Hoechst 33342 Fluorescent Staining Protocols: Live Cell Imaging Dye
Hoechst 33342 is a widely used fluorescent dye for staining cell nuclei in both live and fixed cells. Its ability to selectively bind to DNA allows researchers to visualize nuclear morphology, assess cell cycle status, and track cellular dynamics with high specificity. This versatile dye is compatible with various imaging techniques, including confocal and conventional fluorescence microscopy, as well as flow cytometry. In this article, we provide detailed protocols and practical tips for optimizing Hoechst 33342 staining, helping researchers achieve clear, bright nuclear fluorescence while minimizing background signals. Whether studying mammalian cells, microbial samples, or tissue sections, these protocols ensure reliable and reproducible results for accurate cellular analysis.
Molecular Structure of Hoechst 33342
Hoechst 33342 belongs to the bis-benzimidazole class of small molecule dyes, with the chemical name 2′-(4-ethoxyphenyl)-5-(4-methyl-1-piperazinyl)-2,5′-di(1H-benzimidazol) trihydrochloride trihydrate. Its molecular formula is C₂₇H₂₈N₆O·3HCl·3H₂O, with a molecular weight of approximately 615.99 (as trihydrochloride trihydrate). The molecule consists of two aromatic benzimidazole rings connected by covalent bonds and carries a piperazine cationic group, enabling electrostatic interaction with the DNA phosphate backbone. Structurally, Hoechst 33342 is very similar to its analogue Hoechst 33258, except that Hoechst 33342 contains an ethoxy substituent where Hoechst 33258 has a hydroxyl group. This subtle structural modification significantly enhances the lipophilicity and cell membrane permeability of Hoechst 33342. The dye binds to the minor groove of DNA, with its rigid planar aromatic structure complementing the shape of AT base pairs, and forms hydrogen bonds and electrostatic interactions with the bases and phosphate backbone.
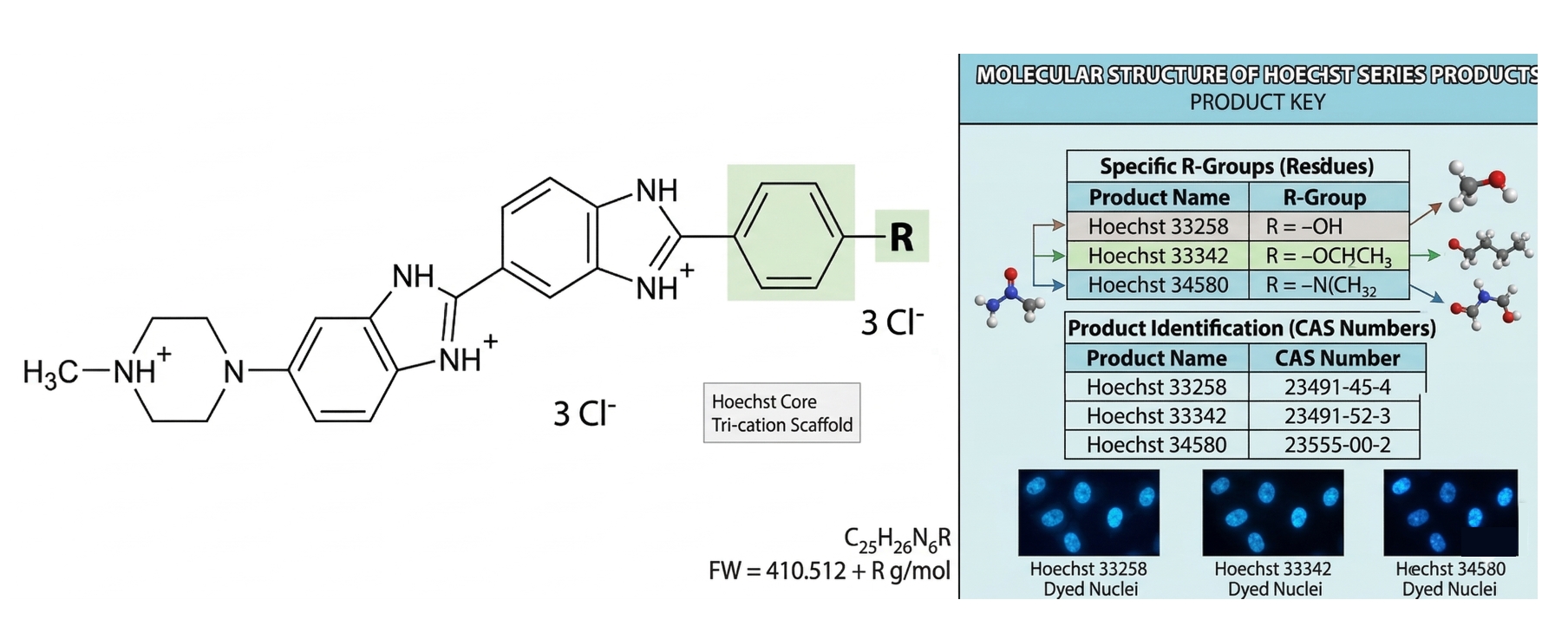 Fig. 1. A detailed structural guide to the Hoechst series of nuclear dyes (BOC Sciences Authorized).
