DAPI Fluorescent Staining Protocols
DAPI is a blue fluorescent dye that binds strongly to DNA, commonly used for nuclear staining. Its blue fluorescence contrasts sharply with other green, yellow, or red fluorescent proteins, making it widely used in multicolor fluorescence applications. DAPI can penetrate cell membranes, allowing staining of both live and fixed cells; however, live-cell staining requires precise control of DAPI concentration, so it is mainly used for fixed-cell staining.
Molecular Structure of DAPI
The chemical name of DAPI is 4',6-diamidino-2-phenylindole, consisting of a nitrogen-containing indole ring fused to a phenyl ring, with amidine (–C(=NH)NH₂) functional groups attached at the 4-position of the phenyl ring and the 6-position of the indole ring. These amidine groups confer a positive charge under physiological conditions, and DAPI is usually supplied as a dihydrochloride salt (molecular formula C₁₆H₁₅N₅·2HCl). The rigid planar aromatic structure and double positive charge of DAPI facilitate its strong binding to the DNA double helix.
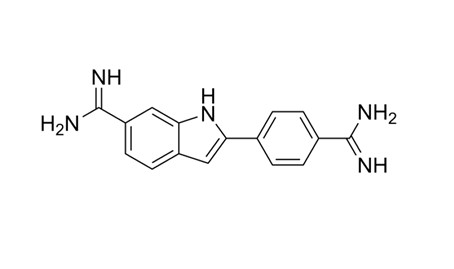 Fig. 1. DAPI structure (BOC Sciences Authorized).
Fig. 1. DAPI structure (BOC Sciences Authorized).
History of DAPI
DAPI was first synthesized in 1971 in the laboratory of German chemist Otto Dann, initially intended as a potential drug against trypanosomiasis. Although it was not successful as a drug, researchers discovered that DAPI binds DNA effectively and exhibits enhanced fluorescence upon binding. This property led to its first use as a fluorescent dye in 1975, labeling mitochondrial DNA in ultracentrifugation experiments. By the late 1970s, DAPI became widely adopted for fluorescent staining of DNA in various cell types. Studies demonstrated its applicability for detecting DNA in plant, microbial, multicellular animal, and bacterial cells. By 1977, DAPI was also confirmed suitable for quantitative measurements of cellular DNA content and was applied in flow cytometry. Its strong DNA fluorescence has made DAPI one of the most commonly used nucleic acid probes and dyes in laboratories for decades.
Principle of DAPI Staining
DAPI is a DNA-specific probe that binds to the minor groove of the DNA double helix. Its elongated, planar structure with a positive charge allows it to fit into the minor groove, preferentially targeting A-T-rich regions. These narrow, negatively charged grooves facilitate stable binding through hydrogen bonds and hydrophobic interactions. Each DAPI molecule occupies approximately 2–3 base pairs. Free in solution, DAPI exhibits weak fluorescence; upon intercalating into DNA, its conformational freedom decreases and the local polarity drops, resulting in enhanced fluorescence. Binding to DNA can increase fluorescence intensity roughly 20-fold, producing bright blue fluorescence.
