some cytoskeleton workflows are less focused on classic stress-fiber imaging and more concerned with broader microfilament-related structure, cell edge organization, or rapid assessment of actin-associated morphology. Microfilament Tracker Green can fit this type of use case when users want a practical fluorescent entry point into microfilament-associated imaging rather than a heavily antibody-dependent endpoint workflow. As with all actin-related readouts, the value of the image depends on whether the staining method aligns with sample processing and whether the final signal supports the specific level of structural interpretation required.
Antibody-based and protein-based cytoskeleton labeling strategies
Antibody-based cytoskeleton staining becomes especially useful when researchers need protein-selective endpoint analysis or want to integrate cytoskeleton readouts into broader immunofluorescence panels. Fluorescent protein-based strategies may be more attractive in live-cell systems that require dynamic tracking, longer observation windows, or stable structural labeling within a compatible model. These methods are not universally better than direct probes; they are simply better matched to certain questions. A strong cytoskeleton workflow often comes from recognizing early whether the study needs a direct structural stain, a protein-selective readout, or a dynamic labeling strategy that can coexist with other channels over time.
How to Choose the Right Cytoskeleton Fluorescent Probes?
Choosing the right cytoskeleton fluorescent probe starts with deciding what the experiment actually needs to reveal. Researchers often ask which reagent is best, but the better question is which probe logic is most appropriate for the structural target, sample state, and imaging design. No single cytoskeleton probe is optimal for every workflow. A probe that performs well in fixed-cell F-actin imaging may be poorly matched to live microtubule observation, and a dye that seems visually bright may still be a weak choice if it creates channel burden or makes structural interpretation ambiguous.
Choose by target: actin, microtubules, or broader cytoskeletal context
The first decision is whether the assay is mainly about actin structure, microtubule organization, or a broader cytoskeletal framework that supports overall morphology analysis. If the main readout is stress fibers, cortical actin, or cell edge organization, phalloidin-based F-actin strategies are often the most direct route. If the readout centers on network continuity, spindle-associated structure, or intracellular tracks, a tubulin-focused probe may be more appropriate. If the aim is broader morphology classification, a microfilament-oriented or mixed structural approach may be sufficient depending on the imaging question.
Choose by sample state: fixed cells or live cells
Sample state is one of the strongest determinants of probe suitability. Fixed-cell workflows often support the best structural preservation for phalloidin-based F-actin imaging and can integrate smoothly with multicolor immunofluorescence panels. Live-cell workflows must place much greater emphasis on membrane permeability, acquisition timing, signal stability, and compatibility with ongoing cellular behavior. Researchers should decide early whether the cytoskeleton channel is intended as a dynamic observation tool or a controlled endpoint structure readout, because that choice usually determines the staining strategy more effectively than product popularity.
Choose by readout goal: morphology, dynamics, or high-content analysis
If the experiment is built around endpoint morphology comparison, the best probe is usually the one that produces clean structural contrast under highly standardized conditions. If the experiment depends on dynamics, repeated imaging, or time-dependent structure tracking, then live-cell-compatible strategies deserve stronger consideration. In higher-content workflows, probe selection should also account for segmentation quality, signal consistency, and whether the cytoskeleton channel can support structure-aware image analysis across many fields and batches.
Choose by spectral region and multicolor compatibility
Cytoskeleton staining is often part of a larger fluorescence panel, so spectral planning matters from the beginning. A green actin stain may be convenient unless the green channel is already needed for another high-priority marker. A red or orange phalloidin conjugate may be more practical in those cases, while blue-labeled options can help preserve visible channels for organelle-specific probes. This type of decision should be guided by the full panel rather than by the cytoskeleton channel in isolation. When broader fluorescence planning is needed, it is often helpful to compare the selection logic with how to choose the right fluorescent dye for cell staining principles before finalizing the panel.
Building a cytoskeleton staining workflow for actin or microtubules?
We can help align probe type, sample compatibility, spectral planning, and multicolor panel design for more reliable cytoskeleton imaging.