Fig. 1. A detailed structural guide to the Hoechst series of nuclear dyes (BOC Sciences Authorized).
Development History of Hoechst 33342
The Hoechst series of dyes was developed in the early 1970s by Hoechst AG in Germany as blue fluorescent bis-benzimidazole compounds for DNA staining. The compounds were numbered sequentially according to their synthesis, with Hoechst 33342 being the 33,342nd compound synthesized. Hoechst 33258 and 33342 are the most commonly used dyes in this series. In 1975, Latt et al. first applied Hoechst 33258 in cytogenetic studies to detect cellular DNA replication. Subsequently, derivatives like Hoechst 33342 were developed to improve live-cell staining efficiency. The additional ethoxy group increased hydrophobicity, making Hoechst 33342 approximately ten times more cell-permeable than Hoechst 33258, making it suitable for live-cell nuclear staining. Over the years, Hoechst 33258/33342 dyes have been widely used in life sciences research, with ongoing improvements and new applications.
Entering the 21st century, researchers explored strategies to combine the Hoechst structure with other chromophores to extend its spectral properties, such as linking Hoechst 33258 with near-infrared dyes to create NIR-emitting DNA probes. Even after more than 45 years since its introduction, Hoechst 33342 and 33258 remain valuable tools in research and diagnostics. Although newer dyes with superior photophysical properties may gradually replace them in imaging, the Hoechst scaffold continues to serve as a key framework for constructing complex nucleic acid sensors and probes.
Mechanism of Action of Hoechst 33342 Staining
Hoechst 33342 functions by specifically binding to the minor groove of DNA. In aqueous solution, the dye itself has weak fluorescence, but upon insertion into AT-rich double-stranded DNA minor grooves, its fluorescence intensity dramatically increases and undergoes a blue shift. Free Hoechst 33342 has an excitation peak around 340–346 nm and an emission peak near 500 nm (weak cyan-green fluorescence), whereas DNA-bound dye exhibits an excitation peak ≈350 nm and a sharpened emission peak at 461 nm, producing bright blue fluorescence. This enhancement results from increased rigidity and hydrophobicity of the dye within the DNA environment, improving its quantum yield.
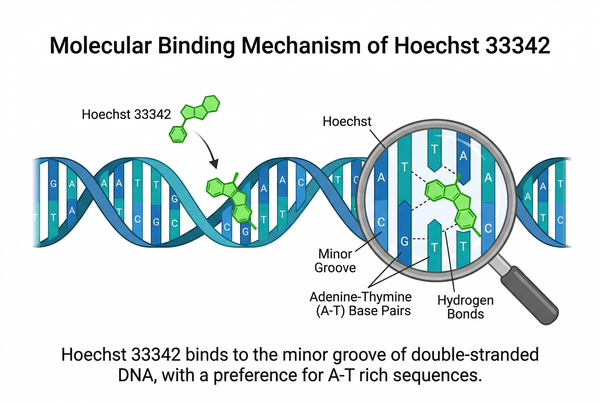 Fig. 2. Schematic of Hoechst 33342 minor groove binding to DNA A-T sequences (BOC Sciences Authorized).