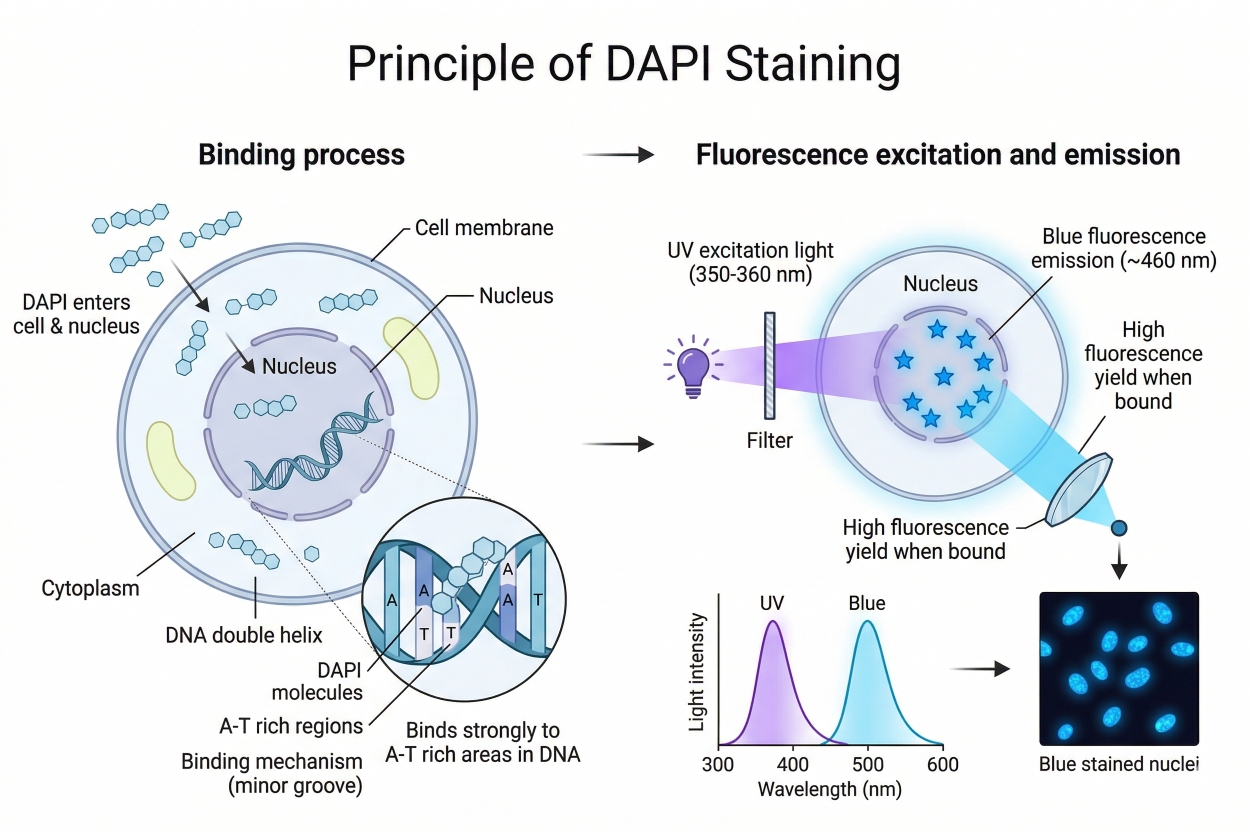 Fig. 2. Schematic illustrating the molecular principle of DAPI nuclear staining (BOC Sciences Authorized).
Fig. 2. Schematic illustrating the molecular principle of DAPI nuclear staining (BOC Sciences Authorized).
It is noteworthy that DAPI exhibits varying fluorescence effects depending on the nucleic acid. It binds most tightly and fluoresces most strongly with A-T-rich double-stranded DNA. Binding to G-C-rich regions occurs differently and results in little or no fluorescence enhancement. DAPI can also bind RNA, but because RNA is often single-stranded or partially double-stranded with different base composition, DAPI produces only slightly increased fluorescence (~5-fold) with a shift toward cyan-green emission. Therefore, in routine applications, DAPI blue fluorescence is considered a specific indicator of double-stranded DNA, primarily nuclear DNA.
DAPI Staining Procedure
Experimental Steps
In the laboratory, DAPI is commonly used to stain cultured cells or tissue sections to label nuclei. A typical protocol includes the following steps:
- Sample Preparation — Choose appropriate fixation and permeabilization methods. For cultured cells, fix with paraformaldehyde (e.g., 4% PFA) and optionally permeabilize with 0.1% Triton X-100 to allow DAPI entry into nuclei. If immunofluorescence staining is also required, perform antibody staining first and apply DAPI at the final step to avoid fluorescence interference. Wash fixed cells or tissue with PBS before staining to remove residual fixative.
- Staining Solution Preparation — Dilute DAPI stock solution to working concentration using PBS or cell culture medium. Typical working concentrations range from 0.1–1 μg/mL (~0.3–3 μM), with 0.5 μg/mL often sufficient. Ready-to-use solutions (e.g., 300 nM, ~0.1 μg/mL) can be used directly. Prepare solutions in light-protected containers, as DAPI is light-sensitive.