Practical Workflow for Cytoskeleton Staining in Cells
A good cytoskeleton staining workflow is not built by reagent choice alone. It depends on defining the structural target, matching sample preparation to probe mechanism, and controlling the handling steps that most strongly affect background, apparent continuity, and interpretive reliability. In many cases, cytoskeleton images fail not because the probe is inherently poor, but because the workflow was optimized for convenience rather than structure-preserving logic.
- Define the cytoskeletal target before selecting the probe: Researchers should decide whether the experiment is primarily about F-actin, microtubules, or a broader cytoskeletal context before comparing products. This avoids the common mistake of selecting a cytoskeleton reagent first and trying to force it into an unsuitable structural question.
- Match sample preparation to probe mechanism: Phalloidin-based actin probes generally fit fixed and permeabilized endpoint workflows, while tubulin trackers or other live-cell-oriented tools may require a more controlled loading-and-imaging sequence. Structural meaning depends on whether the sample handling method supports the probe's intended behavior.
- Control fixation and permeabilization carefully: Cytoskeletal architecture can change visibly if fixation is too harsh, too weak, delayed, or not well matched to the structure being examined. Over-permeabilization can reduce useful signal or disturb fine organization, while insufficient permeabilization can leave the stain incomplete or inconsistent across cells.
- Optimize incubation and wash conditions: Background, signal continuity, and apparent structural sharpness can all be influenced by incubation time and wash intensity. Aggressive washing may reduce useful signal or alter loosely attached cells, while inadequate washing can leave diffuse fluorescence that competes with real filament structure.
- Standardize acquisition settings for comparison: When the experiment compares structural organization across groups, exposure, gain, laser power, and image-processing thresholds should remain fixed. Cytoskeleton patterns are highly sensitive to contrast manipulation, so unstable acquisition settings can easily produce misleading visual differences.
- Use counterstains strategically: Pairing the cytoskeleton channel with nuclear or membrane reference stains often makes structural interpretation easier, especially when cell shape or intracellular orientation matters. In more complex panels, adding organelle channels can help clarify whether the observed cytoskeleton pattern supports or contradicts the rest of the intracellular architecture.
Common Problems in Cytoskeleton Staining and How to Avoid Them
Cytoskeleton staining problems are often as much interpretive as technical. A bright image can still be a weak image if the structural meaning is uncertain, while a subtle but well-controlled signal may be analytically stronger. Effective troubleshooting therefore requires users to look beyond simple brightness and ask whether the signal actually supports the structural question the assay was designed to answer.
- Weak or discontinuous actin signal: Weak actin labeling may arise from poor fixation, over-permeabilization, insufficient probe exposure, low filament preservation, or cell states that genuinely reduce organized actin structure. Before concluding that actin has disappeared or collapsed, researchers should first confirm that the workflow preserved F-actin effectively.
- High background in phalloidin staining: Diffuse fluorescence can arise from incomplete washing, non-ideal probe concentration, residual sample-associated fluorescence, or imaging settings that overemphasize low-level background. In multicolor panels, background in the actin channel can also make cell boundaries appear broader or more irregular than they really are.
- Weak microtubule signal or loss of network clarity: Microtubule patterns may appear faint or incomplete if the staining method is not well matched to the sample, if the cells are stressed, or if acquisition settings do not capture the full dynamic range of the network. Structural clarity often improves more from workflow refinement than from simply increasing concentration.
- Live-cell probe interference with cell behavior: In live-cell cytoskeleton imaging, the stain itself can become part of the experimental problem if it alters cell health, morphology, division behavior, or dynamic structure. Researchers should evaluate whether a live-cell-compatible signal remains informative over the actual acquisition window rather than assuming that short-term visibility guarantees long-term interpretability.
- Misreading structural differences as direct biology: Apparent stress-fiber loss, cortical actin enhancement, spindle change, or network fragmentation may reflect real biology, but they may also reflect handling sensitivity, fixation artifacts, or uneven imaging balance. Strong conclusions require matched controls, a stable workflow, and a probe whose signal mechanism is consistent with the interpretation being made.