Fig. 2. Schematic of Hoechst 33342 minor groove binding to DNA A-T sequences (BOC Sciences Authorized).
Hoechst 33342 exhibits sequence selectivity, preferentially binding to AT-rich regions of double-stranded DNA. Studies indicate that Hoechst 33258/33342 display two binding modes in the minor groove of B-DNA: a high-affinity mode with a dissociation constant of 1–10 nM, corresponding to specific binding to AT-rich sequences, and a low-affinity mode corresponding to non-specific electrostatic interactions with the DNA backbone. The optimal binding sequence is a consecutive AAA/TTT trinucleotide. Incorporation of bromodeoxyuridine (BrdU) in place of thymine disrupts the minor groove, quenching Hoechst fluorescence, a property commonly used to detect DNA replication. The more BrdU incorporated into newly synthesized DNA, the weaker the Hoechst signal, allowing assessment of S-phase progression.
As a non-intercalating minor groove ligand, Hoechst 33342 does not slide between base pairs like ethidium bromide but binds along the groove surface, minimally disturbing DNA secondary structure. However, because it acts via DNA binding, it may impact cellular processes, potentially slowing replication forks or triggering DNA damage responses, and is considered potentially mutagenic and carcinogenic, requiring careful handling.
Another key feature of Hoechst 33342 is its cell permeability. Unlike dyes that require fixation or membrane permeabilization (e.g., DAPI), Hoechst 33342 can freely cross live cell membranes, enabling staining of live cell nuclei. Its small size and relatively lipophilic ethoxy substituent facilitate this property. Observations show that Hoechst 33342 is significantly less toxic to live cells than DAPI, allowing high survival rates at typical staining concentrations. Cells generally remain viable and continue proliferating after staining. However, uptake varies across cell types; many stem cells and cancer cells express high levels of ABC transporters that actively efflux Hoechst 33342, reducing staining intensity.
Hoechst 33342 Staining Procedure
Experimental Steps
Confocal Microscopy Staining
- Prepare Staining Solution
Dilute Hoechst 33342 stock solution (typically 1 mg/mL in water or DMSO) with an appropriate buffer (PBS or culture medium) to a working concentration of 0.5–10 µg/mL. - Add Dye
- Adherent cells: Remove culture medium and add staining solution to cover cells.
- Suspension cells: Add at least three times the volume of cell suspension and mix thoroughly.
- Incubation
- Live cells: Incubate at 37°C in the dark for 10–20 minutes.
- Fixed cells or tissue sections: Incubate at room temperature for 5–15 minutes.
- Washing (Optional)
- Direct observation is possible as unbound dye has low background fluorescence.
- To reduce non-specific background, gently wash 2–3 times with PBS or medium to stop staining and enhance imaging clarity.
- Protect from Photobleaching
Keep stained samples protected from light at all times. - Imaging
- Use appropriate excitation sources (e.g., 405 nm or 351 nm laser) and the DAPI channel on confocal microscopy.
- Stained nuclei exhibit bright blue fluorescence and can be clearly distinguished from other channels.
Other Imaging and Analysis Applications
- Conventional Fluorescence Microscopy
Similar procedure to confocal microscopy; excitation via mercury or xenon UV lamps and observation through a DAPI filter. - Flow Cytometry
- Incubate cells (live cells applicable) in 5–10 µg/mL Hoechst 33342 at 37°C for 30–60 minutes.
- Terminate staining at 4°C and analyze blue fluorescence distribution to evaluate DNA content or identify side populations.
- DNAse or RNAse is often added to ensure the signal mainly derives from DNA.
- Microbial Nucleic Acid Staining
- Can stain bacteria and yeast.
- Live or dead microbes are typically stained at 12–15 µg/mL in PBS or saline for ~30 minutes.
- Yeast and other eukaryotic microbes exhibit weaker staining due to cell wall barriers and dye efflux.
- Experimental Optimization
Adjust working concentration and staining time according to sample type to achieve optimal signal-to-noise ratio.