- Incubation — Apply sufficient DAPI solution to fully cover the sample (e.g., cells on a coverslip or tissue section). Incubate in the dark at room temperature for 5–10 minutes. Thin cell monolayers may only need 5 minutes, while thicker tissue sections may require 10–15 minutes for full penetration. Avoid direct bright light during incubation to prevent photobleaching.
- Washing — Carefully remove excess DAPI and wash the sample 2–3 times with PBS or deionized water, a few minutes each, to reduce background fluorescence. For suspended cells, centrifuge and resuspend in fresh PBS for washes. Keep samples protected from light.
- Mounting — For adherent samples, remove excess liquid, add a drop of anti-fade mounting medium, and cover with a coverslip. Anti-fade agents (e.g., DABCO or PPD) help preserve DAPI fluorescence. Temporary mounting with glycerol: PBS (e.g., 90% glycerol) is possible, but long-term storage requires proper anti-fade media. Avoid bubbles and ensure full coverage of the sample. Store samples protected from light and observe promptly.
- Microscopy — Use a fluorescence or confocal microscope with appropriate excitation/emission settings. DAPI-bound DNA has maximum excitation at ~358 nm (UV) and emission at ~461 nm (blue). Standard fluorescence filters (excitation ~340–365 nm, emission ~420–480 nm) or confocal 405 nm laser with ~450/50 nm emission detection can be used. Adjust exposure or laser intensity to avoid underexposure or saturation. Images can be recorded as needed.
Precautions
- Avoid Photobleaching — DAPI fluorescence decays under UV; minimize strong light exposure, and capture images promptly.
- Reduce Background — Free DAPI can produce non-specific fluorescence. Adequate washing and proper dye concentration minimize background. RNase treatment can remove RNA-derived fluorescence.
- Safety — DAPI binds nucleic acids and may be mutagenic or toxic. Wear gloves and avoid skin contact. In case of contact, rinse with plenty of water. Dispose of DAPI-containing waste according to hazardous chemical regulations.
Limitations of DAPI Staining
Although DAPI is a highly useful nucleic acid probe, there are certain limitations and precautions to consider:
- Limitations for Live-Cell Staining — At conventional concentrations, DAPI cannot efficiently penetrate intact cell membranes, so it is typically used for fixed cells or cells with compromised membranes. Using DAPI in live cells requires higher concentrations, sometimes reaching tens of micromolar, which offers limited effectiveness and can be cytotoxic. In contrast, blue nuclear dyes like Hoechst 33342 are more suitable for live-cell imaging due to better membrane permeability and lower toxicity. Therefore, DAPI is primarily used for fixed samples rather than long-term live-cell nuclear imaging. Conversely, in flow cytometry, DAPI can be employed to distinguish live from dead cells, as live cells exclude the dye while dead cells are stained, effectively taking advantage of its poor penetration into live cells.
- Staining Specificity and Sensitivity — DAPI exhibits sequence preferences, which is both an advantage and a limitation. It binds most strongly to A-T-rich regions in double-stranded DNA and produces the brightest fluorescence, whereas its binding to G-C-rich regions generates weaker fluorescence. In most eukaryotic cells, this does not significantly affect overall nuclear staining. However, in genomes with high G-C content, such as certain bacteria, DAPI staining may be relatively weaker, requiring higher concentrations or complementary probes. Additionally, DAPI can bind RNA and emit slightly different (greenish) fluorescence. In samples with abundant RNA—such as cytoplasm-rich regions or incompletely permeabilized cells—this may cause minor non-specific background, which can be reduced by RNase treatment after fixation.