- Multicolor competition that weakens the cytoskeleton channel: In complex panels, the cytoskeleton stain may become too dominant or too suppressed relative to other channels. This is often a panel-design issue rather than a probe-quality issue. Better channel allocation can improve interpretability more effectively than changing the stain itself.
Applications of Cytoskeleton Staining in Research Workflows
Cytoskeleton staining is widely used across cell-based research workflows because the cytoskeleton provides a direct structural layer for interpreting morphology, intracellular organization, and treatment-dependent phenotypic change. In fluorescence imaging, the actin and microtubule networks often help researchers move beyond simple signal presence and evaluate how the overall architecture of the cell is organized under defined conditions. This makes cytoskeleton staining valuable not only in basic visualization experiments, but also in workflow development, multicolor panel design, comparative analysis, and structure-aware image interpretation.
Cytoskeleton staining in cell morphology studies
One of the most common applications of cytoskeleton staining is the visualization of cell morphology. Actin filaments help define cell shape, spreading status, cortical organization, and stress fiber formation, while microtubules provide information about intracellular support and network arrangement. In morphology-oriented studies, cytoskeleton staining is often used to compare whether cells appear elongated, rounded, polarized, contracted, or reorganized after treatment or under different culture conditions. Because these structural differences can be visually subtle in brightfield imaging, fluorescent cytoskeleton labeling frequently provides a more interpretable and reproducible morphology readout.
Cytoskeleton staining in migration and adhesion workflows
Cytoskeleton staining is also highly valuable in migration- and adhesion-related workflows because actin remodeling and microtubule organization are closely tied to how cells interact with surfaces and reorganize during movement. In these applications, researchers often evaluate lamellipodia-associated actin structure, cell edge organization, stress fiber redistribution, or broader cytoskeletal asymmetry as part of migration-related interpretation. The staining result is especially useful when combined with membrane or nuclear reference channels, because this helps distinguish whether visible changes reflect true migration-associated remodeling or more general alterations in cell shape and attachment.
Cytoskeleton staining in mitosis and spindle analysis
Microtubule-focused cytoskeleton staining plays an important role in mitosis and spindle-related imaging because tubulin organization becomes especially informative during cell division. In these workflows, researchers may want to visualize spindle structure, assess whether microtubule organization appears continuous or disrupted, or compare the spatial relationship between mitotic architecture and other intracellular features. Even when the assay is not primarily about mitosis, the cytoskeleton channel can help identify dividing cells and separate mitotic morphology from non-mitotic structural variation. This improves interpretation in experiments where treatment or timing affects cell-cycle distribution and intracellular organization at the same time.
Cytoskeleton staining in multicolor phenotypic imaging
In multicolor phenotypic imaging, cytoskeleton staining often serves as the structural backbone of the entire panel. When combined with nuclear, membrane, Golgi, mitochondrial, ER, or lysosomal channels, the cytoskeleton layer helps users interpret how the broader intracellular map is arranged and whether multiple organelle-level changes occur in a coordinated way. This is especially useful in higher-content imaging workflows, where the cytoskeleton channel can improve segmentation, support morphology-aware classification, and add structural context to otherwise isolated fluorescent readouts. In that sense, cytoskeleton staining is not just one more marker. It is often the channel that makes the rest of the image analytically coherent.
How We Support Cytoskeleton Staining Workflows?
We support cytoskeleton staining projects with practical probe selection guidance, workflow-aware imaging planning, and flexible support for actin- and microtubule-centered assays. The goal is not simply to provide fluorescent reagents, but to help researchers build a staining strategy that matches the structural target, sample handling sequence, and interpretation needs of the experiment. Because cytoskeleton imaging often operates at the boundary between morphology analysis and broader phenotypic interpretation, a useful support model must address probe logic, spectral design, and workflow reproducibility together rather than treat them as separate decisions.
Actin and Microtubule Probe Selection Support
- Practical comparison of phalloidin conjugates, microfilament-oriented probes, and tubulin-focused staining tools according to structural target and sample type.
- Support for choosing among multiple spectral phalloidin options such as Phalloidin-FITC, BF 488 Phalloidin, BF 568 Phalloidin, BF 594 Phalloidin, and Phalloidin-TRITC for different panel designs.