Precautions
- Phototoxicity and Safety: Since Hoechst 33342 requires UV excitation, excessive or prolonged UV exposure can cause photodamage to live cells (phototoxicity), especially when the dye is bound to DNA, which may exacerbate UV-induced DNA damage. Although Hoechst 33342 is less toxic than DAPI and typically results in high post-staining cell viability, continuous UV excitation and DNA binding can still interfere with DNA replication and cell division. Therefore, during live-cell imaging experiments, it is recommended to minimize excitation intensity and exposure time. When necessary, intermittent illumination or using a 405 nm weak excitation light instead of deep UV can reduce cellular impact. For laboratory personnel, Hoechst 33342 poses potential hazards (possible mutagenicity); appropriate protective measures—such as gloves, lab coats, and avoiding direct skin contact—should be followed, and dye waste should be properly disposed of.
- Photobleaching and Fluorescence Stability: Like most fluorescent dyes, Hoechst 33342 fluorescence gradually diminishes over time. In unfixed samples, the dye may be gradually effluxed by cells or diluted during metabolism, leading to signal weakening. Fixed samples remain relatively stable after mounting, but photobleaching can still occur. Strong UV exposure may also induce photoconversion of Hoechst 33342, generating green- or red-emitting products. While this phenomenon can be exploited in single-molecule localization microscopy for super-resolution imaging, it may introduce background signals in conventional confocal imaging. Therefore, imaging is recommended on the same day as staining, avoiding prolonged storage or repeated exposure. For longer-term observation or storage, anti-fade mounting media can be used to slow fluorescence decay. During imaging, unnecessary intense illumination should be avoided, minimal effective excitation intensity should be used, and excitation light should be promptly turned off. Samples and unused staining solution should always be protected from light to prevent premature photodegradation.
- Effects on Live Cells: Hoechst 33342 can stain live cells, but dye concentration and incubation time may affect cell physiology. It is generally recommended to use the lowest possible dye concentration and shortest incubation time that provide detectable signal, minimizing interference with cellular processes. High concentrations (e.g., >10 µg/mL) or prolonged incubation (>1 hour) may increase cell stress, cause cell cycle arrest, or induce apoptosis. Pre-experiments should optimize staining conditions according to cell type. In long-term live-cell imaging (e.g., time-lapse studies), continued presence of Hoechst may affect proliferation, and fluorescence is diluted during cell division. In such cases, milder nuclear dyes, such as fluorescent proteins tagged with nuclear localization sequences, may be considered. Although Hoechst 33342 is considered low-toxicity, careful experimental design is essential to balance staining efficiency and cell health.
- Compatibility with Other Dyes: The spectral properties of Hoechst 33342 make it compatible with other fluorescent dyes. Its excitation and emission are in the UV/blue range, generally avoiding overlap with common green or red fluorophores. In multiplex experiments, Hoechst can stain nuclei while FITC (green) and Texas Red or Cy5 (red) label other targets, with minimal signal interference. Avoid using two nucleic acid dyes simultaneously (e.g., Hoechst and DAPI or PI), as they may compete for binding sites or cause Förster resonance energy transfer, reducing staining intensity. Hoechst may partially overlap DNA binding sites with intercalating dyes like ethidium bromide or 7-AAD; sequential staining or stepwise protocols are recommended. For experiments labeling both DNA and RNA, Hoechst primarily stains double-stranded DNA and has weak RNA staining, so RNA-specific dyes may be used additionally. In BrdU incorporation assays, the quenching effect of BrdU on Hoechst fluorescence should be considered when analyzing results.
Limitations
- Cell Type and Status Limitations: Hoechst 33342 staining efficiency varies among cell types. It penetrates eukaryotic nuclei readily but is less efficient in organisms with thick cell walls (e.g., yeast, plant spores), often requiring higher concentrations and longer incubation while still yielding weaker signals. In mixed live and dead cell samples, Hoechst preferentially stains dead cells due to increased membrane permeability, resulting in stronger nuclear fluorescence from dead cells and potential overestimation of total DNA content or false positives in flow cytometry or imaging. In live-cell experiments, membrane integrity dyes (e.g., PI or 7-AAD) should be used to distinguish dead cell contributions. Additionally, some stem cells or cancer cells actively efflux Hoechst, reducing signal; careful interpretation and, if necessary, inhibition of efflux pumps may be required to validate results.