- Cytotoxicity and Safety — As a minor-groove DNA-binding agent, DAPI carries potential toxic risks to both cells and laboratory personnel. High concentrations can be cytotoxic to live cells, causing cell death or chromosomal damage, so direct use on live cells is generally not recommended. In animal studies, large doses of DAPI have been shown to cause toxicity in mice (reported LD₅₀ ~37.3 mg/kg). DAPI is also considered potentially mutagenic and carcinogenic because it intercalates into DNA and may interfere with replication or induce mutations. Laboratory safety protocols must be strictly followed: avoid direct contact and inhalation, and dispose of waste as hazardous material. Although routine low-dose use does not usually cause immediate adverse effects, potential risks should always be kept in mind, minimizing unnecessary exposure.
- Spectral and Imaging Limitations — DAPI emits blue fluorescence, which generally distinguishes it well from common green fluorescent proteins (GFP) or dyes. However, its emission spectrum is relatively broad and includes a minor cyan-green tail. In multiplex fluorescence experiments with very strong green signals, spectral crosstalk correction may be needed to prevent misinterpretation of DAPI's weak green component. Overall, DAPI pairs well with green and red fluorophores, with minimal spectral overlap, rarely affecting most multi-label experiments. Additionally, DAPI requires UV excitation, and prolonged exposure or limited UV source intensity may cause sample photodamage. Modern confocal microscopes often use a 405 nm laser instead of UV to reduce phototoxicity. Photobleaching over time should also be considered, so anti-fade mounting media and prompt image acquisition are recommended.
Applications of DAPI Staining
DAPI has broad utility in life science research, particularly for labeling nucleic acids, especially cell nuclei:
Nuclear Fluorescence Staining
The most common use of DAPI is as a nuclear stain or counterstain in fluorescence microscopy. Its bright blue fluorescence makes it suitable for combination with green or red fluorescent probes for multicolor labeling without spectral interference. In immunofluorescence and tissue section staining, DAPI is often included in mounting media to counterstain nuclei, facilitating clear visualization of each cell nucleus under the microscope.
Cell Viability and Apoptosis Detection
Since DAPI cannot penetrate intact live-cell membranes at typical concentrations, it is often used as a dead-cell indicator in live-cell experiments. For example, in apoptosis assays, DAPI can distinguish live and dead cells—apoptotic or necrotic cells take up the dye and exhibit blue nuclear fluorescence, while healthy live cells remain unstained. This difference can be analyzed using fluorescence microscopy or flow cytometry to assess cell death rates.
Flow Cytometry and Cell Cycle Analysis
DAPI can stain fixed or ethanol-treated cell suspensions for flow cytometric quantification of nuclear DNA content, enabling discrimination of cell cycle stages (G₀/G₁, S, G₂/M). Fluorescence intensity correlates with DNA content, allowing researchers to perform cell cycle sorting and ploidy analysis. DAPI is also commonly used to detect sub-G₁ peaks indicative of apoptosis.
Detection of Microbial Contamination
In cell culture experiments, DAPI is frequently used to detect mycoplasma or viral contamination. Small contaminants in culture media are stained by DAPI and fluoresce blue under the microscope, allowing easy detection. Many laboratories regularly stain cell culture supernatants with DAPI to ensure cultures are free of mycoplasma.
Chromosome and Tissue Staining
DAPI can stain fixed nuclei and chromosome preparations. In genetics and cell biology, DAPI-stained metaphase chromosomes display bright banding patterns under fluorescence microscopy (DAPI banding), aiding in chromosome identification and detection of structural abnormalities. DAPI is also used as a nuclear counterstain in in situ hybridization and certain tissue sections to highlight nuclei. While it can stain DNA in agarose gels, specialized gel dyes such as ethidium bromide are more commonly used, making DAPI's gel applications less frequent.