- Guidance on when Tubulin Tracker Green or Microfilament Tracker Green is better aligned with the actual structure and readout goal of the assay.
- More structured selection support for users comparing direct structural stains with antibody-based or protein-based cytoskeleton labeling strategies.
Workflow Optimization for Fixed-Cell and Live-Cell Imaging
- Support for aligning staining method with fixation, permeabilization, incubation sequence, wash logic, and image acquisition timing.
- Practical troubleshooting for weak actin signal, high phalloidin background, poor microtubule continuity, or unstable live-cell structural readout.
- Better workflow design for assays that rely on cytoskeleton structure as a comparative endpoint rather than a purely descriptive image feature.
- Assistance with documenting workflow variables so the staining logic remains reproducible across projects, operators, or transferred assays.
Multicolor Panel Design for Cytoskeleton-Centered Imaging
- Support for integrating the cytoskeleton channel with nuclear, membrane, mitochondrial, Golgi, ER, lysosomal, or other intracellular signals without avoidable channel burden.
- Guidance on selecting blue, green, orange, red, or other spectral positions according to marker hierarchy and instrument configuration.
- More balanced planning for assays where the cytoskeleton channel must function as a structural framework rather than compete with the rest of the panel.
- Practical design support for morphology studies, migration analysis, phenotypic imaging, and multichannel fluorescence microscopy workflows.
Support for Morphology, Migration, and Phenotypic Analysis
- Useful support for studies comparing cytoskeletal remodeling across treatment groups, culture conditions, or cell states.
- Better alignment of staining strategy with morphology-focused readouts, adhesion-related imaging, and structure-aware phenotypic analysis.
- Assistance with selecting probe types that preserve interpretability when cytoskeleton staining is part of a larger multiparameter workflow.
- Related fluorescence resources may also connect with fluorescent protein services, fluorescent nanoparticle services, fluorescent bead services, high-throughput screening, and molecular imaging.
Do You Need A Consultation?
BOC Sciences integrates cutting-edge fluorescence technologies to accelerate your research, driving next-generation solutions for drug discovery and diagnostics.
Transform Your Studies with Cutting-Edge Fluorescent Products
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A16-0004 | Phalloidin-FITC | 915026-99-2 | Bulk Inquiry |
| A16-0113 | Tubulin Tracker Green | N/A | Bulk Inquiry |
| A16-0125 | Sulforhodamine 101-X Phalloidin | N/A | Bulk Inquiry |
| A16-0100 | BF 350 Phalloidin | N/A | Bulk Inquiry |
| A16-0102 | BF 488 Phalloidin | N/A | Bulk Inquiry |
| A16-0106 | BF 568 Phalloidin | N/A | Bulk Inquiry |
| A16-0111 | BF 594 Phalloidin | 330626-83-0 | Bulk Inquiry |
| A16-0138 | Biotin-XX phalloidin | N/A | Bulk Inquiry |
| A16-0003 | Phalloidin-TFAX 488 | 289620-19-5 | Bulk Inquiry |
| A16-0002 | Phalloidin-TRITC | 915013-10-4 | Bulk Inquiry |
| A16-0202 | Microfilament Tracker Green | N/A | Bulk Inquiry |
High-Performance Fluorescent Tools for Your Research
- Cell Proliferation Tracer Fluorescent Probes Long-term tracking of cell division processes.
- Mitochondrial Fluorescent Probes Targeted dyes for mitochondrial structure and function.
- Endoplasmic Reticulum Fluorescent Probes ER-targeted dyes for organelle structure analysis.
- Cell membrane Fluorescent Probes Surface-labeling dyes for membrane dynamics studies.
- Other Cell Fluorescent Probes Functional probes for diverse cellular imaging studies.
- Ion Fluorescent Probes Indicators for real-time ion concentration imaging.
- Nuclear Fluorescent Probes DNA-binding dyes for nucleus visualization.
- pH Indicators Fluorescent sensors for intracellular pH monitoring.
Explore More Topics
Online Inquiry