- Long-Term Imaging and Stability: Hoechst 33342 is best suited for endpoint staining (immediate observation or analysis). In long-term imaging (hours to days), signal stability and cellular interference are limiting factors. Live cells gradually efflux or dilute the dye, weakening fluorescence. Even fixed samples should not be exposed to ambient light for extended periods, as significant photobleaching occurs. Prolonged dye exposure and repeated UV illumination may alter cell states. Therefore, Hoechst is generally used for staining cells or tissues immediately before observation, and is not ideal for long-term continuous monitoring. For long-term nuclear tracking, lower-toxicity dyes requiring visible light excitation (e.g., Syto series) or genetically encoded nuclear markers are recommended.
- Limitations in Thick Tissue and In Vivo Applications: Hoechst 33342 has limited penetration in thick tissue sections or intact tissues (e.g., organoids, embryos). UV excitation is strongly attenuated and tissue autofluorescence is high, making deep nuclei difficult to stain or image effectively. Hoechst is better suited for thin tissue sections (e.g., frozen or paraffin sections after antigen retrieval) or cleared samples. In vivo applications are generally avoided due to DNA-binding properties and potential genotoxicity, although ex vivo staining of isolated cells or tissues is possible. Reports exist of using Hoechst in live cells (in vitro) for sperm sorting, but safety risks must be weighed and strict ethical and protective measures followed.
Applications of Hoechst 33342 Staining
Cell Imaging
Hoechst 33342's membrane permeability and DNA specificity make it a common nuclear probe in cell and molecular biology. In fluorescence microscopy (including confocal), it serves as a nuclear counterstain, facilitating multiplex experiments. Upon UV laser excitation (345–355 nm line or 405 nm laser), DNA-bound Hoechst emits bright blue fluorescence detectable through DAPI filter sets. Large Stokes shift minimizes spectral overlap with green or red fluorophores, enabling combination with GFP, RFP, or other visible-light probes for nuclear counting and localization.
Cell Cycle Analysis
Hoechst 33342 quantitatively stains DNA, allowing cell cycle distribution analysis and cell sorting. In flow cytometry, cells stained with Hoechst exhibit fluorescence differences based on DNA content (2N, 4N, etc.), enabling determination of cell cycle phase proportions or sorting by DNA content. Similarly, confocal imaging with fluorescence intensity analysis can provide approximate DNA quantification and mitotic chromosome observation.
Apoptosis Studies
Hoechst 33342 is widely used to detect nuclear changes during apoptosis. Fluorescence microscopy reveals chromatin condensation and nuclear fragmentation into apoptotic bodies: normal nuclei stain uniformly, early apoptotic nuclei appear bright and condensed, and late apoptotic nuclei show highly fragmented fluorescence. Due to its high affinity for apoptotic nuclei, fluorescence from apoptotic cells is often stronger than in viable cells. Combined with Annexin V (phosphatidylserine) or membrane-impermeable dyes like PI, Hoechst enables discrimination of live, early apoptotic, and late apoptotic/dead cells in flow cytometry.
Stem Cell and Side Population Analysis
Hoechst 33342 is used to identify "side population" (SP) cells in stem cell research. Some stem and cancer stem cells highly express ATP-binding cassette (ABC) transporters that actively efflux Hoechst. After staining, flow cytometry with dual-wavelength detection (blue and red emission) reveals a small low-fluorescence SP, which disappears when efflux inhibitors like verapamil are applied, confirming cell identity. This method, originally established in mouse bone marrow hematopoietic stem cells, is now widely used for various tissue and cancer stem cell studies.