Fluorescent Dyes Recommended for Your Research Project
| Catalog | Name | CAS | Inquiry |
|---|---|---|---|
| A19-0040 | Hoechst 33342 | 23491-52-3 | Bulk Inquiry |
| A19-0005 | Nuclear Yellow | 74681-68-8 | Bulk Inquiry |
| A16-0201 | DAPI dihydrochloride | 28718-90-3 | Bulk Inquiry |
| A19-0103 | SYBR Green I | 178918-96-2 | Bulk Inquiry |
| A19-0042 | Pyronin Y | 92-32-0 | Bulk Inquiry |
| A19-0101 | Propidium Iodide | 25535-16-4 | Bulk Inquiry |
| A19-0060 | Hoechst 34580 | 23555-00-2 | Bulk Inquiry |
| A19-0034 | DAPI dilactate | 28718-91-4 | Bulk Inquiry |
| A19-0102 | SYBR Green II | 195199-08-7 | Bulk Inquiry |
| A19-0041 | Hoechst 33258 | 23491-45-4 | Bulk Inquiry |
Corporate Profile
BOC Sciences is a premier independent supplier of high-quality fluorescent chemicals, fluorescent dyes, and nucleic acid probes, serving research laboratories worldwide. With decades of expertise in chemical synthesis and fluorescent probe development, we specialize in providing reagents such as DAPI, Hoechst dyes, and other DNA- and RNA-specific stains that deliver reliable, high-performance results. Our mission is to combine cutting-edge chemistry with rigorous quality standards to enable precise and reproducible outcomes in cell biology, molecular imaging, and fluorescence-based assays.
Our capabilities extend beyond standard reagent supply. BOC Sciences offers custom synthesis of fluorescent probes and conjugates, tailored to specific experimental requirements. Through structural modification, functional tagging, and optimization for brightness, photostability, and target specificity, we provide researchers with reagents that meet the highest scientific standards. In addition, our team of technical specialists supports clients with protocol optimization, dye selection guidance, and assay troubleshooting, ensuring maximal signal-to-noise performance across diverse cell types and experimental conditions. All products undergo stringent quality control to guarantee identity, purity, and stability, complying with international laboratory regulations for safe and reliable use.
As a trusted partner in fluorescence research, BOC Sciences continuously expands its portfolio to include emerging dyes, high-sensitivity probes, and next-generation staining reagents. We support both small-scale laboratory studies and larger preclinical programs with flexible supply solutions, helping scientists maintain continuity in their research. By bridging innovative chemistry with practical laboratory applications, BOC Sciences enables precise visualization, quantification, and analysis of biological systems. Researchers choosing BOC Sciences gain access to premium fluorescent reagents, expert technical support, and a partner dedicated to advancing reliability and excellence in fluorescence-based research.
Custom Fluorescent Solutions Designed for Your Experiments
- DNA StainingPrecise fluorescent dyes for clear DNA visualization and analysis.
- Lipid StainingFluorescent solutions for effective lipid structure imaging.
- Cell StainingAdvanced staining for detailed cell morphology and analysis.
- Protein StainingHigh-quality staining for accurate protein detection and imaging.
- Bacteria ImagingFluorescent solutions to visualize bacterial structures and activity.
- Cell ImagingVisualize and analyze live or fixed cells using advanced fluorescence.
- Molecular ImagingCutting-edge fluorescent solutions for deep molecular analysis.
- Fluorescence ImagingHigh-resolution imaging solutions for detailed fluorescence studies.
- BioconjugationCustom bioconjugation services for protein, peptide, and dye linking.
- Drug DeliveryTailored fluorescent solutions for efficient drug delivery research.
- Molecular DiagnosticsFluorescent probes and markers for precise molecular diagnostics.
- Flow CytometryFluorescent dyes and reagents for enhanced flow cytometry analysis.
High-Performance Fluorescent Tools for Your Research
- Endoplasmic Reticulum Fluorescent Probes ER-targeted dyes for organelle structure analysis.
- Cytoskeleton Fluorescent Probes Probes for actin and microtubule visualization.
- Calcium, Chloride and Other indicators Fluorescent indicators for intracellular ion flux monitoring.
- Ion Fluorescent Probes Indicators for real-time ion concentration imaging.
- Nuclear Fluorescent Probes DNA-binding dyes for nucleus visualization.
- Fluorescent Probes Versatile tools for biomolecular and cellular imaging.
- Metal Fluorescent Probes Selective sensors for intracellular metal ions.
- Apoptosis Fluorescent Probes Probes detecting programmed cell death events.
Explore More Topics
Online Inquiry