Fluorescent Dyes Recommended for Your Research Project
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A19-0051 | Ortho-iodoHoechst 33258 | 158013-41-3 | Bulk Inquiry |
| A19-0053 | Para-iodoHoechst 33258 | 158013-43-5 | Bulk Inquiry |
| A19-0052 | Meta-iodoHoechst 33258 | 158013-42-4 | Bulk Inquiry |
| A19-0060 | Hoechst 34580 | 23555-00-2 | Bulk Inquiry |
| A19-0046 | Hoechst 33342 analog 2 | 106050-84-4 | Bulk Inquiry |
| A19-0056 | Hoechst 33258 analog 2 | 23491-54-5 | Bulk Inquiry |
| A19-0058 | Hoechst 33258 analog 3 | 23554-98-5 | Bulk Inquiry |
| A19-0057 | Hoechst 33258 analog 5 | 23491-55-6 | Bulk Inquiry |
| A19-0047 | Hoechst 33258 Analog 6 | 129244-66-2 | Bulk Inquiry |
| A19-0041 | Hoechst 33258 | 23491-45-4 | Bulk Inquiry |
Corporate Profile
BOC Sciences is a leading independent supplier of high-quality fluorescent chemicals, dyes, and fluorescent probes, dedicated to supporting advanced life science research. With decades of expertise in chemical synthesis, molecular labeling, and analytical services, we provide reliable reagents for applications ranging from live-cell imaging to flow cytometry and tissue analysis. Our mission is to empower researchers with precise, reproducible, and application-ready fluorescent reagents, such as Hoechst 33342, that enable accurate cellular and molecular studies.
Our capabilities include a comprehensive portfolio of fluorescent dyes, custom synthesis and modification services, and rigorous quality control to ensure purity, fluorescence consistency, and minimal background interference. We provide expert guidance on staining protocols, imaging optimization, and multi-color labeling strategies, helping researchers achieve clear, high-contrast results. BOC Sciences also supports advanced applications, including high-throughput assays, side-population analysis, and complex multi-channel imaging experiments.
Committed to scientific excellence and innovation, BOC Sciences combines chemical expertise with practical, application-focused support. By delivering dependable reagents and tailored solutions, we enable laboratories to enhance experimental accuracy, sensitivity, and reproducibility. Our focus on quality, technical know-how, and customer collaboration makes us a trusted partner for researchers in cell biology, molecular diagnostics, and fluorescence-based investigations.
Custom Fluorescent Solutions Designed for Your Experiments
- DNA StainingPrecise fluorescent dyes for clear DNA visualization and analysis.
- Lipid StainingFluorescent solutions for effective lipid structure imaging.
- Cell StainingAdvanced staining for detailed cell morphology and analysis.
- Protein StainingHigh-quality staining for accurate protein detection and imaging.
- Bacteria ImagingFluorescent solutions to visualize bacterial structures and activity.
- Cell ImagingVisualize and analyze live or fixed cells using advanced fluorescence.
- Molecular ImagingCutting-edge fluorescent solutions for deep molecular analysis.
- Fluorescence ImagingHigh-resolution imaging solutions for detailed fluorescence studies.
- BioconjugationCustom bioconjugation services for protein, peptide, and dye linking.
- Drug DeliveryTailored fluorescent solutions for efficient drug delivery research.
- Molecular DiagnosticsFluorescent probes and markers for precise molecular diagnostics.
- Flow CytometryFluorescent dyes and reagents for enhanced flow cytometry analysis.
High-Performance Fluorescent Tools for Your Research
- Nuclear Fluorescent Probes DNA-binding dyes for nucleus visualization.
- Lysosomal Fluorescent Probes Acidic organelle markers for lysosome tracking.
- Cell Proliferation Tracer Fluorescent Probes Long-term tracking of cell division processes.
- Golgi Fluorescent Probes Targeted probes for Golgi apparatus visualization.
- Lipid Fluorescent Probes Dyes for lipid droplets and membranes.
- Cytoskeleton Fluorescent Probes Probes for actin and microtubule visualization.
- Endoplasmic Reticulum Fluorescent Probes ER-targeted dyes for organelle structure analysis.
- Calcium, Chloride and Other indicators Fluorescent indicators for intracellular ion flux monitoring.
Explore More Topics
Online Inquiry


